

Background: Current standard therapies for systemic lupus erythematosus (SLE), including glucocorticoids and conventional immunosuppressants, often deliver inadequate responses. In addition, toxicity concerns have created the need for treatments with better benefit-risk profiles to improve long-term prognoses of patients with SLE. B-cell–depleting therapies have gained attention due to the important role of B cells in the pathogenesis of SLE. Obinutuzumab is a recombinant, monoclonal, humanised and glycoengineered type II CD20 antibody with greater B-cell–depleting capacity compared with type I anti-CD20 monoclonal antibodies. In the Phase III REGENCY study (NCT04221477), the number of patients achieving complete renal response at Week 76 was significantly higher in those treated with obinutuzumab plus standard therapy (mycophenolate mofetil plus glucocorticoids) (46.4%) compared with standard therapy alone (33.1%) (adjusted difference: 13.4%; 95% CI, 2.0% to 24.8%; P =0.0232), with an acceptable safety profile.
Objectives: The ALLEGORY (NCT04963296) study aims to evaluate the efficacy, safety, pharmacokinetics and pharmacodynamics of obinutuzumab compared with placebo in participants with SLE when added to standard therapy.
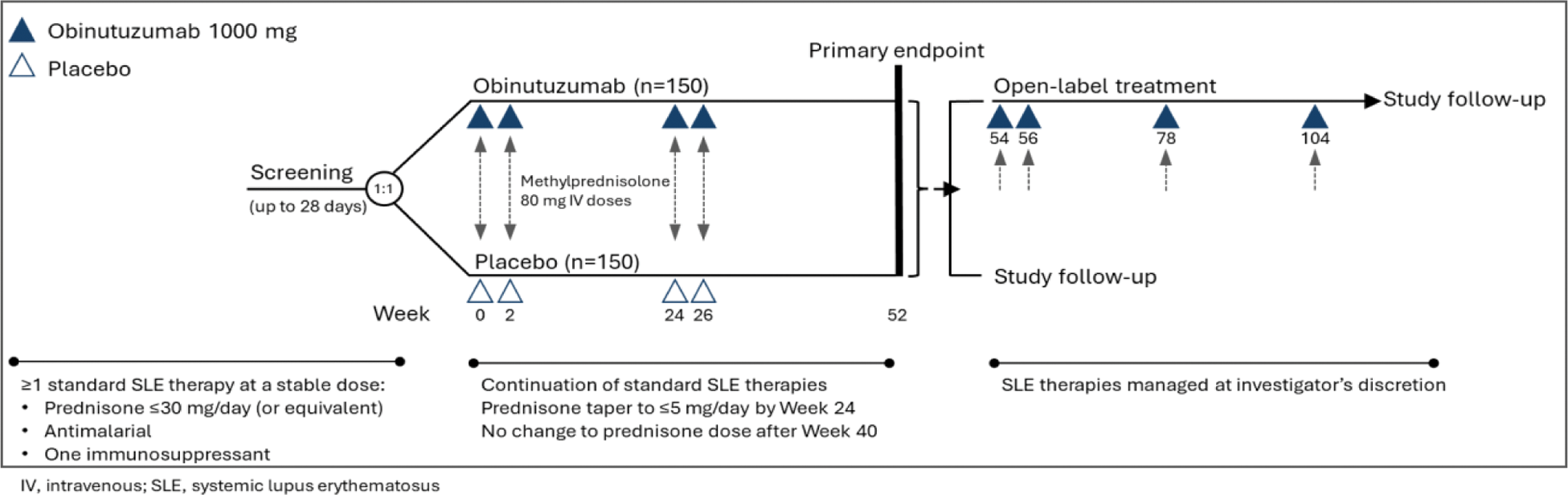
Methods: Participants with SLE (per 2019 EULAR/ACR Classification Criteria) with high disease activity (Systemic Lupus Erythematosus Disease Activity Index 2000 [SLEDAI-2K] ≥8 and British Isles Lupus Assessment Group 2004 [BILAG-2004] Category A severity in ≥1 domain and/or Category B severity in ≥2 domains), and receiving at least one standard therapy for SLE (at least one or a combination of immunosuppressants, antimalarials and glucocorticoids), were randomised 1:1 to receive blinded infusions of obinutuzumab (1000 mg) or placebo on Day 1 (Week 0) and at Weeks 2, 24 and 26 (Figure 1). After completing blinded treatment at Week 52, participants from both groups may be eligible to receive an additional 18 months of open-label treatment (OLT), in which obinutuzumab will be administered at Weeks 54, 56, 78 and 104. After completion of study infusions during the blinded stage or the OLT, participants will be followed for at least 12 months from their last infusion to monitor safety and disease activity, with no further obinutuzumab infusions. The primary endpoint is the difference between the proportions of obinutuzumab-treated versus placebo-treated patients who achieve the SLE Responder Index-4 (SRI-4) at Week 52, defined as a reduction of ≥4 points in the SLEDAI-2K, <1 new BILAG A or <2 new BILAG B items and no worsening of ≥0.3 points on a 3-point Physician’s Global Assessment-Visual Analogue Scale. Secondary endpoints include the proportions of participants who achieve BILAG-based Composite Lupus Assessment or SRI-6 at Week 52, sustained glucocorticoid control from Weeks 40 to 52 (for those having entered the study on prednisone [or equivalent] ≥10 mg/day), sustained SRI-4 response from Weeks 40 to 52 and the time to first BILAG flare over 52 weeks.
Results: ALLEGORY is fully enrolled with 303 participants from 64 sites across 14 countries worldwide. Participants are distributed across Latin America (54.1%), eastern Europe (14.9%), North America (12.2%), western Europe (9.2%), Africa (8.9%) and Asia–Pacific (0.7%) regions. Enrolled patients have a median (range) age of 41.0 (18–70) years and 90.4% are female (Table 1).
Conclusion: Full results of the ALLEGORY study will be available upon study completion and will assess the efficacy and safety of obinutuzumab in SLE.
ALLEGORY Study Design
Demographic Characteristics of Participants Enrolled in the ALLEGORY Study
| Characteristic | All Participants
|
|---|---|
| Age, years, mean (SD ) | 41.2 (12.4) |
| Male/ Female, n (% ) | 29 (9.6) / 274 (90.4) |
| Race, n (% ) | |
| American Indian or Alaska Native | 86 (28.4) |
| Asian | 6 (2.0) |
| Black or African American | 47 (15.5) |
| Native Hawaiian or other Pacific Islander | 1 (0.3) |
| White | 125 (41.3) |
| Multiple | 18 (5.9) |
| Unknown | 20 (6.6) |
| Region, n (% ) | |
| Africa | 27 (8.9) |
| Asia–Pacific | 2 (0.7) |
| Eastern Europe | 45 (14.9) |
| Latin America | 164 (54.1) |
| North America | 37 (12.2) |
| Western Europe | 28 (9.2) |
*Data as of 2 November 2024.
REFERENCES: NIL.
Acknowledgements: Funded by F. Hoffmann-La Roche Ltd. Editorial assistance was provided by Nucleus Global, an Inizio company, and funded by F. Hoffmann-La Roche Ltd.
Disclosure of Interests: Edward M. Vital consulting fees from AbbVie, AstraZeneca, Eli Lilly and Company, F. Hoffmann-La Roche Ltd/Genentech, Inc., Merck, Novartis, Otsuka, Pfizer and UCB, Zahir Amoura consulting fees from Amgen, AstraZeneca, Genentech, Inc., GlaxoSmithKline, Kezar and Novartis, research support from Amgen, AstraZeneca, Genentech, Inc., GlaxoSmithKline, Kezar and Novartis, Stella Botha: None declared, Maria Antonietta D’ Agostino speaker and consulting fees from AbbVie, Alfasigma Global, Amgen, AstraZeneca, BMS, Eli Lilly, Galapagos, GlaxoSmithKline, J&J, MSD, Novartis, Sanofi and UCB, Sunil Kumar: None declared, Fedra Irazoque-Palazuelos consulting fees from AbbVie, Amgen Inc. Eli Lilly, F. Hoffmann-La Roche Ltd, Novartis and UCB, research support from AbbVie, Amgen Inc. Eli Lilly, F. Hoffmann-La Roche Ltd, Novartis and UCB, Grzegorz Rozumek: None declared, Elizabeth Zuta Santillan: None declared, Viviane Souza: None declared, Biruh Workeneh: None declared, Justine Maller shareholder of F. Hoffmann-La Roche Ltd, employee of Genentech, Inc., Oliver Meier shareholder of F. Hoffmann-La Roche Ltd, employee of F. Hoffmann-La Roche Ltd, Jay P. Garg shareholder of Hoffmann-La Roche Ltd, employee of Genentech, Inc., Richard A. Furie consulting fees from Genentech, Inc., and GlaxoSmithKline, research support from Genentech, Inc., and GlaxoSmithKline.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (