

Background: Population-based data regarding the incidence and mortality of Interstitial lung disease (ILD) with and without progressive pulmonary fibrosis (PPF) in patients with Connective Tissue Diseases (CTDs) are limited.
Objectives: The aim of the study was to assess the incidence and mortality of patients with CTD-ILD compared to CTD without ILD in France, at the whole country-level, and to estimate the proportion of ILD with PPF.
Methods: We performed a nation-wide population-based study of CTD-ILD using the French national claims database (SNDS), which contains pseudonymized data for >67 million inhabitants. Patients with rheumatoid arthritis (RA), primary Sjögren syndrome (pSS), mixed connective tissue disease (MCTD), systemic lupus erythematosus (SLE), idiopathic inflammatory myopathies (IIM), systemic sclerosis (SSc) were identified in the SNDS based on Long-Term Disease and/or ICD10 codes. Incident patients were identified as having a first CTD diagnosis during the 2015 to 2022 study period, and by excluding those with a CTD diagnosis in 2010-2015. ILD was identified by the J841 ICD code. PPF was identified using a proprietary algorithm based on age, diagnosis codes and changes in healthcare consumption [1]. A multivariable Cox model was used to estimate the mortality risk of patients with CTD-ILD compared to CTD without ILD.
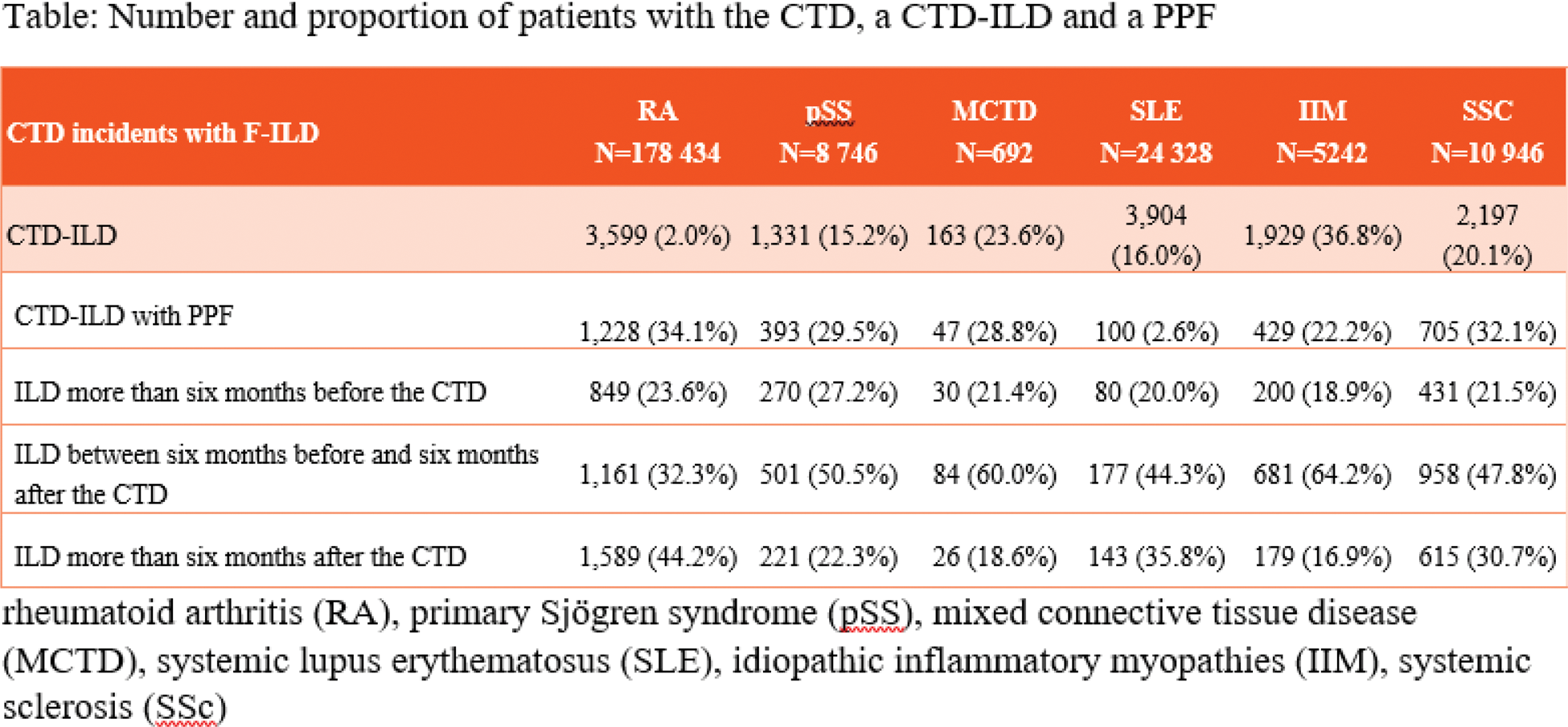
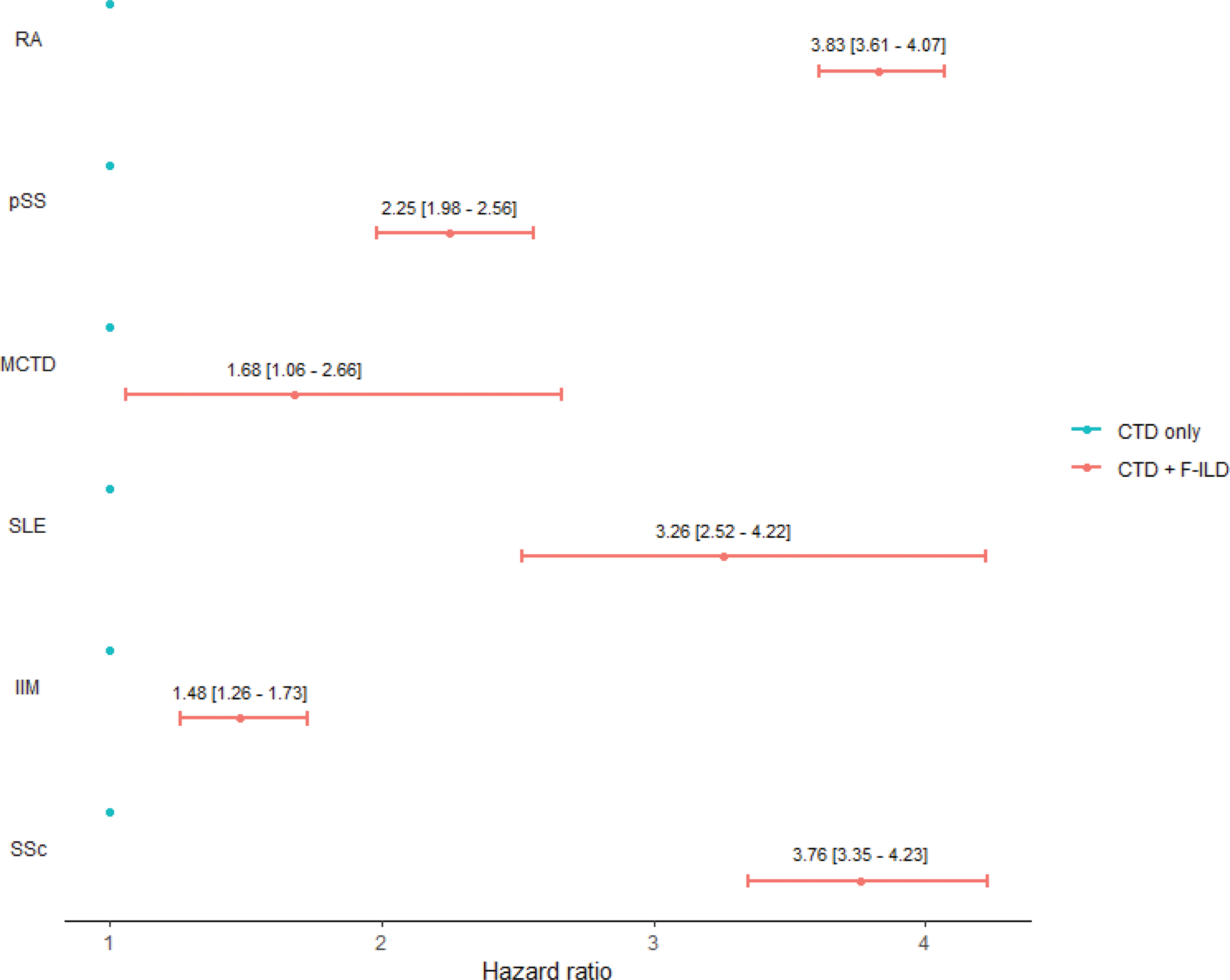
Results: A total of 228,388 incident CTD patients were identified in the 8-year study period (Figure 1), including 178,434 patients with RA, 8746 with pSS, 692 with MCTD, 24328 with SLE, 5242 with IIM, and 10946 with SSc. The mean ±SD age at identification of the CTD was: 63.0 ±16.1 years for RA, 63.4 ±16.6 for pSS, 49.7 ±19.6 for MCTD, 46.1 ±17.2 for SLE, 55.3 ±20.2 for IIM, and 58.2 ±16.6 for SSc. The proportion of incident CTD patients who had ILD varied between 2.0% in RA and 36.8% in IIM (Table 1). Among incident CTD-ILD patients, the proportion who had PPF varied between 2.6% for SLE and 34.1% for RA (Table 1). The proportion of patients for whom the ILD was identified more than 6 months before the diagnosis of CTD varied between 18.9% (IIM) and 27.2% (pSS) (Table 1) and the proportion of patients for whom ILD was identified more than 6 months after the CTD varied between 16.9% (IIM) and 44.2% (RA). For the remaining patients, ILD was identified in the 6-month period before and after the CTD diagnosis. After adjustment for age and sex, a significant increase in mortality was observed in patients with ILD (whether progressive or not) in all six CTDs compared to patients with CTD without ILD (Figure 1).
Conclusion: The results of this nation-wide population-based study underline the significant mortality risk associated with ILD among patients with CTD. These results can inform additional strategies for screening, diagnosis and follow-up of ILD in patients with CTDs.
REFERENCES: [1] Nasser M, Larrieu S, Boussel L, Si-Mohamed S, Bazin F, Marque S, Massol J, Thivolet-Bejui F, Chalabreysse L, Maucort-Boulch D, Hachulla E, Jouneau S, Le Lay K, Cottin V. Estimates of epidemiology, mortality and disease burden associated with progressive fibrosing interstitial lung disease in France (the PROGRESS study). Respir Res. 2021;22:162.
Table 1. Number and proportion of patients with the CTD, a CTD-ILD and a PPF.
Overmortality of patients with a CTD-ILD
Acknowledgements: NIL.
Disclosure of Interests: Laurent Arnaud Abbvie, AlfaSigma, Alpine, Astra-Zeneca, Biogen, BMS, Boehringer-Ingelheim, Chugaï, GSK, Grifols, Janssen, Kezar, LFB, Lilly, Medac, Merck, Novartis, Novo-Nordisk, Pfizer, Roche, UCB, Abbvie, AlfaSigma, Alpine, Astra-Zeneca, Biogen, BMS, Boehringer-Ingelheim, Chugaï, GSK, Grifols, Janssen, Kezar, LFB, Lilly, Medac, Merck, Novartis, Novo-Nordisk, Pfizer, Roche, UCB, Yurdagul Uzunhan Boehinger Ingelheim, Pfizer, GSK, Sanofi and CSL Vifor and support for attending meetings from Oxyvie, Boehinger Ingelheim, Pfizer, GSK, Sanofi and CSL Vifor and support for attending meetings from Oxyvie, Boehinger Ingelheim, Pfizer, GSK, Sanofi and CSL Vifor and support for attending meetings from Oxyvie, Boehinger Ingelheim, Pfizer, GSK, Sanofi and CSL Vifor and support for attending meetings from Oxyvie, Pierre-Louis Lleu Boehringer Ingelhelm Paris, France, Stéphane Bouée Boehringer Ingenheim for this study, Theo Cerceau Boehringer Ingenheim for this study, Chloé Grimal Boehringer Ingenheim for this study, Vincent Cottin Abbvie, Avalyn, Boehringer Ingelheim, BMS, CSL, Ferrer/United Therapeutics, Gossamer, Liquidia, Pliant, Pulmovant, PureTech, Roche, Sanofi, Shionogi.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (