

Background: Glucocorticoids (GCs) are widely used in the treatment of various diseases, including rheumatic diseases, and improve their outcomes. However, GCs cause bone loss and bone fragility by increasing early bone resorption followed by the progression of impaired bone formation. The Wnt/β-catenin signaling pathway is involved in increasing bone formation and suppressing bone resorption and is thought to be one of the pathogenic mechanisms of glucocorticoid-induced osteoporosis (GIOP). We previously reported that GC therapy increased serum sclerostin, an inhibitor of Wnt signaling, and decreased Wnt3a, a ligand for Wnt signaling, suggesting the potential of inhibiting sclerostin as a treatment for GIOP [1,2]. Romosozumab (ROMO), a monoclonal antibody that binds to sclerostin, has a unique dual effect of increasing bone formation and decreasing bone resorption. Large randomized controlled trials on postmenopausal osteoporosis patients showed that bone mineral density (BMD) was higher with ROMO than with a placebo or bisphosphonates (BP) after 12 months of treatment. We recently presented that ROMO increased lumbar spine BMD over one year compared to denosumab (DMAb) or BP in patients with rheumatic diseases newly treated with GCs [3], but the long-term effect in patients started on ROMO is not clear. Since ROMO administration is restricted to one year, DMAb has been shown to be effective in postmenopausal osteoporosis as sequential therapy following ROMO [4]. However, the efficacy of sequential therapy with DMAb following ROMO in GIOP is not clear.
Objectives: The purpose of this study is to evaluate the long-term efficacy in BMD changes of ROMO-DMAb (12-month ROMO followed by 24-month DMAb) for GIOP compared to 36-month DMAb or BP, in patients starting GC therapy.
Methods: This was a randomized, open-label, prospective study. Patients with rheumatic diseases who had not previously received GCs or osteoporosis treatment and newly started treatment with prednisolone (PSL) at 15 mg/day or higher were recruited. Eligible patients were randomly assigned to receive either ROMO (ROMO-DMAb group), DMAb, or BP using a dynamic allocation randomization approach. Stratification factors for assignment were age (≥65 years vs <65 years), sex, GC dose at the start of the study (PSL equivalent of ≥45 mg/day, ≥30 mg/day <45 mg/day, or <30 mg/day), and the T-score of the lower lumbar spine or femoral neck (≥-1.0, <-1.0 ≥-2.0, or <-2.0). After the 12-month restricted period of ROMO, the treatment in the ROMO-DMAb group was switched to DMAb. We measured BMD of the lumbar spine, femoral neck, and total hip every 6 months, and bone turnover markers and molecules regulating bone metabolism every 3 months for 36 months. We also assessed the incidence of new fractures, and adverse events.
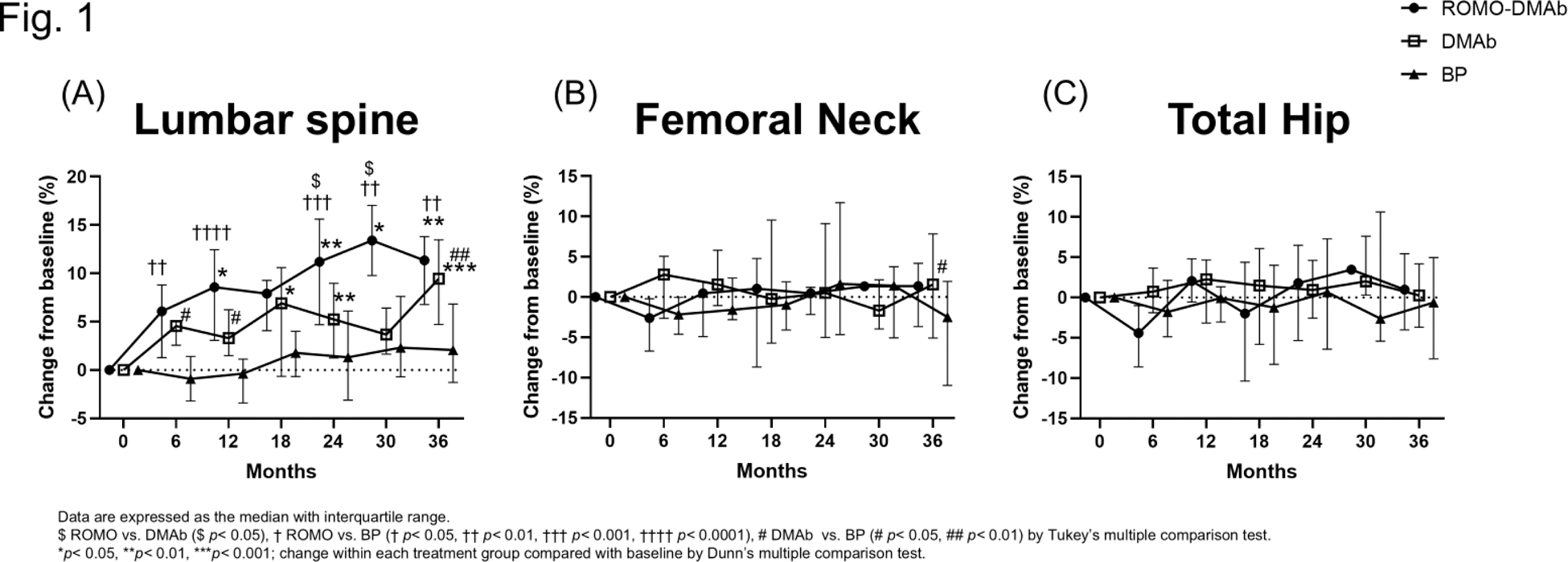
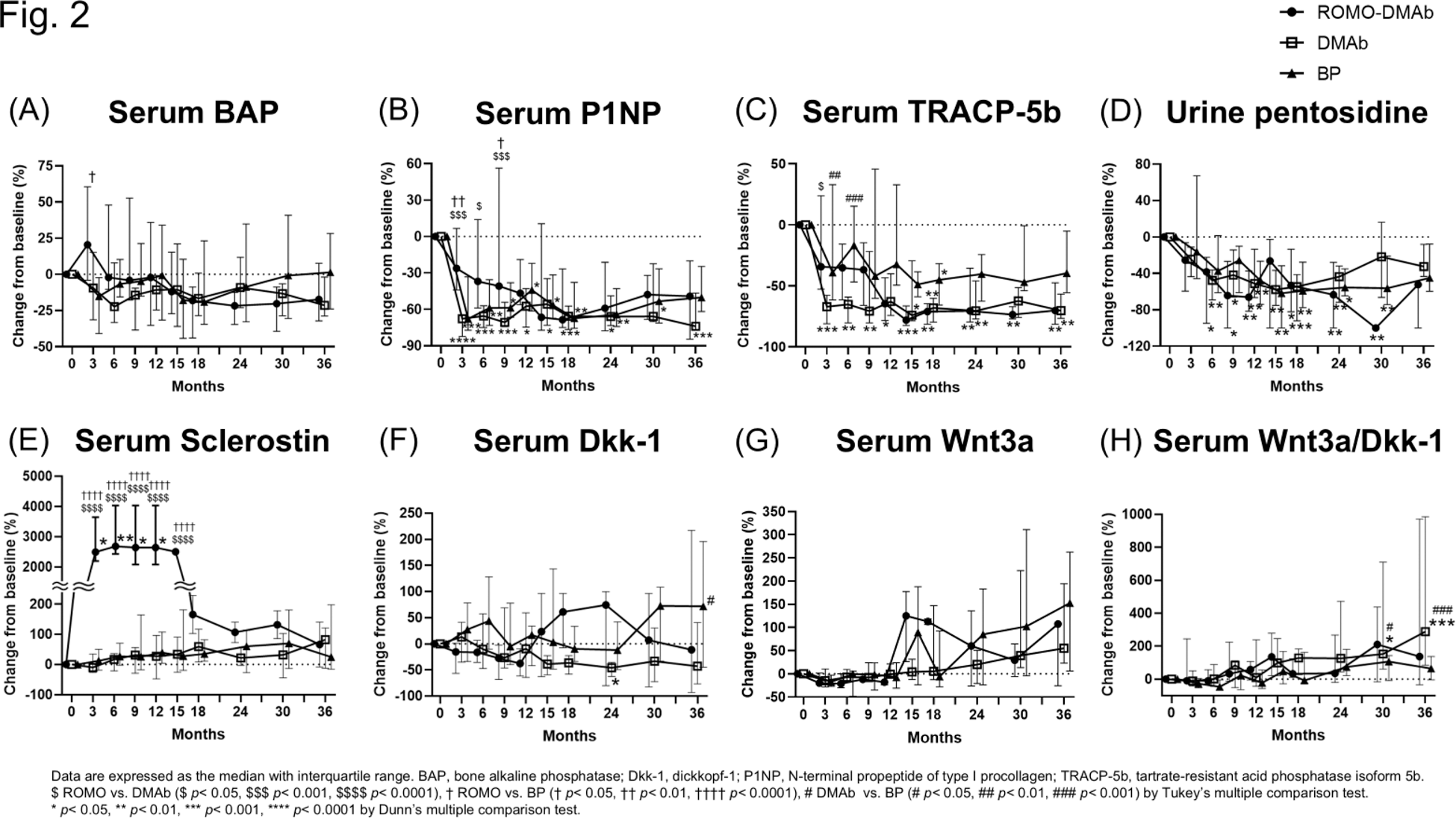
Results: Eleven patients were assigned to the ROMO-DMAb group, 14 to the DMAb group, and 14 to the BP group. The median age of all three groups was in the 70s (between 53 and 92 years), with more women, all of whom were postmenopausal. The median daily prednisolone dose ranged from 15.0 to 25.0 mg, with no significant differences among the three groups. The cumulative GC doses at 12, 24, and 36 months in each group were also not significantly different among the three groups. The median [25th percentile to 75th percentile] percent change in lumbar spine BMD from baseline at 36 months was greatest for the ROMO-DMAb group among the three groups (ROMO-DMAb; 11.3 [6.8 – 13.8] %, DMAb; 9.4 [4.7 – 13.5] %, BP; 2.1 [-1.3 – 6.8] %) (Figure 1). The BMD of both the ROMO-DMAb and DMAb groups increased significantly from baseline at most time points. At 36 months, the median percent change in total hip BMD was also greatest for the ROMO-DMAb group (ROMO-DMAb; 0.99 [-4.0 – 5.4] %, DMAb; 0.27 [-3.7 – 4.2] %, BP; -0.61 [-7.6 – 5.0] %), and the median percent change in femoral neck BMD was greatest for the DMAb group (ROMO-DMAb; 1.4 [-3.7 – 4.2] %, DMAb; 1.6 [-5.1 – 7.8] %, BP; -2.5 [-11.0 – 1.9] %). Serum bone alkaline phosphatase (BAP) level, a marker of bone formation, increased at 3 months only in the ROMO-DMAb group, but decreased thereafter, as in the other two groups (Figure 2). Serum N-terminal propeptide of type I procollagen (P1NP) level, another marker of bone formation, decreased in all three groups, but the change was smaller in the ROMO-DMAb group. Serum BAP and P1NP in the ROMO-DMAb group decreased rapidly at 15 months, probably because of the change to DMAb after 12 months, but showed an increasing trend from 24 months. Serum tartrate-resistant acid phosphatase isoform 5b, a marker of bone resorption, decreased in all groups. Urine pentosidine, a bone quality marker, decreased in all groups. Serum Dickkopf-1 (Dkk-1), an inhibitor of Wnt signaling, Wnt3a, and also the ratios of Wnt3a to Dkk-1 increased from 15 months in the ROMO-DMAb group. The incidence of new fractures was not statistically different among the three groups.
Conclusion: The BMD of the lumbar spine, femoral neck, and total hip increased with ROMO-DMAb for 3 years, even under GC therapy. In particular, the BMD of the lumbar spine increased significantly compared to baseline and more than DMAb and BP. Furthermore, no significant differences were observed in the incidence of adverse effects, suggesting that long-term ROMO-DMAb treatment is effective and safe for GIOP.
REFERENCES: [1] Kawazoe M, et al. Clin Rheumatol 2018; 37: 2169-78.
[2] Kawazoe M, et al. Clin Rheumatol 2021; 40: 2947-54.
[3] Kawazoe M, et al. J Clin Endocrinol Metab 2024; Epub ahead of print.
[4] Cosman F, et al. N Engl J Med 2016; 375: 1532-43.
Acknowledgements: NIL.
Disclosure of Interests: Mai Kawazoe GlaxoSmithKline plc., Asahi Kasei Pharma Corp., Amgen K.K., Astellas Pharma Inc., AstraZeneca K.K., Chugai Pharmaceutical Co., Ltd., Kissei Pharmaceutical Co., Ltd., Mitsubishi Tanabe Pharma Corp., Nippon Boehringer Ingelheim Co., Ltd., Taiho Pharmaceutical Co., Ltd., and Taisho Pharmaceutical Co., Ltd., Kaichi Kaneko AbbVie GK, Astellas Pharma Inc., Chugai Pharmaceutical Co., Ltd., Daiichi Sankyo Co. Ltd., Eisai Co., Ltd., Eli Lilly Japan K.K., Janssen Pharmaceutical K.K., Mitsubishi Tanabe Pharma Co., Ltd., GlaxoSmithKline K.K., Pfizer Japan Inc., and Takeda Pharmaceutical Co. Ltd., Shotaro Masuoka: None declared, Soichi Yamada: None declared, Zento Yamada: None declared, Sei Muraoka Taisho Pharmaceutical, Karin Furukawa Asahi Kasei Pharma, Hiroshi Sato: None declared, Eri Watanabe GlaxoSmithKline plc., and Bayer Yakuhin Co., Ltd., Keiko Koshiba: None declared, Izumi Irita: None declared, Miwa Kanaji: None declared, Takahiko Sugihara AbbVie GK, Asahi Kasei Pharma Corp., Astellas Pharma Inc., Ayumi Pharmaceutical, Bristol Myers Squibb K.K., Chugai Pharmaceutical Co., Ltd., Eli Lilly Japan K.K., Mitsubishi-Tanabe Pharma Co., Ono Pharmaceutical, Pfizer Japan Inc., and Taisho Pharmaceutical Co., Ltd., Asahi Kasei Corp., Daiichi Sankyo Co., Ltd., Chugai Pharmaceutical Co., Ltd., and Ono Pharmaceutical., Junko Nishio Asahi Kasei Pharma Corp., and Taisho Pharmaceutical Co., Ltd., Asahi Kasei Pharma Corp., Chugai Pharmaceutical Co., Ltd., Ono Pharmaceutical Co., Ltd., AbbVie GK, GlaxoSmithKline plc., Nippon Boehringer Ingelheim Co., Ltd., Nippon Kayaku Co., Ltd., Ayumi Pharmaceutical Corp., Abbott Japan LLC, and Taisho Pharmaceutical Co., Ltd., Toshihiro Nanki Ono Pharmaceutical Co., Ltd., Chugai Pharmaceutical Co., Ltd., Eisai Co., Ltd., Astellas Pharma Inc., Janssen Pharmaceutical K.K., Pfizer Japan Inc., Asahi Kasei Pharma Corp., Eli Lilly Japan K.K., AbbVie GK, Takeda Pharmaceutical Co., Ltd., Ayumi Pharmaceutical Corp., Daiichi Sankyo Co., Ltd., Mitsubishi Tanabe Pharma Corp., UCB Japan Co. Ltd., Nippon Boehringer Ingelheim Co., Ltd., AstraZeneca K.K., GlaxoSmithKline plc., Novartis Pharma K.K. and Taisho Pharmaceutical Co., Ltd., UCB Japan Co., Ltd., Eisai Co., Ltd., and Chugai Pharmaceutical Co., Ltd., Chugai Pharmaceutical Co., Ltd., Eisai Co., Ltd., Eli Lilly Japan K.K., Bristol Myers Squibb Co., Ono Pharmaceutical Co., Ltd., Asahi Kasei Pharma Corp., Mitsubishi Tanabe Pharma Corp., Ayumi Pharmaceutical Corp., Nippon Kayaku Co., Ltd., AbbVie GK, Teijin Pharma Ltd., Taisho Pharmaceutical Co., Ltd., Nippon Boehringer Ingelheim Co., Ltd., and Shionogi & Co., Ltd.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (