

Background: Ixekizumab (IXE), a high-affinity monoclonal antibody that selectively targets interleukin (IL)-17A, has demonstrated efficacy in Phase 3/4 clinical trials for patients with psoriatic arthritis (PsA) who were biologic disease-modifying anti-rheumatic drug (DMARD)-naïve [1, 2] and tumor necrosis factor inhibitor (TNFi)-experienced [3]. Other advanced therapies have been approved for PsA treatment. However, there is limited real-world (RW) effectiveness on the use of IXE and IL-23p19 inhibitors (IL-23i) for the treatment of PsA.
Objectives: Here, we present the final results of the prospective site-based Psoriatic Arthritis Real-World Study in the US (PARTUS), describing demographic and patient characteristics, as well as clinical and patient-reported outcomes at baseline, Week (W) 4, and W12, in patients with active PsA treated with IXE or an IL-23i.
Methods: PARTUS, a prospective observational study, enrolled adult patients with active PsA (clinical Disease Activity Index for PsA [cDAPSA] >4), meeting the Classification Criteria for Psoriatic Arthritis (CASPAR). IXE or an IL-23i (guselkumab [GUS] or risankizumab [RZB]) was initiated in accordance with the Food and Drug Administration (FDA) labelling in a US clinical setting. The primary objective was to assess the RW effectiveness in patients with PsA treated with IXE as measured by the change from baseline in cDAPSA at W12. The study was not powered for comparative analysis, therefore only descriptive results are presented for IXE and IL-23i. At baseline, demographics, clinical or disease severity measures, medical history, and medication use history were collected. Clinical and patient-reported outcomes were measured at baseline, W4, and W12. Among the clinical outcomes, disease activity (as measured by the cDAPSA), remission of disease activity (measured by the proportion of patients achieving cDAPSA ≤4), low disease activity (measured by the proportion of patients achieving cDAPSA ≤13, in patients with cDAPSA score >13 at baseline), and the body surface area (BSA) affected by psoriasis (measured by the proportion of patients who achieved a BSA score <3%, in patients with BSA ≥3% at baseline) were evaluated at W4 and W12. Among the patient-reported outcomes, health-related quality of life (measured by the Psoriatic Arthritis Impact of Disease [PsAID-12] score), pain (measured by the change from baseline in Patient Pain visual analogue scale [VAS] score in patients with baseline pain >0), fatigue (measured by a ≥4 score increase from baseline in the Functional Assessment of Chronic Illness Therapy-Fatigue [FACIT-Fatigue] score), and the global treatment satisfaction (measured by the absolute score of the abbreviated Treatment Satisfaction Questionnaire for Medication-9 [TSQM-9]) were evaluated at W4 and W12. Mean and standard deviations (SD) are reported for continuous variables, while counts and percentages are reported for categorical variables.
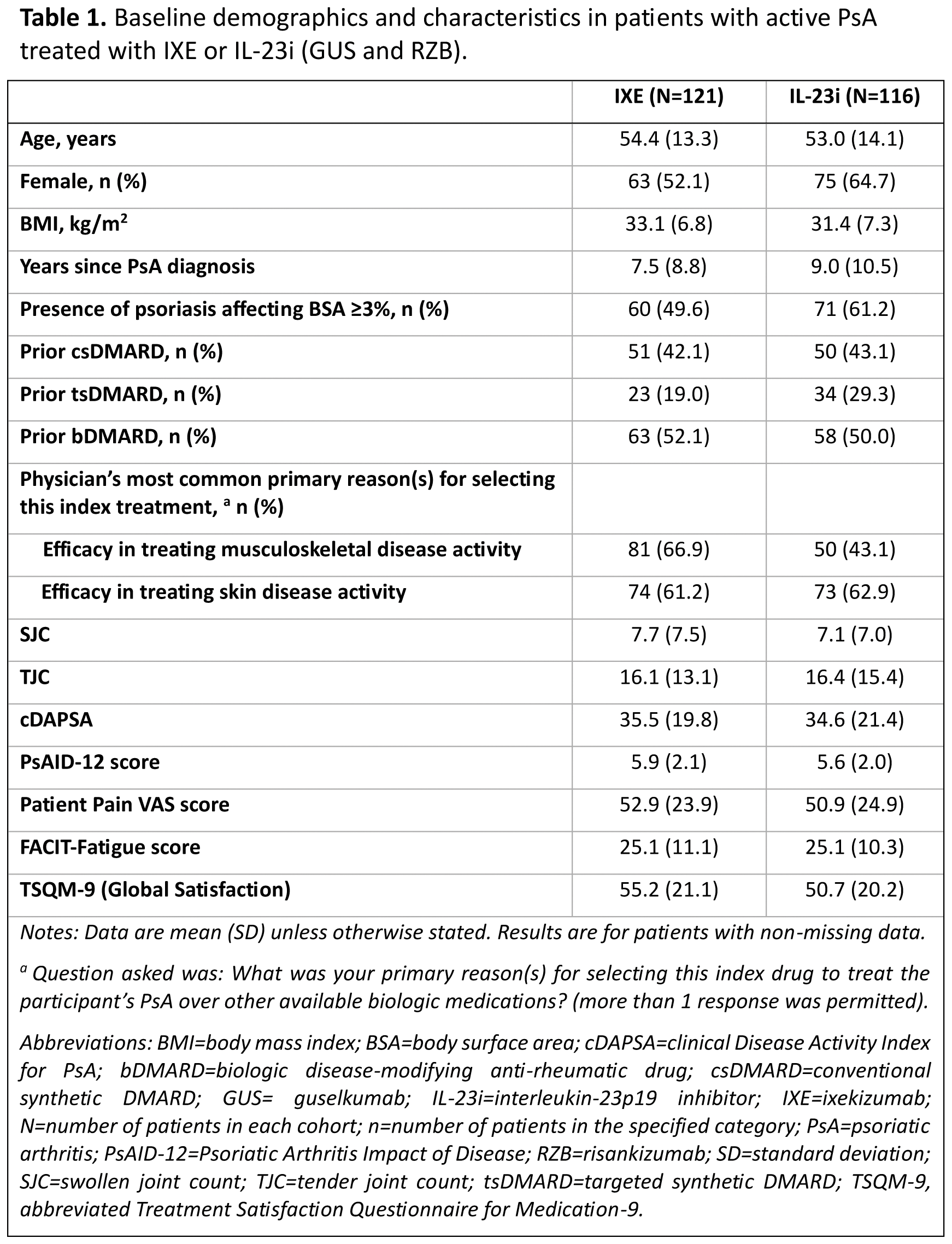
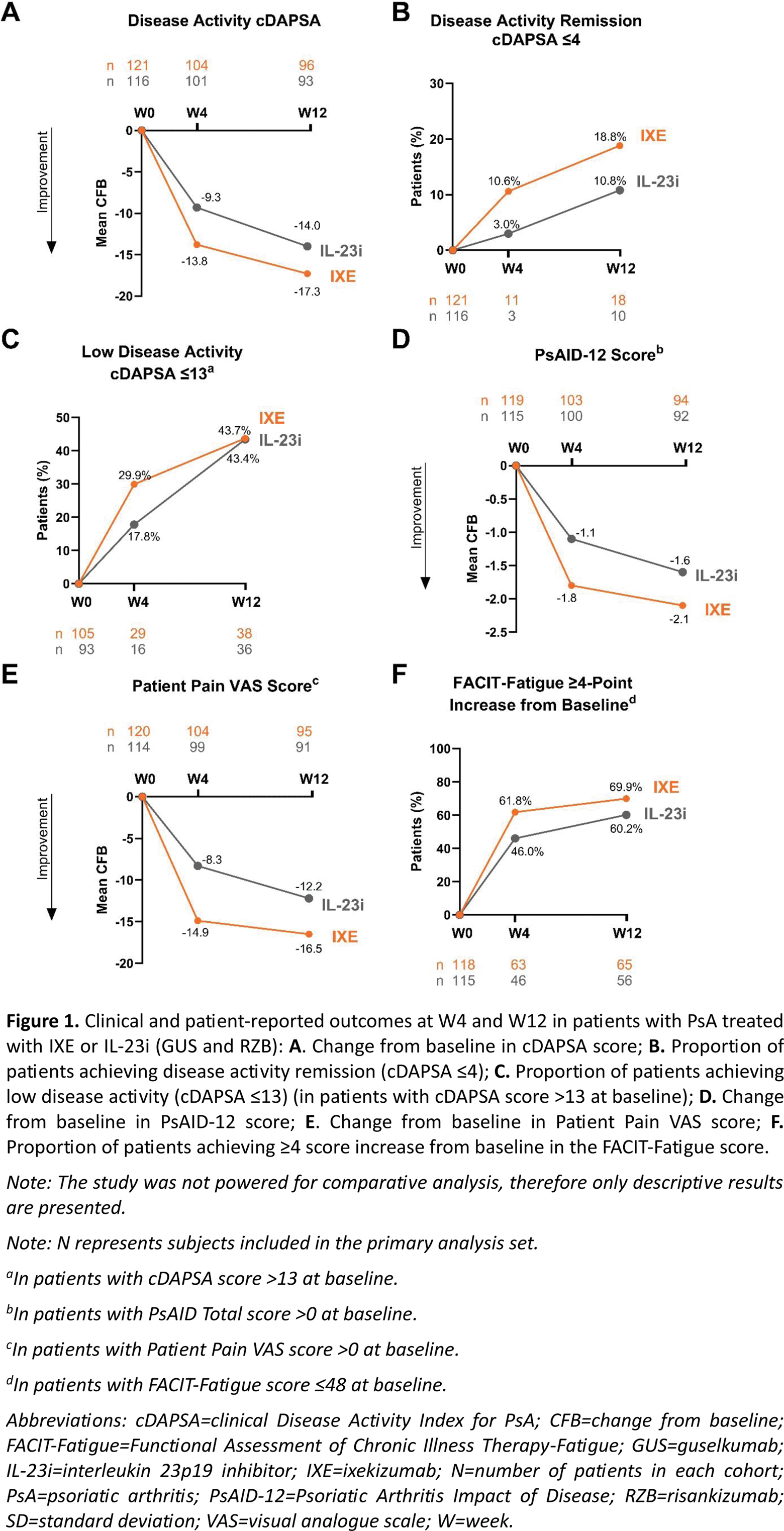
Results: Overall, 121 patients in the IXE cohort and 116 patients in the IL-23i cohort were included in this analysis. Baseline characteristics were similar between the two cohorts of patients (Table 1). Efficacy in treating musculoskeletal disease activity was among the physician’s most common primary reasons for choosing IXE (66.9%), while efficacy in treating skin disease activity was the most common reason for choosing IL-23i (62.9%). At W4, the mean (SD) change from baseline in the cDAPSA score was -13.8 (15.9) in the IXE cohort and -9.3 (15.4) in the IL-23i cohort. At W12, the mean (SD) change from baseline in the cDAPSA score was -17.3 (19.6) in the IXE cohort and -14.0 (19.3) in the IL-23i cohort (Figure 1A). At W4, the proportion of patients achieving remission of disease activity (cDAPSA ≤4) and low disease activity (cDAPSA ≤13) were 10.6% (n/Nx=11/104) and 29.9% (n/Nx=29/97) in the IXE cohort and 3.0% (n/Nx=3/101) and 17.8% (n/Nx=16/90) in the IL-23i cohort, respectively. These proportions increased through W12 in both cohorts of patients (Figure 1B, 1C). At W4, in patients with BSA ≥3% at baseline, the proportion of patients who achieved a BSA score <3% was 47.1% (n/Nx=24/51) in the IXE cohort and 27.4% (n/Nx=17/62) in the IL-23i cohort. These proportions increased by W12 in both cohorts of patients (67.4% and 57.4%, respectively). At W4, the mean (SD) change from baseline in the PsAID-12 score was -1.8 (1.9) in the IXE cohort and -1.1 (1.9) in the IL-23i cohort, with further improvements reported by W12 in both cohorts of patients (Figure 1D). Similar trends of improvements were reported for Patient Pain VAS (Figure 1E). At W4, the proportions of patients achieving clinically significant improvements in fatigue (FACIT-Fatigue ≥4-point increase from baseline) were 61.8% in the IXE cohort and 46.0% in the IL-23i cohort, with further improvements reported by W12 in both cohorts of patients (Figure 1F). In both the IXE and IL-23i cohorts, the TSQM-9 score regarding global satisfaction increased from baseline (55.2 and 50.7, respectively) to W4 (65.0 and 61.7, respectively) and remained stable at W12 (63.9 and 60.1, respectively).
Conclusion: The results from the prospective observational RW study PARTUS showed that patients with active PsA treated with IXE achieved meaningful early improvements by W4 in both disease activity and patient-reported outcomes. Disease outcomes continued to improve up to W12. Patients treated with IXE reached disease activity remission and low disease activity rapidly and through W12. Trends in improvement were also observed in the patients treated with IL-23i.
REFERENCES: [1] Mease PJ, et al. Ann Rheum Dis. 2017;76:79-87. 10.1136/annrheumdis-2016-209709.
[2] Smolen JS, et al. Rheumatol Ther. 2020;7:1021-1035. 10.1007/s40744-020-00250-3.
[3] Nash P, et al. Lancet. 2017;389:2317-2327. 10.1016/S0140-6736(17)31429-0.
Acknowledgements: NIL.
Disclosure of Interests: Kurt Oelke is on the speaker’s bureau for: AbbVie, Amgen, AstraZeneca, Janssen, and UCB Pharma, Emily Edson-Heredia is an employee and shareholder of: Eli Lilly and Company, Sarah Ross is an employee and shareholder of: Eli Lilly and Company, Jeffrey Lisse is a former employee and shareholder of: Eli Lilly and Company, Jennifer Pustizzi is an employee and shareholder of: Eli Lilly and Company, Ali Sheikhi Mehrabadi is an employee and shareholder of: Eli Lilly and Company, Natalia Bello Vega is an employee and shareholder of: Eli Lilly and Company, Frederick Murphy is on the speaker’s bureau for: AbbVie, Shikha Singla is on the speaker’s bureau for: Janssen, has received consulting fees from: AbbVie, Janssen and UCB Pharma, has received research grants from: Eli Lilly and Company and Prometheus Biosciences, Siba P Raychaudhuri is on the advisory board consultancy for: UCB Pharma, has received research grants from: AbbVie, Eli Lilly and Company, and Novartis.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (