

Background: In psoriatic arthritis (PsA), EULAR treatment recommendations advise a treat-to-target approach and suggest a more intensive therapy for those with poor prognostic factors. Two recent studies [1,2] showed no significant benefit of early biologics over standard step-up care with methotrexate, but these studies did not select for poor prognosis.
Objectives: The aim of the Severe Psoriatic arthritis – Early intervEntion to control Disease (SPEED) trial was to compare PASDAS response in PsA patients with poor prognostic factors treated with standard step-up conventional systemic disease-modifying anti-rheumatic drugs (csDMARDs), combination csDMARDs or early tumour necrosis factor inhibitor (TNFi) induction therapy.
Methods: Participants were identified from eligible patients in the MONITOR inception cohort of PsA using a Trials Within Cohorts design. Patients with newly diagnosed PsA and ≥1 poor prognostic factor (polyarthritis, CRP>5mg/dL, HAQ>1, radiographic erosions) were randomised equally and open label to standard care with ‘step up’ csDMARD therapy, combination csDMARDs (methotrexate plus sulfasalazine/leflunomide) or early TNFi induction therapy (methotrexate plus adalimumab for 24 weeks) (Figure 1). Randomisation was stratified by centre, arthritis subtype (poly vs oligoarticular) and symptom duration (</≥12 months). The primary endpoint was the mean PsA disease activity score (PASDAS) at 24 weeks (intention-to-treat analysis). A sample size of 192 subjects (64 per arm) was needed to achieve 80% power with level of significance 0.05 (using PASDAS MCID=0.8, SD=1.5). A hierarchical method of testing was performed: An initial global test (ANOVA) was carried out to test the null hypothesis of no difference between the PASDAS mean scores in the three treatment groups. If the null hypothesis of the ANOVA was rejected, then two pairwise comparisons (combination csDMARDs versus step-up csDMARDs; early TNFi versus step-up csDMARDs) were performed. If they both rejected the null hypothesis of equality of PASDAS mean scores, a final pairwise comparison (combination csDMARDs versus early TNFi) was completed. Each pairwise comparisons fitted a linear mixed effect model of PASDAS score versus treatment adjusting for baseline PASDAS score, arthritis subtype and symptom duration as fixed effects and centre as random effect. Adjusted mean differences, 95% confidence intervals and p-values were calculated. Key secondary outcomes were analysed with the same hierarchical structure by fitting repeated measures mixed effect models of PASDAS score including both week 24 and week 48 timepoints with the same adjustments as for the primary outcome and with the addition of a treatment by time interaction. The results are based on a complete case analysis.
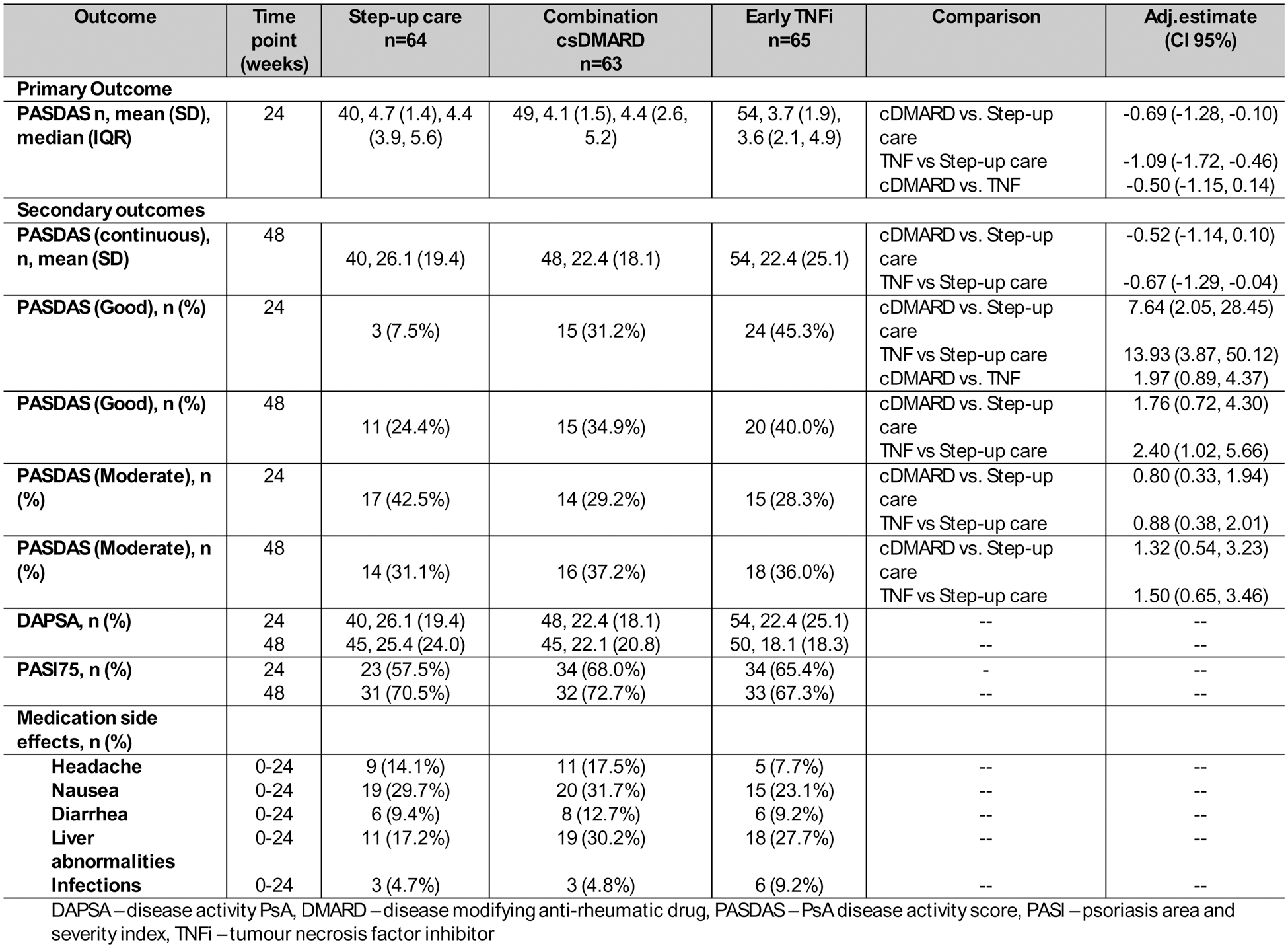
Results: From July 2019 to January 2024, 192 participants were recruited, 100 (52.1%) were male with average age (SD) of 47.6 (15.1) years. 159 (82.8%) had polyarticular disease with 33 (17.2%) oligoarthritis patients having CRP>5mg/dL (23, 69.7%), HAQ>1 (13, 39.4%) or erosive disease (8, 24.2%). Baseline mean (SD) PASDAS score was 5.6 (1.2), 5.6 (1.3) and 5.7 (1.1) in the step up, combination csDMARD and early TNFi groups, respectively. At week 24, an ANOVA global test rejected the null hypothesis of no difference in PASDAS mean score between treatment groups (p-value = 0.012). In pairwise testing at week 24, the early TNFi group showed evidence of a difference when compared to standard step-up care (PASDAS mean (SD) of 3.7 (1.9) vs 4.7 (1.4), adjusted mean difference -1.09 (95%CI -1.72, -0.46), p<0.001). The combination DMARD group showed evidence of a difference when compared to standard step-up care (PASDAS mean (SD) of 4.1 (1.5) vs 4.7 (1.4), adjusted mean difference -0.69 (95%CI -1.28, -0.10), p=0.02). There was no evidence of a difference comparing the early TNFi group to the combination DMARD group (adjusted mean difference -0.50 (-1.15, 0.14), p=0.127). At week 24, secondary outcomes showed that achievement of high-level responses such as PASDAS good response showed evidence of a benefit for either of the intervention arms compared to standard care. However, the proportion of participants meeting a PASDAS moderate response was similar. For exploratory outcomes, better responses in arthritis (DAPSA score) and skin (PASI response) are suggested in both intervention arms. At week 48, the benefit was only seen for early TNFi therapy. The early TNFi group showed evidence of a difference in continuous PASDAS and PASDAS good response when compared to standard step-up care (Table 1). Rescue therapy, where treatment was escalated beyond the initial randomised treatment was required before week 24, in 12 (27.9%) in the combination DMARD group and 14 (26.9%) in the early TNFi group. Only 4 serious adverse events occurred, 2 in the standard care group (cerebral haemorrage and perineal abscess) and 2 in the early TNF group (gastroenteritis, lower respiratory tract infection). Headache, nausea, diarrhoea and liver abnormalities were seen most frequently in the combination DMARD arm whilst infections were seen most frequently in the early TNF arm (9.2%).
Conclusion: These data show that initial intensive therapy with early biologics or combination DMARDs are superior for rapid control of disease for patients with early moderate-severe PsA. Even with only 6 months of early biologic therapy, better outcomes are maintained at one year in those initially receiving TNF inhibitor therapy. Intensive early therapy is associated with a likely small increase in non-serious side effects related to the medications.
REFERENCES: [1] De Marco et al. GolMePsA study [abstract]. Ann Rheum Dis 2024; 83: 153-154.
[2] Koc et al. STAMP study [abstract]. Arthritis Rheumatol 2024; 76 (suppl 9).
Table 1. Primary and secondary outcomes results.
The summary descriptive statistics for each treatment group are presented as n, mean (SD), median (IQR). For the primary outcome, the adjusted means, 95% CI and P-value for each comparison are reported (after fitting a linear mixed effect model adjusted for PASDAS at baseline, arthritis subtype and symptom duration as fixed effect and centre as random effect). For secondary outcomes, adjusted means (for PASDAS continuous at 48 weeks) and odds ratios (for PASDAS good and moderate response) with (95% Cis are reported after fitting a repeated measure model for PASDAS score including both week 24 and week 48 timepoints adjusted for PASDAS as baseline, arthritis subtype and symptom duration as fixed effect, centre as random effect and a treatment by time interaction). DAPSA aned PAS175 are reported descriptively via n, mean(SD) and n(%), respectively. Side effects are reported with n(%).
Acknowledgements: The SPEED trial project is an Investigator-Initiated Study supported by NIHR (via a National Institute for Health Research Clinician Scientist award) and Abbvie. This study is funded by the National Institute for Health and Care Research (NIHR) Oxford Biomedical Research Centre (BRC). The views expressed are those of the author(s) and not necessarily those of the NIHR or the Department of Health and Social Care.” Ethical approval for the study was granted by the South Central Research Ethics Committee (ref 18/SC/0107). Registration - ClinicalTrials.gov (NCT03739853) and EudraCT (2017-004542-24).
Disclosure of Interests: Sofia Massa: None declared, Elnaz Saeedi: None declared, Anne Francis: None declared, Nicola Gullick AbbVie, Celgene, Eli Lilly, Janssen, Novartis and UCB, AbbVie, Alfasigma, Celgene, Eli Lilly, Janssen, Novartis, and UCB, Abbvie, Astra Zeneca, Eli Lilly, Galapagos, Izana and Novartis, Deepak Jadon: None declared, William Tillett AbbVie, Amgen, Bristol Myers Squibb, Celgene, Eli Lilly, GSK, Janssen, Novartis, Ono-Pharma, Pfizer and UCB, AbbVie, Amgen, Bristol Myers Squibb, Celgene, Eli Lilly, GSK, Janssen, Novartis, Ono-Pharma, Pfizer and UCB, Abbvie, Celgene, Eli Lilly, Janssen, Pfizer and UCB, Raphael Hurtubise Biojamp, Celltrion, UCB, Jean-Guillaume Letarouilly Abbvie, Amgen, Biogen, BMS, Galapagos, Janssen, Lilly, Novartis, and Pfizer, AbbVie, Celltrion, Janssen, and MSD, Pfizer, Yvonne Sinomati: None declared, Laura Tucker: None declared, Nadia Mian: None declared, Laura C. Coates AbbVie, Amgen, Eli Lilly, Janssen, Novartis, Pfizer and UCB, AbbVie, Amgen, Bristol Myers Squibb, Eli Lilly, Enlivex, Janssen, Moonlake, Novartis, Pfizer, Takeda and UCB, Abbvie, Amgen, Janssen and UCB.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (