

Background: Giant cell arteritis (GCA) and polymyalgia rheumatica (PMR) are closely related, immune-mediated chronic inflammatory diseases that often occur concurrently in individuals over the age of 50 [1,2]. It has been proposed that these conditions share common pathophysiology under the term GCA-PMR spectrum disease [3]. GCA causes headache, scalp tenderness, jaw claudication, visual impairment and other ischemic complications, as well as extracranial events such as aortitis and aortic aneurysm [2,4]. PMR causes muscle pain and stiffness, especially in the neck, shoulders, and pelvis [1,2]. Both conditions are associated with constitutional symptoms and significantly impact quality of life. While glucocorticoids are the mainstay of treatment for both, relapses are common, and long-term use leads to significant side effects, highlighting the need for effective glucocorticoid-sparing therapies [2]. Secukinumab, a monoclonal antibody that selectively neutralizes interleukin (IL)-17A, has demonstrated efficacy and a favourable safety profile in immune-mediated diseases, including psoriasis, psoriatic arthritis, axial spondyloarthritis, and hidradenitis suppurativa [5-8]. In the phase 2 TitAIN study (NCT03765788), secukinumab treatment resulted in a higher sustained remission rate in GCA patients compared to placebo at week 28 (70% vs. 20%) with effects lasting 52 weeks (59% vs. 8%); safety outcomes were consistent with the known safety profile of secukinumab [9]. A phase 3 trial in GCA (GCAptAIN; NCT04930094) is underway. Secukinumab is also being evaluated in patients with PMR. REPLENISH (NCT05767034) is a phase 3 trial evaluating its efficacy and safety in relapsed PMR patients in combination with a prednisone taper regimen, with primary end point at 52 weeks.
Objectives: To perform a post hoc analysis of the TitAIN study to evaluate secukinumab treatment vs. placebo in the subgroup of GCA patients with PMR symptoms. This subgroup consisted of patients who had PMR symptoms within 12 weeks prior to and/or at baseline.
Methods: TitAIN was a randomised, double-blind, placebo-controlled, multicentre, phase 2 study in patients aged 50 years or older with new-onset or relapsing GCA (unequivocal cranial symptoms of GCA and/or symptoms of PMR) who were naive to biological therapy and already receiving glucocorticoids at baseline with a prednisolone equivalent dose of 25 to 60 mg/day [9]. Patients were assigned (1:1) to receive 300 mg secukinumab or placebo subcutaneously once a week up to week 4, and every 4 weeks thereafter. In both treatment arms, prednisolone dose was tapered down to 0 mg over a 26-week period [9]. Data are presented as observed.
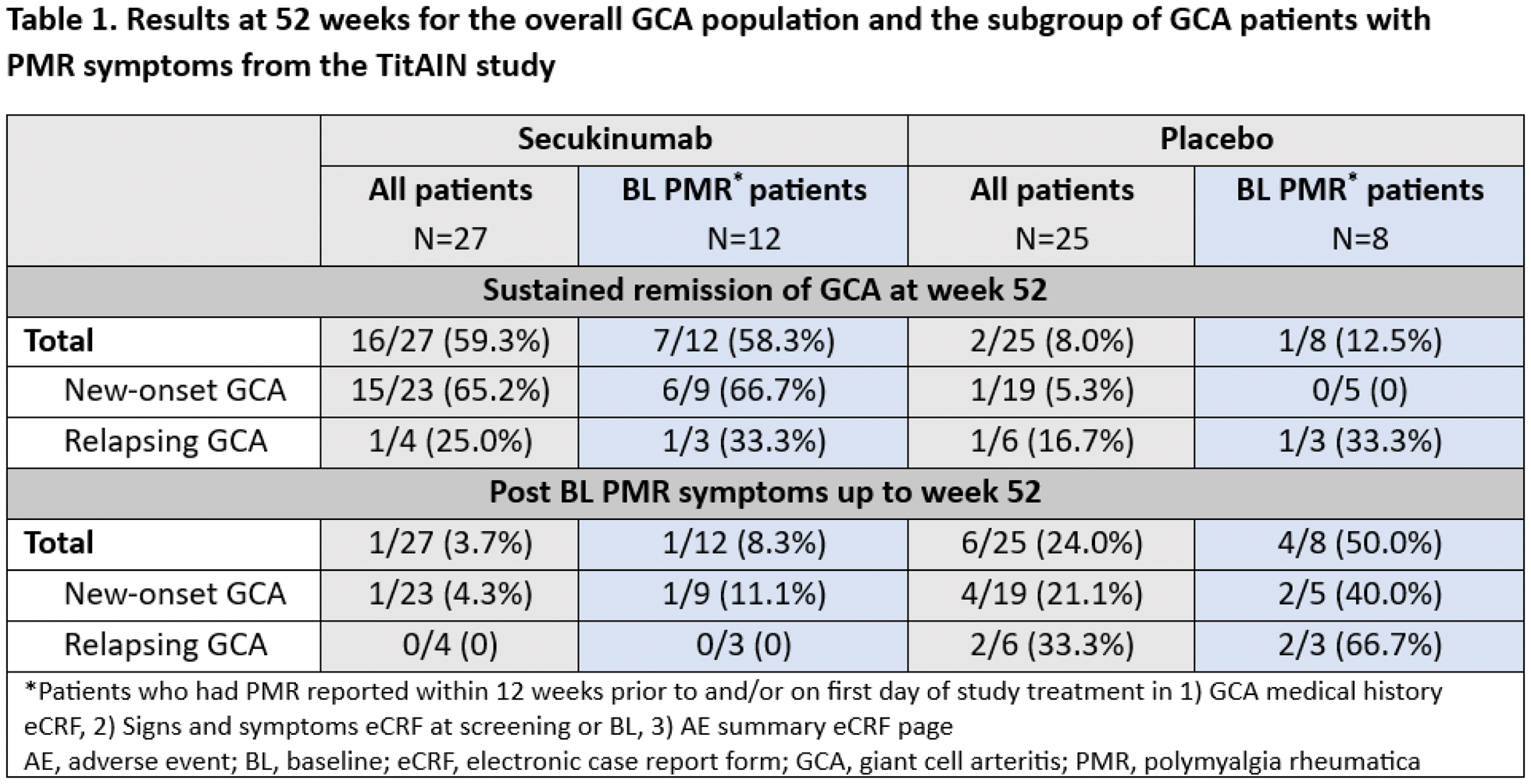
Results: In the TitAIN study, 27 patients received secukinumab and 25 patients received placebo; 12 of 27 patients (44.4%) in the secukinumab arm vs. 8 of 25 patients (32.0%) in the placebo arm had PMR symptoms and are included in this post hoc analysis. Among patients with PMR symptoms, 7 of 12 (58.3%) in the secukinumab arm vs. 1 of 8 (12.5%) in the placebo arm were in sustained GCA remission at week 52, in line with what has been reported previously for the overall study population (59% vs. 8%) [9]. Overall, only 1 of 27 patients (3.7%) in the secukinumab arm vs. 6 of 25 patients (24.0%) in the placebo arm experienced PMR symptoms post baseline up to week 52. In the subgroup of patients with PMR symptoms, only 1 of 12 (8.3%) patients in the secukinumab arm experienced PMR symptoms post baseline compared to 4 of 8 (50.0%) in the placebo arm (Table 1). Data for new-onset and relapsing GCA subgroups are presented in Table 1. All GCA patients with PMR symptoms had at least one adverse event (AE), across both treatment arms. The most common AEs in this subgroup were overall in line with those reported in the study population [9]. Serious AEs were reported in 8.3% of patients (n=1; facial paralysis) in the secukinumab arm and 37.5% of patients (n=3; gastrointestinal pain, dizziness, and chronic obstructive pulmonary disease) in the placebo arm.
Conclusion: This post hoc analysis shows a numerical reduction in patients experiencing PMR symptoms when treated with secukinumab compared to placebo. The safety profile of secukinumab in the subgroup of patients with PMR was similar to the overall GCA study population and consistent with its known safety profile. In addition to the results of the phase 2 TitAIN study fulfilling proof-of-concept efficacy for IL-17A inhibition with secukinumab in GCA,[9] these results further support the development of secukinumab for the treatment of PMR.
REFERENCES: [1] Espígol-Frigolé G, et al. Lancet . 2023;402(10411):1459-72.
[2] Man-Ger Sun M, et al. Best Pract Res Clin Rheumatol . 2022;36(4):101822.
[3] Tomelleri A, et al. Nat Rev Rheumatol . 2023;19(7):446-459.
[4] Mahr A, et al. Front. Med . 2021;8:732934.
[5] Blair HA, Drugs . 2021;81(4):483-494.
[6] Aparicio M, et al. Rheumatol Ther . 2022;9(1):73-94.
[7] Kimball AB, et al. Lancet . 2023;401(10378):747-761.
[8] Sun R, et al. Dermatol Ther (Heidelb ). 2024;14(3):729-743.
[9] Venhoff N, et al. Lancet Rheumatol. 2023;5:e341-50.
Acknowledgements: The authors thank Kshama Chitnis and Divya Chandrasekhar (Novartis, India) for providing editorial and medical writing support, and Alvaro Arjona Saz (Novartis, Spain) for providing medical and editorial guidance in accordance with Good Publication Practice 2022 guidelines.
Disclosure of Interests: Nils Venhoff declares speaker honorarium from AbbVie, AstraZeneca, Novartis, Boehringer Ingelheim, Bristol Myers Squibb, Chugai, Roche, UCB, GlaxoSmithKline, and Vifor, being an advisory board member for AbbVie, Chugai, Novartis, UCB, and Vifor, consulting fees from Novartis, AbbVie, Chugai, and Vifor, research grants from Bristol Myers Squibb and Novartis, meeting or travel grants from Bristol Myers Squibb, Novartis, AbbVie, and Vifor, expert testimony for Novartis, Vifor, and AbbVie, Wolfgang Schmidt is a part of speaker bureaus for AbbVie, Medac, Novartis, Roche, and Sanofi, is an advisory board member for AbbVie, Boehringer Ingelheim, GlaxoSmithKline, Novartis, and Sanofi, received research grants from AbbVie, GlaxoSmithKline, Novartis, and Sanofi, Raoul Bergner is part of speaker bureaus for AbbVie, Bristol Myers Squibb, Chugai, Novartis, Lilly, MSD, Janssen, Galapagos, and Glaxo Smith Kline, is an advisory board member for Galapagos, GlaxoSmithKline, Abbvie, and Vifor, received research grants from Vifor, and is an unpaid board member for Commission für student education—German Rheumatology Society, Jürgen Rech is part of speaker bureaus for AbbVie, Biogen, Bristol Myers Squibb, Chugai, GlaxoSmithKline, Janssen, Lilly, MSD, Novartis, Roche, Sanofi, Sobi, and UCB, received consulting fees from AbbVie, Biogen, Bristol Myers Squibb, Chugai, GlaxoSmithKline, Janssen, Lilly, MSD, Novartis, Roche, Sanofi, Sobi, and UCB, Leonore Unger received payments for speeches and seminar presentations from Novartis, Stephanie Finzel speaker honorarium from AbbVie, Alfasigma/Galapagos, Biotest, Celltrion, Chugai, Johnson&Johnson, Novartis, and UCB, received consulting fees from Johnson & Johnson, Novartis, NovoNordisk and UCB, meeting or travel grants from Biotest, Eli Lilly, Johnson&Johnson, Novartis, UCB, Galapagos, and Sobi, Ioana Andreica is part of speaker bureaus for AbbVie, Chugai, Gilead, Lilly, MSD, Novartis, Pfizer, Sobi, and UCB, received consulting fees from Amgen, Boehringer Ingelheim, Chugai, Galapagos, Lilly, Novartis, Pfizer, Sobi, Takeda, and UCB, received payments for other services (receipt of equipment, materials, drugs, medical writing, and gifts) from Novartis; and received research grants from Lilly, David M Kofler is an advisory board member for AbbVie and UCB, Stefan Weiner is an advisory board member for Novartis, received speaker honorarium from AbbVie, Amgen, AstraZeneca, Bayer, Bristol Myers Squibb, Daiichi-Sankyo, MSD, Novartis, Otsuka, Pfizer, and Roche, received meeting or travel grants from Abbvie, Amgen, Bristol Myers Squibb, Otsuka, Pfizer, and UCB, and is an unpaid board member for Saarländisch-Pfälzische Internistengesellschaft and Arbeitskreis Nephrologie Saar-Pfalz Mosel, Peter Lamprecht received speaker honorarium from AstraZeneca, Boehringer Ingelheim, Bristol Myers Squibb, GlaxoSmithKline, Janssen, Novartis, UCB, and Vifor, is an advisory board member for AstraZeneca, GlaxoSmithKline, UCB, and Vifor, received research grants from Bundesministerium für Bildung und Forschung, Deutsche Forschungsgemeinschaft, John Grube Foundation, and Vifor, Hendrik Schulze-Koops is part of speaker bureaus for AbbVie, Bristol Myers Squibb, Chugai, GlaxoSmithKline, Janssen, Medac, Novartis, Pfizer, Roche, Sanofi, and UCB, is an advisory board member for AbbVie, Bristol Myers Squibb, Chugai, Janssen, Medac, Pfizer, Novartis, Roche, Sanofi, and UCB, received research grants from AbbVie and Novartis, Meryl Mendelson shareholder of Novartis, employee of Novartis, Weibin Bao shareholder of Novartis, employee of Novartis, Monica Keyport shareholder of Novartis, employee of Novartis, Meron Maricos shareholder of Novartis, employee of Novartis, Valeria Jordan Mondragon shareholder of Novartis, employee of Novartis, Jens Thiel declares speaker honorarium from Novartis, GlaxoSmithKline, Bristol Myers Squibb, Roche, AstraZeneca, and Vifor, is an advisory board member for Novartis; received consulting fees from Novartis, Janssen, and GlaxoSmithKline, received research grants from Bristol Myers Squibb and Novartis.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (