

Background: Currently oral xanthine oxidase inhibitor and uricosuric drugs for gout are limited by requirements for multiple titrations and potential adverse events, which creates unmet therapeutic needs. SAP-001 is a new first-in-class compound, since it mainly targets a major renal urate reabsorption transporter other than URAT1. In completed Phase 1 and 2a studies in gout patients with hyperuricemia, the compound displayed strong urate-lowering effects beyond those obtained with currently approved.
Objectives: To evaluate the safety and efficacy of SAP-001 in refractory gout patients with or without palpable tophi.
Methods: This was a six-month, multi-center, randomized, double-blinded, placebo-controlled phase 2b clinical trial conducted in the US. N=87 gout patients with or without tophi, and refractory to conventional therapy. Subjects were enrolled using a 1:1:1:2 randomization ratio to placebo, or SAP-001 10, 30, and 60 mg treatment groups. The comparison of each dose group against placebo in the proportion of subjects achieving various sUA thresholds at different time points was performed using CMH test stratified by the baseline tophi status. Linear dose response in these proportions was assessed by testing the coefficient for dose in a logistic regression model.
Results: Mean baseline serum urate (sUA) level was 8.37 (±1.29) mg/dL and was similar among all treatment groups. Baseline characteristics, such as race, ethnicity, gender, Body Mass Index, comorbidities, patients with renal impairment, or with palpable tophi were comparable among the treatment groups. The majority of the subjects were male, obese, and on background SOC treatment (more than 90% with either 100 mg, or 300 mg allopurinol for ≥3 months, and with sUA above 7 mg/dL during screening in all patients). Average duration of diagnosed gout was ~10 years. About 20% had palpable tophi.
Efficacy
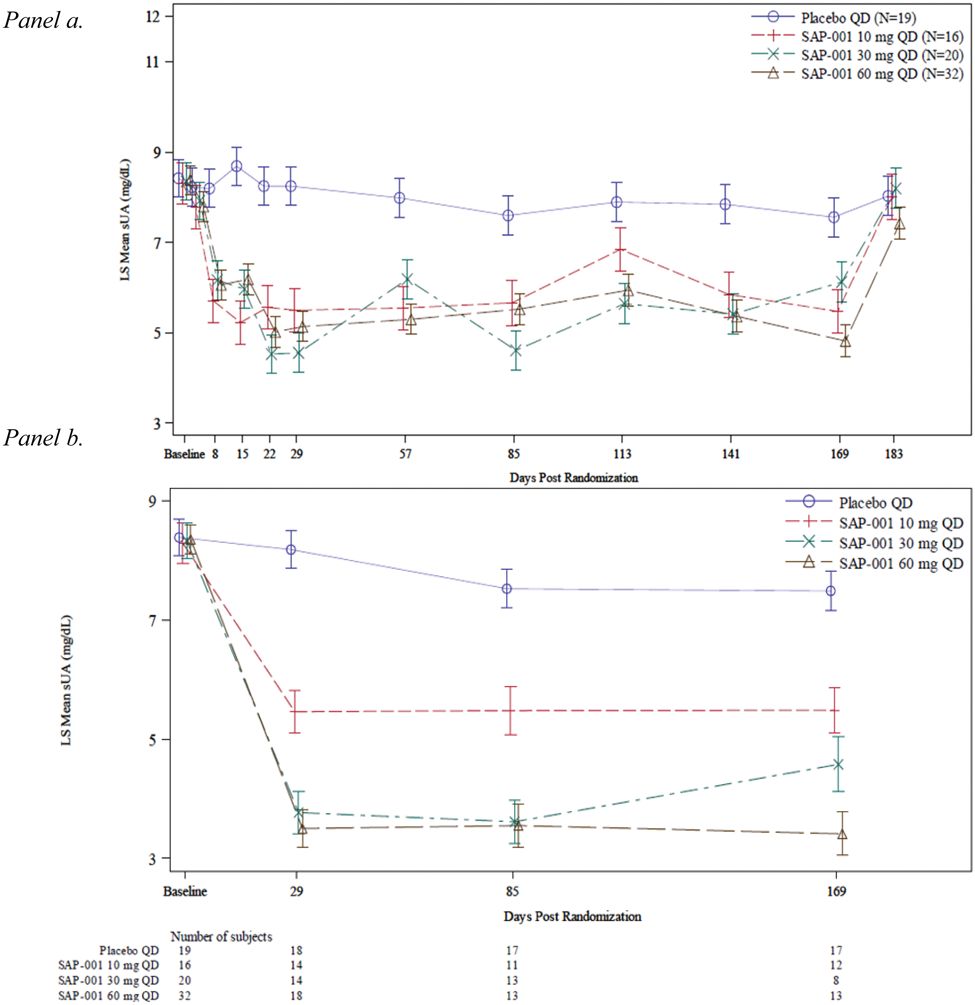
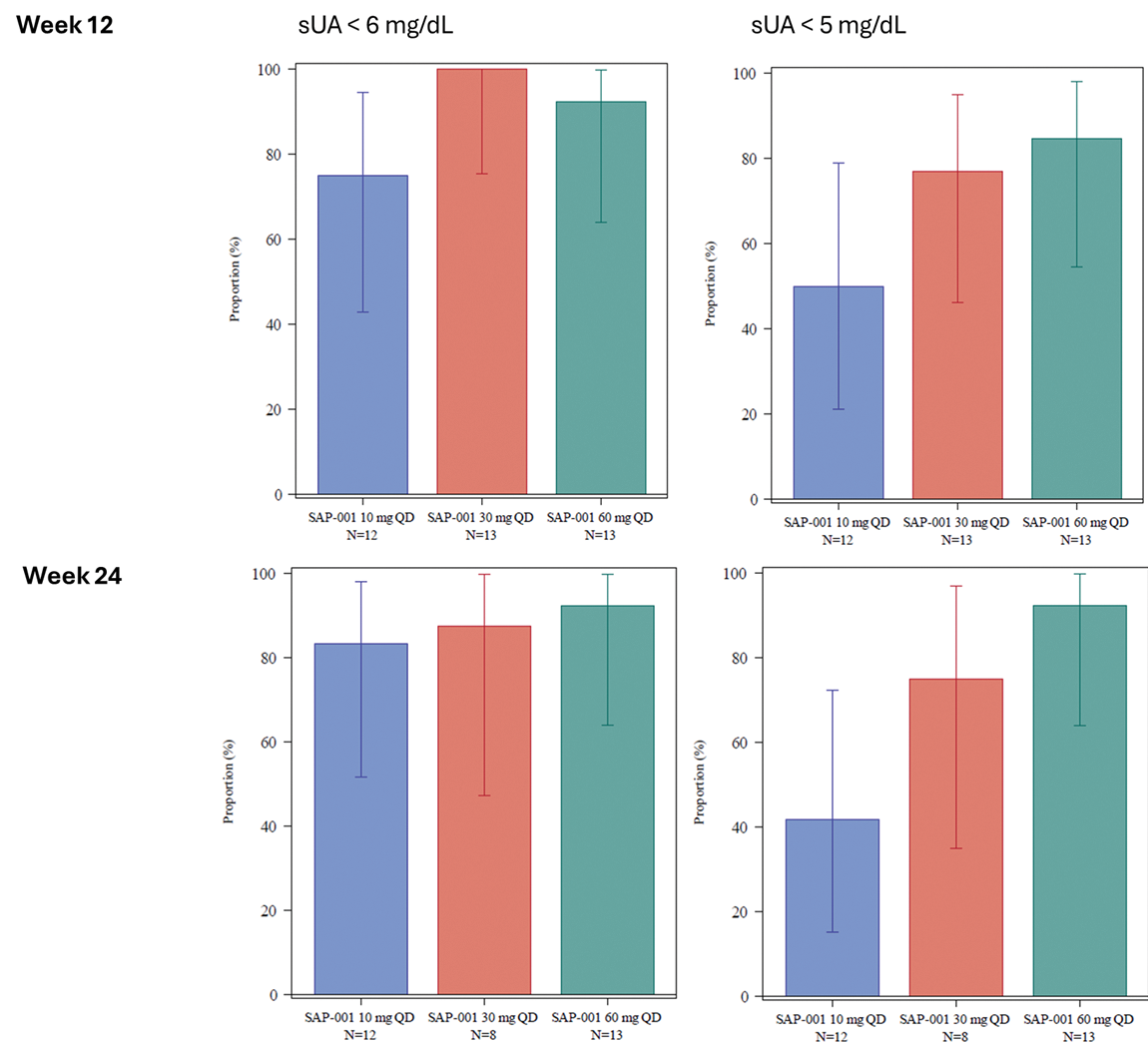
A significant sUA lowering effect that treated with SAP-001 three dose groups (10, 30, and 60 mg QD) superimposed on baseline ULT SOC with a xanthine oxidase inhibitor observed as early as one week post randomization, sUA further reduced by the end of dosing titration at Week 3. The urate lowering effects were maintained throughout the treatment period (Figure 1 panel a). A significant proportion of subjects achieved sUA below 6 mg/dL; 43.8% to 70% of patients in the three dose groups achieved sUA below 6 mg/dL compared to 10.5% in the continuing XOI SOC treatment group at 12 weeks. By end of treatment, 50 - 79% of patients achieved sUA below 6 mg/dL and 37 -56% achieved sUA below 5 mg/dL in the three dose groups. The study uncovered a fraction of patients not exposed to SAP-001, which was confirmed by pharmacokinetics (PK) analyses prior to dosing at protocol-defined timepoints. These findings were strongly associated with poor adherence by other sources (patient daily diary, pill counts, post-study patient interviews). We thus conducted additional analyses on patients with quantifiable plasma SAP-001 demonstrated consistent and dose-dependent efficacy (Figure 1. panel b, and Figure 2). At week 4, all groups treated with SAP-001 had an excellent response, by week 12, almost all patients treated with targeted dose of 30 and 60 mg achieved target sUA level < 6 mg/dL. Over 50% of patients achieved sUA < 3 mg/dL in those on SAP-001 60 mg/day, with effects maintained well until end of treatment at Month 6 with SAP-001.
Panel a. Efficacy – Average sUA Over Time (Full Analysis Set)panel b. Efficacy – Average sUA Over Time (Subjects in the Full Analysis Set who were exposed to SAP-001 confirmed by PK analyses at the pre-dose at defined time points).
sUA response rate based on additional analysis (patients with quantifiable plasma SAP-001 at analysis time points at 12, and 24 weeks
Safety
Overall, SAP-001 was well tolerated. There was no treatment-emergent SAE in SAP-001 treated patients. All AEs were mild-moderate in severity in active treatment groups. The most frequently reported adverse event was gout flare in ~30% of patients overall, with similar distribution in placebo and SAP-001 treated groups. Isolated mild or moderate TEAE, including diarrhea, headache, anemia, and sinus bradycardia, were observed. Due to SAP-001 being a uricosuric, we paid special attention to any potential renal safety signal. Transient creatine elevations/decreased eGFR were reported as Adverse Events of Special Interest based on protocol criteria (9 AESIs in 6 subjects). None of these events met Serious AE criteria and all resolved during the study or shortly thereafter (for one case of mild creatinine increase with a start date at End of Study). One event of moderate decrease in eGFR led to discontinuation of investigational product per protocol requirement.
Conclusion: Among patients exposed to study drug, SAP-001 demonstrated robust, sustained urate-lowering effect in refractory gout patients with or without palpable tophi. A first in class inhibitor of a distinct renal urate transporter, SAP-001 may offer a mechanistically novel, effective and safe oral ULT drug for difficult- to-treat gout populations.
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: Shanshan Cui Shanton Pharma, Carmen Arencibia Shanton Pharma, li li Shanton Pharma, Qian Zhang Shanton Pharma, Ouhong Wang Shanton Pharma, Jean-Louis Saillot Shanton Pharma, Rachel Peterson Shanton Pharma, Hyon Choi Shanton Pharma, Kenneth Saag Shanton Pharma, Nicola Dalbeth Shanton Pharma, Robert Terkeltaub Shanton Pharma, Wenfeng Miao Shanton Pharma.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (