

Background: Rheumatoid arthritis requires long-term treatment, and tapering of medication should be considered after achieving the treatment target. However, standardised strategies for the medication tapering in patients treated with a tumor necrosis factor (TNF) inhibitor and methotrexate (MTX) have not yet been established.
Objectives: This study aimed to assess the efficacy and safety of spacing of ozoralizumab (OZR), a next generation of TNF inhibitor that binds TNFα and albumin, or dose reduction of MTX compared to continued treatment in patients with rheumatoid arthritis who maintained remission or low disease activity with the combination of OZR and MTX.
Methods: This multicenter, open-label, randomised, parallel-group non-inferiority study enrolled 149 patients who maintained remission or low disease activity in the preceded trial [1]. These patients had received combination therapy with OZR and MTX for approximately 3-4 years and achieved remission or low disease activity based on clinical disease activity index (CDAI, ≤10). The participants were randomised into three groups: continued treatment, OZR spacing (from every 4 to 6 to 8 weeks), or MTX dose reduction (by 2 mg/week every 8 weeks). The primary endpoint was the proportion of patients who maintained CDAI ≤10 at week 48. Additionally, the proportions of patients who achieved MTX-free status in the MTX dose reduction group and an 8-week OZR dosing interval in the OZR spacing group were evaluated. Other efficacy indices and safety were also assessed.
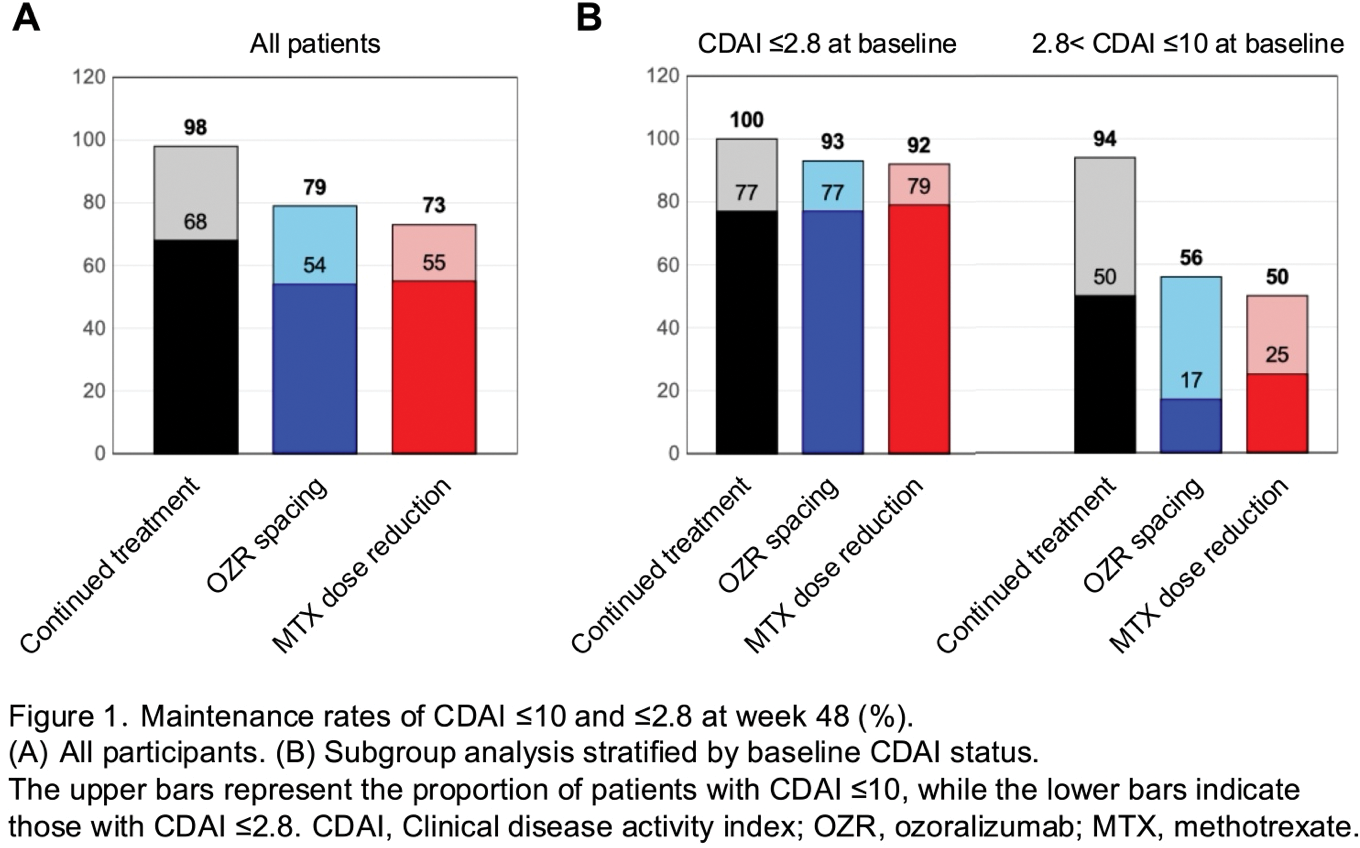
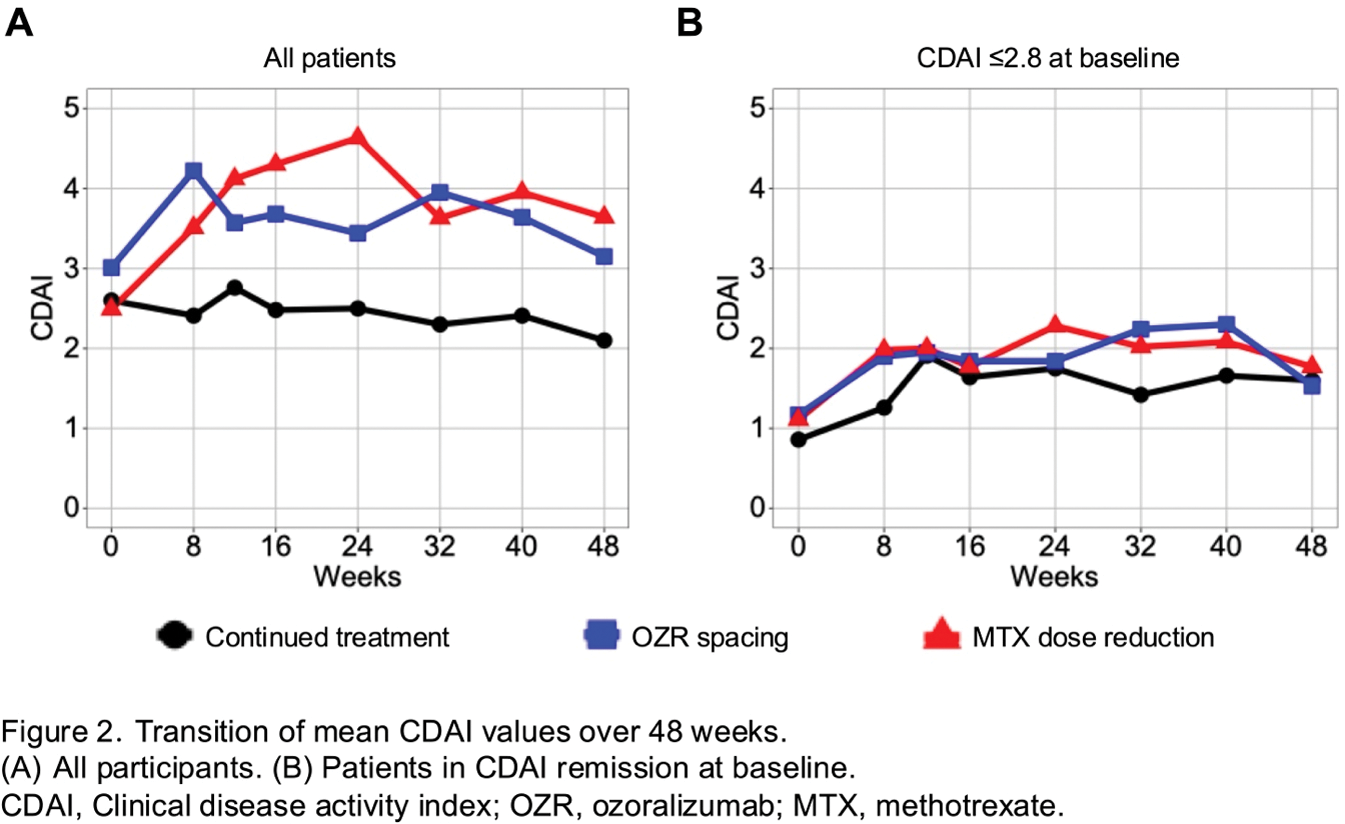
Results: A total of 144 patients were included in the per-protocol set analysis. The mean age was 58.2 years, and 75 % were female. Eighty nine patients (62%) were in CDAI remission (≤2.8). At week 48, the proportions of patients with CDAI ≤10 were 98% in the continued treatment group, 79% in the OZR spacing group (difference: -22%, 95% CI: -40 to -6%), and 73% in the MTX dose reduction group (difference: -30%, 95% CI: -54% to -11%) (Figure 1A). These results did not meet the pre-specified non-inferiority margin (lower limit of 95% CI: -18%). However, in the subgroup who were in CDAI remission at baseline, CDAI ≤10 was maintained in 100% of the continued treatment group, 93% of the OZR spacing group, and 92% of the MTX dose reduction group, and remission was maintained comparably among the three groups (77–79%, Figure 1B). The mean CDAI trajectory is illustrated in Figure 2. Maintenance of CDAI ≤10 at week 48 was associated with baseline CDAI remission (vs. low disease activity; OR 11.345, p <0.001) and serum albumin >3.8 g/dL (vs. ≤3.8 g/dL; OR 4.061, p = 0.012) in logistic regression analysis adjusted for treatment group. MTX-free status was achieved in 49% of patients in the MTX dose reduction group, although no significant predictive factors were identified. In contrast, 80% of patients in the OZR spacing group reached an 8-week OZR dosing interval, which was associated with baseline CDAI remission (OR 10.182, p = 0.007) and serum albumin >3.8 g/dL (OR 5.500, p = 0.027). Among patients experiencing disease flares, 89% in the OZR spacing group and 83% in the MTX dose reduction group achieved CDAI ≤10 again after rescue therapy (an increase to the original dosage of OZR or MTX). The mean change in modified Total Sharp Score over 48 weeks indicated structural remission (<0.5) in all groups (range: 0.14–0.36). Adverse events were observed in 68% in the continued treatment group, 59% in the OZR spacing group, and 58% in the MTX dose reduction group. Two serious adverse events occurred in each group.
Conclusion: In patients who have achieved remission with a combination of TNF inhibitor and MTX therapy, extending OZR interval or reducing MTX dose can be feasible and favorable under careful clinical monitoring. This study also re-emphasises the importance of achieving remission for the subsequent successful treatment reduction.
REFERENCES: [1] Effect of the extended dosing interval of anti-TNF-α NANOBODY® compound ozoralizumab in patients with low disease activity rheumatoid arthritis. Mod Rheumatol. 2024;34:678–685.
Acknowledgements: The authors would like to express their sincere gratitude to all participants in this study. The authors also thank Linical Co., Ltd. and Medical Edge Co., Ltd. for their support in study administration, monitoring, data management, and statistical analysis.
Disclosure of Interests: Yuki Imai: None declared, Tatsuya Atsumi Bristol-Myers Squibb Co., Eli Lilly Japan K.K., Nippon Boehringer Ingelheim Co., Ltd., Eisai Co. Ltd., AbbVie Inc., Pfizer Inc., Gilead Sciences, Inc., Daiichi Sankyo Co., Ltd., AstraZeneca plc., Asahi Kasei Pharma Co., Janssen Pharmaceutical K.K., AstraZeneca plc, Idorsia Pharmaceuticals Ltd, Otsuka Pharmaceutical Co., Gilead Sciences, Inc., GlaxoSmithKline plc., Sanofi K.K, Eli Lilly Japan K.K., Nippon Boehringer Ingelheim Co., Ltd., Janssen Pharmaceutical K.K., UCB Japan Co. Ltd., KISSEI PHARMACEUTICAL CO., LTD., Novartis Pharma K.K., GlaxoSmithKline plc., Bristol-Myers Squibb Co., Zenyaku Kogyo Company, Ltd., Amgen K.K., KISSEI PHARMACEUTICAL CO., LTD., AbbVie Inc., Mitsubishi Tanabe Pharma, Eisai Co. Ltd., Asahi Kasei Pharma Co., Nippon Boehringer Ingelheim Co., Otsuka Pharmaceutical Co., Ltd., Chugai Pharmaceutical Co., Ltd., Tomonori Ishii Asahi Kasei, Chugai, Eli Lilly, GSK, Pfizer, Boehringer-ingelheim, and AstraZeneca., Asahi Kasei., Toshihisa Kojima: None declared, Shintaro Hirata: None declared, Yoshiya Tanaka YT has received speaking fees and/or honoraria from Chugai, UCB, Abbvie, AstraZeneca, Eli-Lilly, Behringer-Ingelheim, GlaxoSmithKline, Eisai, IQVIA, Daiichi-Sankyo, Otsuka, Taisho, Gilead, Bristol-Mayers., Eiichi Tanaka ET has received lecture fees from AbbVie Japan GK, Asahi Kasei Corp., Astellas Pharma Inc., Ayumi Pharmaceutical Co., Boehringer Ingelheim Japan, Inc., Bristol Myers Squibb Co., Ltd., Chugai Pharmaceutical Co., Ltd., Daiichi-Sankyo, Inc., Eisai Co., Ltd., Eli Lilly Japan K.K., Gilead Sciences, Inc., Pfizer Japan Inc, Nichi-Iko Pharmaceutical Co., Ltd., Taisho Pharmaceutical Co., Ltd, Takeda Pharmaceutical Co., Ltd, Mitsubishi Tanabe Pharma Co., UCB Japan Co. Ltd. and Viatris Inc., Koichiro Ohmura: None declared, SHUNSUKE OKAMOTO Taisho Pharmaceutical Co., Ltd., Rumiko Matsumoto Taisho Pharmaceutical Co., Ltd., Yuri Sato Taisho Pharmaceutical Co., Ltd., Takashi Yamamura Taisho Pharmaceutical Co., Ltd., Tsutomu Takeuchi AbbVie GK, Chugai, Eli Lilly Japan, Eisai, Gilead Sciences Inc, Pfizer Japan Inc, Taisho Pharma., AbbVie GK, Eli Lilly Japan, Gilead Sciences, Inc. Mitsubishi-Tanabe, Taisho Pharma., Yuko Kaneko Taisho Pharmaceutical Co. Ltd, Taisho Pharmaceutical Co. Ltd.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (