

Background: A EULAR task force recently published a systematic review and meta-analysis on targeted therapies in patients with inflammatory arthritis and a history of cancer as well as points to consider when initiating targeted therapies in these patients [1,2]. The task force’s meta-analysis showed no indications of increased risk of new primary cancers or cancer recurrence with all biological disease-modifying anti-rheumatic drugs (bDMARDs) combined, nor for tumour necrosis factor inhibitors (TNFi) or rituximab individually [1]. However, in patients with rheumatoid arthritis (RA) and a solid cancer in remission, safety concerns relating to the immunomodulatory nature of bDMARDs primarily pertain to the risk of cancer recurrence. Despite this, only few of the observational studies in the systematic review and meta-analysis focused solely on cancer recurrence risk, with most studies investigating new primary cancers or pooling together cancer recurrence and new primary cancers into one outcome.
Objectives: To investigate if treatment with any type of bDMARD, TNFi, and rituximab, respectively, in patients with RA and a solid cancer in remission was associated with an increased risk of cancer recurrence compared with patients who received only conventional synthetic DMARDs (csDMARDs).
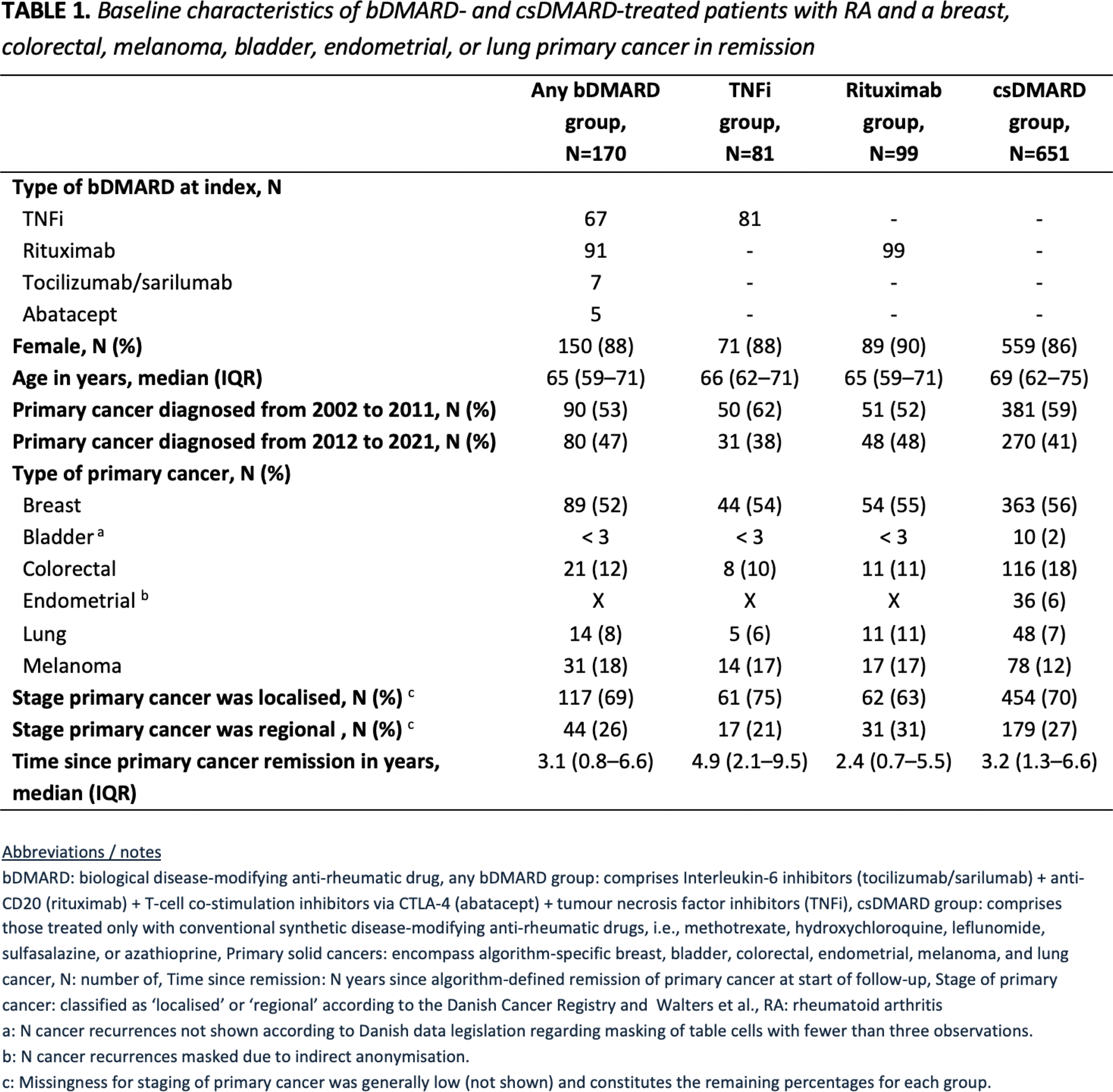
Methods: We performed a nationwide observational cohort study of Danish RA patients and one of six algorithm-specific solid cancers in remission who initiated treatment with a bDMARD or a csDMARD from 2002 to 2021. All patients with RA were identified in the Danish Rheumatology Quality Register (DANBIO). We utilised six validated and published cancer-specific algorithms to define cancer remission and to identify recurrences of breast, colorectal, melanoma, bladder, endometrial, and lung cancer in nationwide Danish health registers. These algorithms have demonstrated positive predictive values ranging between 86% and 95% for the identification of cancer recurrences. We defined three bDMARD exposure groups and one csDMARD comparator group according to the type(s) of DMARD initiated after algorithm-defined cancer remission: 1) any bDMARD, i.e., TNFi, rituximab, tocilizumab/sarilumab, or abatacept, 2) TNFi, 3) rituximab, and 4) csDMARD. Employing an ‘ever-treated’ approach, patients were followed from the corresponding DMARD initiation date (index date) and until: cancer recurrence, new primary cancer, death, emigration, bDMARD initiation (csDMARD group censoring only), or 31 December 2021. Hence, the csDMARD-treated group was naïve to bDMARD treatment after algorithm-defined cancer remission. The primary outcome was a composite cancer recurrence definition of the six cancer types. With breast cancer being the most incident primary cancer in patients with RA, breast cancer recurrence was also assessed separately. Inverse probability of treatment weighting (IPTW) was used to account for covariate differences between treatment groups. IPTW models included age at index date, sex, calendar period of primary cancer diagnosis, stage of primary cancer, time since algorithm-defined cancer remission, and type of primary cancer. IPTW-weighted cause-specific Cox models with death as a competing risk were used to calculate hazard ratios (HRs) and 95% confidence intervals (CI) for cancer recurrence.
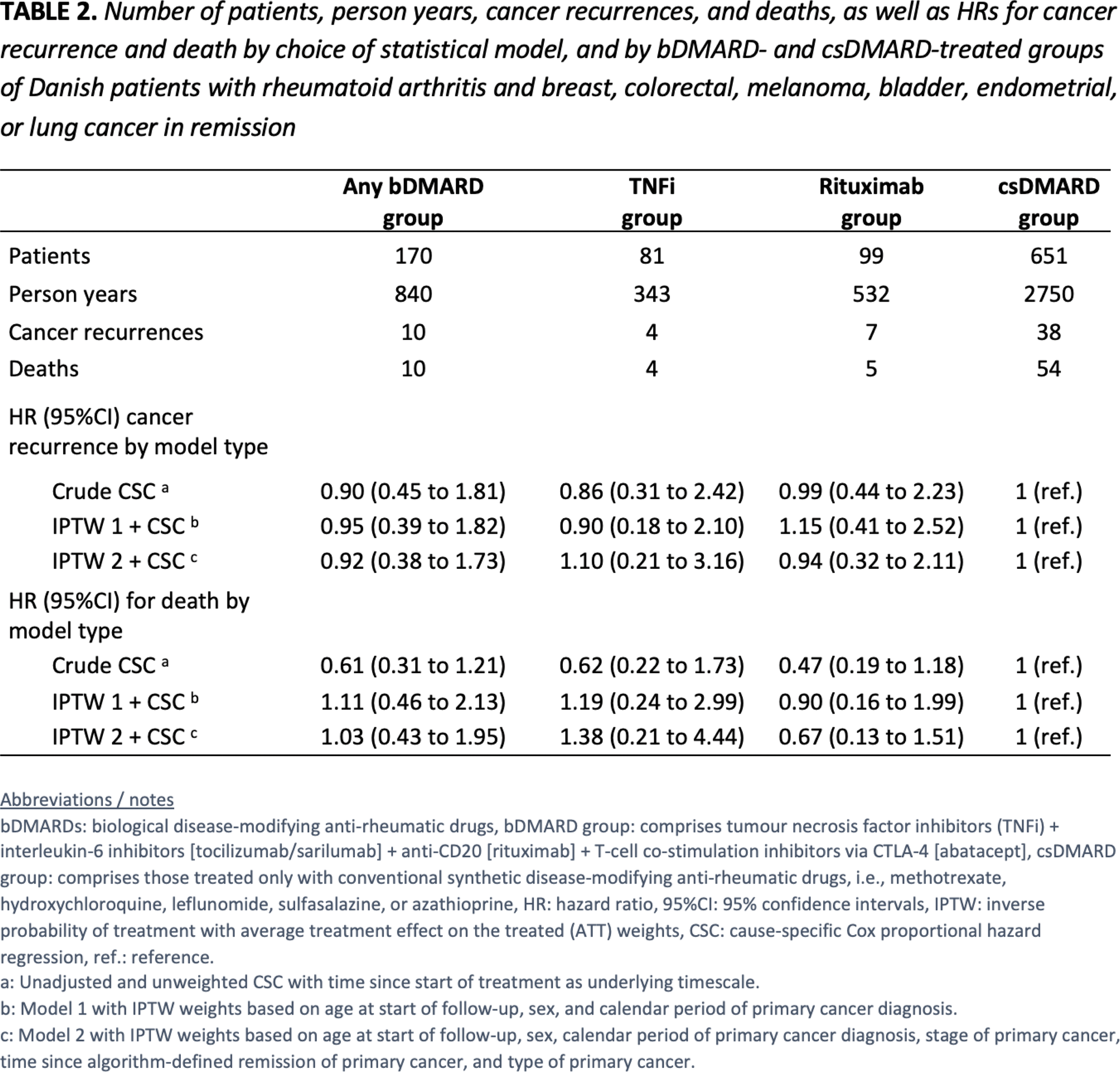
Results: Among 720 unique patients with RA and a solid cancer in remission, 170 initiated treatment with any bDMARD, 81 initiated a TNFi, 99 initiated rituximab, and 651 initiated a csDMARD after the date of algorithm-defined cancer remission. Of particular note, the TNFi group had a markedly higher median time since primary cancer remission (4.9 years) compared with the other groups. See Table 1. The crude incidence rates for cancer recurrence were 11.9 per 1000 person years (any bDMARD group), 11.6 (TNFi group), 13.2 (rituximab group), and 13.8 (csDMARD group). See Table 2. Compared with csDMARD treatment, we found no statistically significant increased HRs for cancer recurrence with any type of bDMARD 0.92 (95%CI 0.38 to 1.73), TNFi 1.10 (95%CI 0.21 to 3.16), or rituximab 0.94 (95%CI 0.32 to 2.11). Assessing breast cancer recurrence separately, no increased HRs were demonstrated for any of the three bDMARD exposure groups. Data not shown .
Conclusion: Treatment with bDMARDs as used in Danish clinical practice in patients with RA and a solid cancer in remission was not associated with increased risks of cancer recurrence compared with csDMARD treatment. This study corroborates the findings made by the EULAR task force regarding bDMARD treatment in patients with a history of cancer, and—in line with the EULAR task force’s call for improved reporting within this research area in respect of differentiating cancer recurrence from new primary cancers—constitutes one of the first studies with an explicit definition of cancer recurrence risk. However, in view of the limited study power we cannot rule out a clinically important excess risk of cancer recurrence with bDMARD treatment in RA patients.
REFERENCES: [1] Sebbag E, Molina-Collada J, Ndoye R, et al. Systematic literature review and meta-analysis informing the EULAR points to consider on the initiation of targeted therapies in patients with inflammatory arthritis and a history of cancer. Ann Rheum Dis 2024 2025/01/01. DOI: 10.1136/ard-2024-225981.
[2] Sebbag E, Lauper K, Molina-Collada J, et al. 2024 EULAR points to consider on the initiation of targeted therapies in patients with inflammatory arthritis and a history of cancer. Ann Rheum Dis 2024 2025/01/01. DOI: 10.1136/ard-2024-225982.
Acknowledgements: The authors would like to acknowledge all Danish departments of rheumatology for reporting and contributing to the DANBIO register. We also thank the Danish Cancer Society and the Danish Rheumatism Association for providing funding for the study.
Disclosure of Interests: Rasmus Westermann: None declared, René Lindholm Cordtz is employed by Novo Nordisk outside of the present study, Kirsten S. Duch: None declared, Lene Mellemkjær has an immediate family member employed at Novo Nordisk and has an immediate family member who owns stocks in Novo Nordisk, Merete Lund Hetland has received research grants (paid to her institution) from AbbVie, BMS, Eli Lilly Denmark A/S, MSD, Novartis, Pfizer, Sandoz, Nordforsk, UCB; honoraria (paid to her institution) from Pfizer, Medac, Sandoz, Novartis and UCB; participated in an advisory board (AbbVie) (paid to her institution). MLH co-chairs EuroSpA, which generates real-world evidence of treatment of psoriatic arthritis and axial spondylorthritis based on secondary data and is partly funded by Novartis and UCB, Linda A Rasmussen: None declared, Lene Dreyer has received research grant (paid to her institution) from BMS and Abbvie outside the current study. She is member of the steering committee of the Danish Rheumatology Quality Registry (DANBIO, DRQ), which receives public funding from the hospital owners and funding from pharmaceutical companies.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (