

Background: Recently, the disease activity of rheumatoid arthritis (RA) was improved due to the ‘treat-to-target’ strategy. However, some patients remain various symptoms despite recommended treatment was performed. Then, the term of ‘difficult-to-treat RA (D2TRA)’ is widely recognized. Inability to taper glucocorticoids (below 7.5 mg/day prednisone or equivalent) was included in the EULAR definition of D2TRA. However, there are few reports on the effects of glucocorticoid with D2TRA patients.
Objectives: The aim of this study was to examine the effects of glucocorticoid use on the course of treatment in patients with D2TRA. In addition, we investigated the risk factor of inability to taper glucocorticoid in patients with D2TRA.
Methods: This study used the multicenter database included 673 RA patients treated with biological disease-modifying antirheumatic drugs (bDMARDs)/Janus kinase inhibitors (JAKi). In these 673 patients, 137 patients met D2TRA criteria (tocilizumab: n=21, sarilumab: n=10, abatacept: n=31, tofacitinib: n=30, baricitinib: n=30, Upadacitinib: n=10, peficitinib: n=4, filgotinib: n=1). The subjects were divided into glucocorticoid use group and glucocorticoid non-use group. Disease activity change of each group was evaluated at 24 weeks after bDMARDs/JAKi switching. In glucocorticoid use group, reduction in glucocorticoids was also investigated, and multivariate logistic regression analysis was performed to clarify the risk factors of inability to taper glucocorticoid in patients with D2TRA. Changes in disease activity score based on 28 joints count and erythrocyte sedimentation rate (DAS-ESR), simplified disease activity index (SDAI), clinical disease activity index (CDAI), and Health Assessment Questionnaire (HAQ) scores between baseline and 24weeks were analysed using a Wilcoxon Signed-Rank test. The improvement ratios of DAS28-ESR, SDAI, and CDAI between glucocorticoid use group and glucocorticoid non-use group were analysed using a Mann–Whitney U-test. Logistic regression analysis was used to calculate odds ratios (ORs) and 95% confidence intervals (CIs) for the risk factors of inability to taper glucocorticoid after 24 weeks after bDMARDs/JAKi switching in patients with D2TRA. In the logistic regres-sion analysis, ORs were adjusted for age, sex, body mass index, disease durations, titer of rheumatoid factor, titer of antibodies against cyclic citrullinated peptide, DAS-ESR, HAQ, methotrexate dose, glucocorticoid dose, type of bDMARDs/JAKi, number of past bDMARDs/JAKi, history of lymphoproliferative disorders, history of pancytopenia and history of interstitial pneumonia based on the results of the univariate analysis and general risk factors of inability to taper glucocorticoid.
Results: In 137 patients, 71 patients were treated with glucocorticoid and 66 patients were treated without glucocorticoid. The methotrexate usage rate was lower in glucocorticoid use group than in glucocorticoid non-use group (34.7 vs 56.1%, p=0.02), but there was no difference in the methotrexate dose between groups (8.6 ± 3.1 vs 9.1 ± 4.2 mg/week, p=0.76). Drug retention rate of bDMARDs/JAKi at 24 weeks was 70.8% in glucocorticoid use group, 62.1% in glucocorticoid non-use group. Drug retention rate was not different between groups (p=0.34). DAS-ESR, SDAI, CDAI, and HAQ at 4, 12, 24 weeks decrease in both groups (Figure 1). There were no diferences between the groups in improvement ratio at 24 weeks of DAS-ESR (p=0.97), SDAI (p=0.74), CDAI (p=0.84) and HAQ (p=0.13). In glucocorticoid use group (n=71), 29 patients (40.9%) could reduce glucocorticoid dosage after 24 weeks after bDMARDs/JAKi switching. Average dosage of glucocorticoid was 5.6 ± 3.1 mg/day from baseline to 2.5 ± 1.7 mg/day after 24 weeks. Seven patients (9.9%) could stop glucocorticoid within 24 weeks after bDMARDs/JAKi switching. DAS-ESR, SDAI, CDAI, and HAQ at 4, 12, 24 weeks in both 29 patients in which glucocorticoid reduction was possible (glucocorticoid tapering group) and 42 patients in which glucocorticoid reduction was not possible (glucocorticoid non-tapering group) were shown in Figure 2. Improvement ratio at 24 weeks of DAS-ESR, SDAI, and CDAI were higher in glucocorticoid tapering group than glucocorticoid non-tapering group (p<0.01, respectively). Multivariate logistic regression analysis revealed that the risk factors of inability to taper glucocorticoid in patients with D2TRA were high number of past bDMARDs/JAKi use (ORs: 0.15, 95% CI: 0.03-0.78, p=0.02) and history of lymphoproliferative disorders (ORs: 0.001, 95% CI: 0.00-0.76, p=0.04). Type of bDMARDs/JAKi (ORs: 0.32, 95% CI: 0.05-2.01, p=0.22), DAS28-ESR at baseline (ORs: 2.96, 95% CI: 0.74-11.9, p=0.13), and glucocorticoid dose at baseline (ORs: 1.29, 95% CI: 0.74-2.24, p=0.37) did not affect the reduction in glucocorticoid dosage after 24 weeks after bDMARDs/JAKi switching.
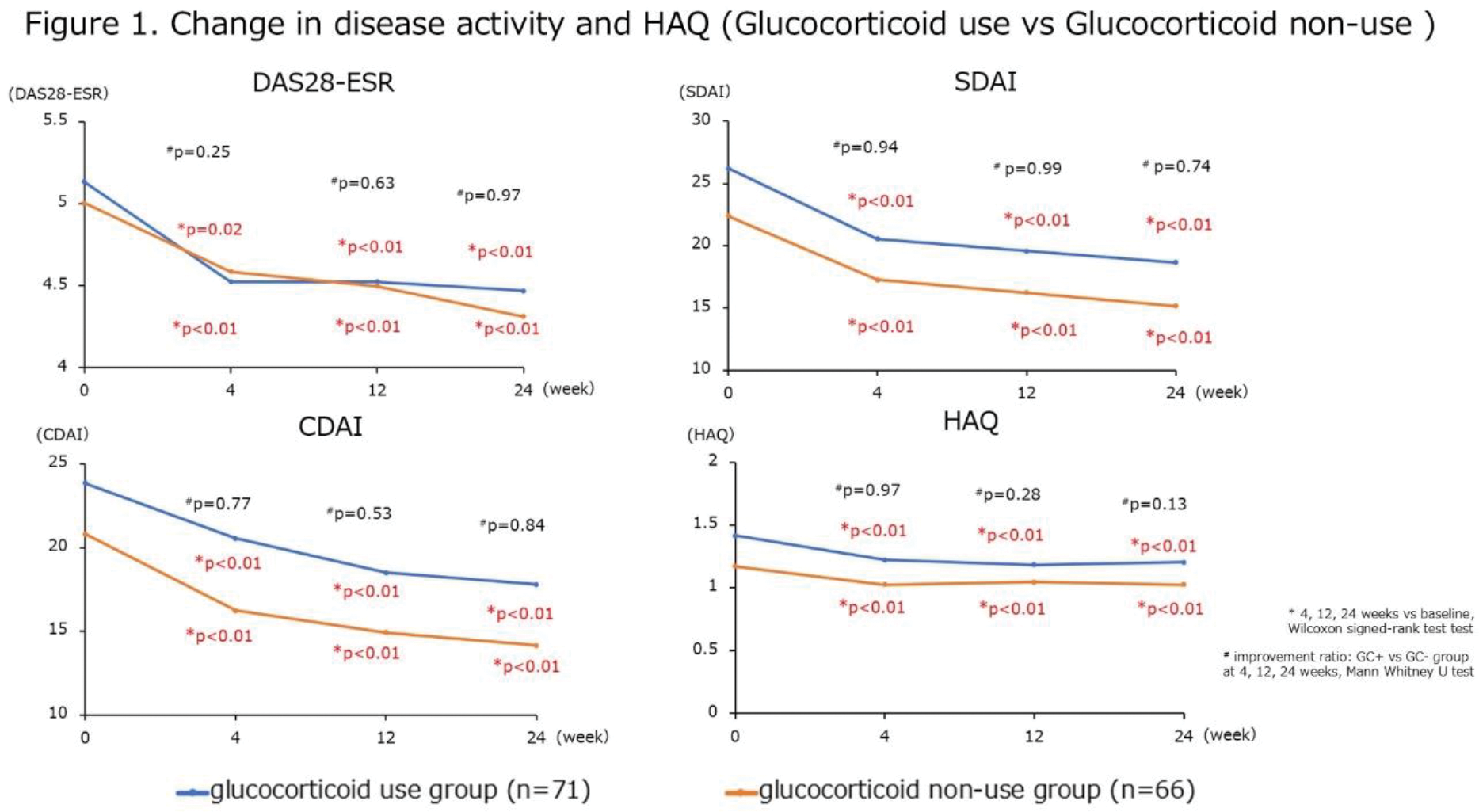
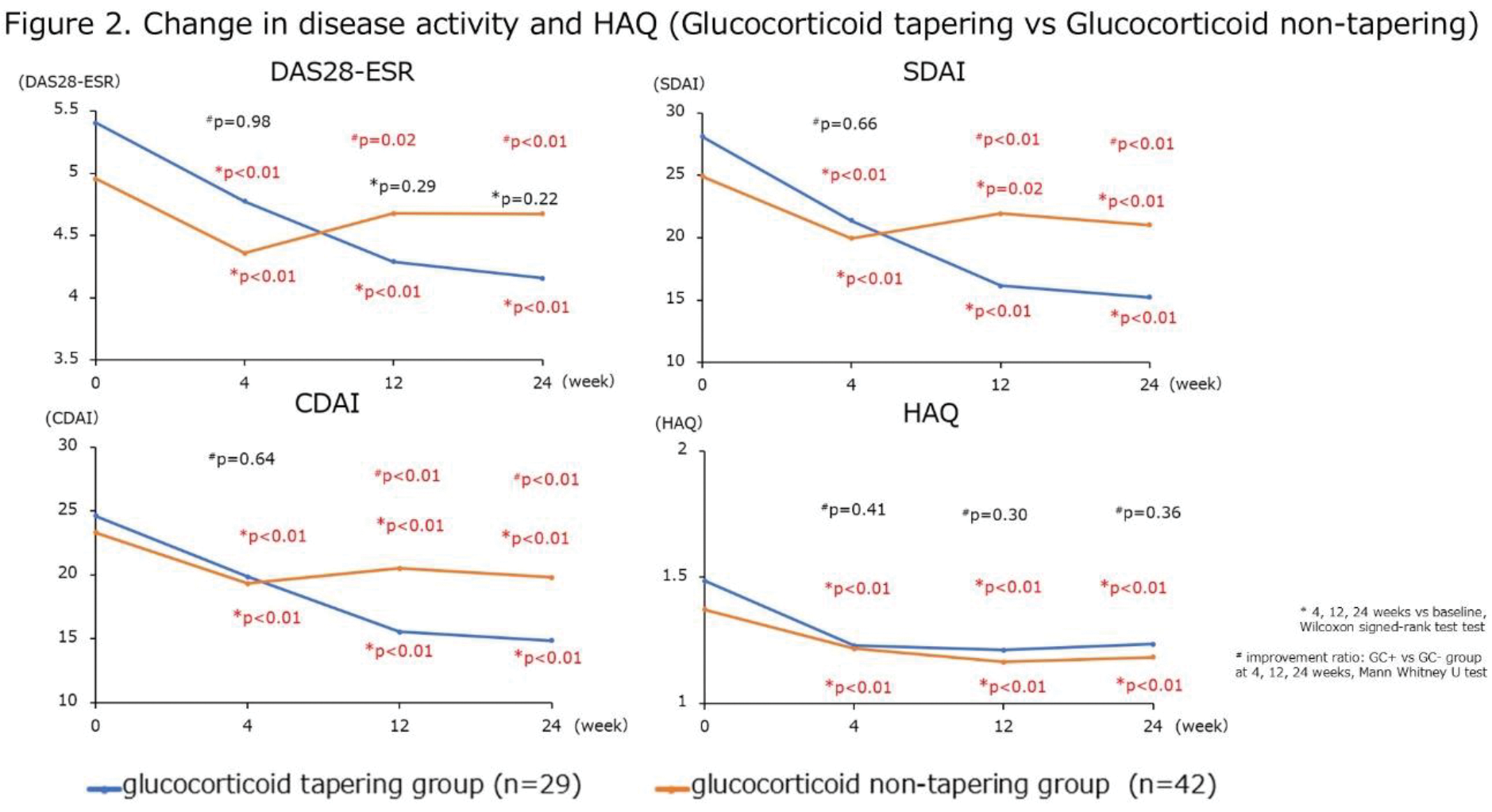
Conclusion: Drug retention rate and clinical efficacy of D2TRA patients were not different between glucocorticoid use group and glucocorticoid non-use group. In glucocorticoid use group, 40.9% could reduce glucocorticoid dosage after 24 weeks after bDMARDs/JAKi switching. Risk factors of inability to taper glucocorticoid in patients with D2TRA were high number of past bDMARDs/JAKi use and history of lymphoproliferative disorders.
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (