

Background: Drug survival is a comprehensive outcome encompassing effectiveness, safety, and patient and physician preferences. It is particularly suitable for chronic diseases that require long-term treatment, such as systemic lupus erythematosus (SLE). The EULAR recommended that add-on treatment with belimumab should be considered in patients with SLE who do not respond to combinations of hydroxychloroquine and glucocorticoid (GC) with or without immunosuppressants. However, the best use of belimumab remains to be elucidated, and the situation has become more complicated since anifrolumab was approved for the treatment of extrarenal SLE in 2021.
Objectives: We aimed to investigate the drug survival of belimumab and explore the clinical parameters that predict belimumab retention in patients with SLE in a real-world setting.
Methods: This is a noninterventional, longitudinal, observational study. The clinical data of consecutive patients with SLE who were newly treated with belimumab from 2018 to 2023 at our institution were retrospectively reviewed. Patients initiated on belimumab within 1 year of data collection were excluded. Belimumab was administered at the discretion of the treating physician. Other treatments, including tapers for GCs, were also decided by the treating physicians. The observation period began on the first day of belimumab administration. The data were censored when belimumab was discontinued, the patient was transferred to another hospital, or the observation period ended (December 2024). Data on medication, serum anti-dsDNA antibody titers, and complement levels, the SLE Disease Activity Index 2000 (SLEDAI-2K), Physician Global Assessment (PGA), definition of remission in SLE (DORIS), lupus low disease activity state (LLDAS), and adverse events were collected upon initiation of belimumab, at 3, 6, and 12 months after initiation, and annually until the last visit. The primary outcome was the 1-year retention rate of belimumab. The secondary outcomes included the overall discontinuation rate of belimumab, reduction in the dosage of prednisolone (PSL)-equivalent GC, attainment of LLDAS and DORIS, and adverse events. No correction for multiple comparisons was made.
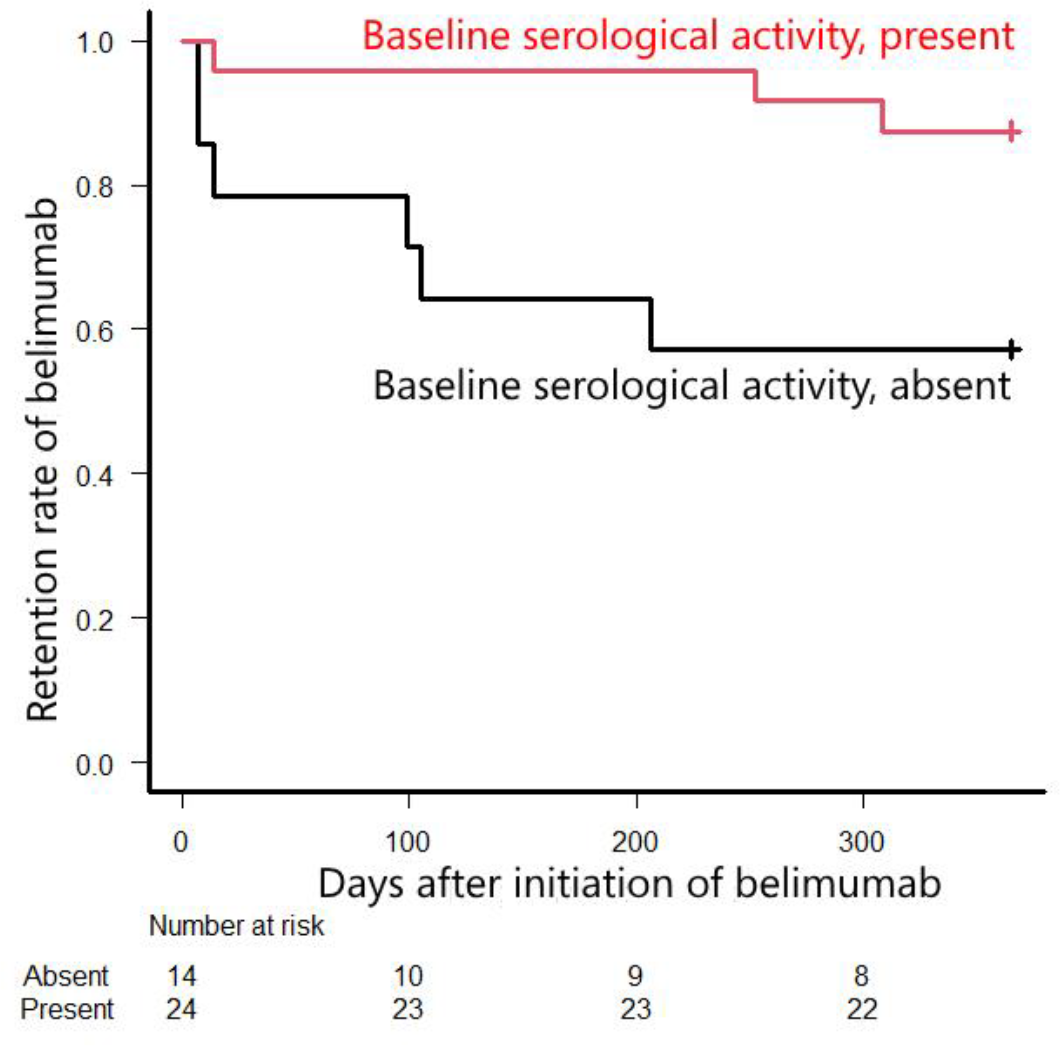
Results: All 38 consecutive patients with SLE who received belimumab during the study period were included. At baseline (at the time of belimumab initiation), 35 patients (92%) were female, the age was 18–68 years (median 42 years), and the disease duration was 0–37 years (median 16 years). At baseline, the dosage of PSL-equivalent GC was 4–28 mg/day (median 10 mg/day), and hydroxychloroquine and immunosuppressants were used concomitantly in 22 (58%) and 32 (84%) patients, respectively. Fifteen (39%) patients discontinued belimumab, and 4 patients discontinued the drug after only one infusion. The reasons for discontinuation were as follows: lack of efficacy in 10, patient preference in 4, and pregnancy in 1. Seven patients were switched to other biologics, including anifrolumab ( n = 5), among the 10 patients who discontinued belimumab because of its lack of efficacy. The overall discontinuation rate was 13.4 per 100 person-years. The 1-year drug retention rate of belimumab was 29/38 (76%). An absence of serological activity (increased anti-dsDNA and/or hypocomplementemia) at baseline was a significant risk factor for discontinuation of belimumab within 1 year ( p = 0.034), with a relative risk (95% CI) of 3.43 (1.01 to 11.6). Higher serum anti-dsDNA antibody titers and lower serum C3 levels at baseline were significantly associated with the 1-year drug retention of belimumab ( p = 0.045 and 0.021, respectively). The time-to-event analysis also indicated that serological activity at baseline was significantly associated with the 1-year drug retention of belimumab ( p = 0.022 by the log-rank test) (Figure 1). The following factors were not significantly associated with the 1-year drug retention of belimumab ( p >0.05 in all assessments): baseline SLEDAI-2K, PGA, PSL-equivalent GC dosage, and concomitant use of immunosuppressants or hydroxychloroquine; attainment of LLDAS at 3 or 6 months; decrease in SLEDAI-2K scores, serum anti-dsDNA antibody titers, and PSL-equivalent GC dosages at 3 or 6 months; and increase in serum C3 or C4 levels at 3 or 6 months. The GC dosage was significantly reduced among the 30 patients who used belimumab ≥12 months ( p <0.001). In 20 (67%) patients, the PSL-equivalent GC dosage was reduced, with a median reduction of 3.0 mg at 1 year after the initiation of belimumab. Among the 33 patients who were not in LLDAS at baseline, 19 (58%) patients were in LLDAS at their last visit ( p <0.001). 9 and 11 patients newly attained LLDAS at 12 months and thereafter, respectively: 1 patient, who attained LLDAS at 12 months, was not in LLDAS at her last visit. Although no patient was in DORIS at baseline, 12 patients were in DORIS at their last visit. Six patients temporarily discontinued belimumab because of infection, herpes zoster in 4, and other infections in 2; however, all of these patients were restarted on belimumab and continued until the last visit. No other serious adverse events or adverse events resulting in permanent discontinuation of belimumab were observed.
Conclusion: This study showed that the retention rate of belimumab was high among patients with SLE in real-world settings; however, an absence of serological activity upon initiation of belimumab was a significant risk factor for discontinuing belimumab within 1 year. Belimumab was well tolerated and demonstrated a GC-sparing effect.
REFERENCES: NIL.
Acknowledgements: We thank all the physicians who treated the enrolled patients for their engagement in data collection.
Disclosure of Interests: Natsuko Hara: None declared, Yasuhiro Katsumata received speaker fees from Asahi Kasei Pharma, AstraZeneca K.K., Ayumi Pharmaceutical Co., Chugai Pharmaceutical Co., Ltd., Eli Lilly Japan K.K., GlaxoSmithKline K.K., Janssen Pharmaceutical K.K., Mitsubishi Tanabe Pharma Corporation, Otsuka Pharmaceutical Co., Ltd., Pfizer Japan Inc., Taiho Pharmaceutical Co., Ltd., and Taisho Pharmaceutical Co., Ltd.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (