

Background: Radiographic axial spondyloarthritis (r-axSpA), also known as ankylosing spondylitis (AS), is a chronic inflammatory autoimmune disease. Interleukin-17A (IL-17A), a cytokine, plays a key role in the pathogenesis of AS. Xeligekimab (GR1501), a fully human monoclonal antibody that selectively neutralizes IL-17A, has demonstrated potential efficacy in treating r-axSpA. This study represents the first phase 3 clinical trial evaluating Xeligekimab as a treatment for r-axSpA.
Objectives: This phase 3 study aimed to determine the efficacy and safety of Xeligekimab for treating Chinese patients with active r-axSpA.
Methods: This was a 48-week, phase 3, multicenter study conducted in China, comprising a 16-week core treatment period, a 16-week maintenance period, and a 16-week follow-up period. Eligible patients were randomly assigned (1:1:1) to receive Xeligekimab 100 mg, eligekimab 200 mg, or placebo. Randomization was stratified by medication history (biologic-experienced vs. biologic-naïve) and weight (≥70 kg vs. <70 kg). At week 16, patients receiving placebo were re-randomized to receive either Xeligekimab 100 mg or 200 mg. The primary endpoint was the proportion of patients achieving an ASAS20 response at week 16. Secondary endpoints included assessments of disease activity and functional outcomes, evaluated using ASDAS-ESR, ASDAS-CRP, BASDAI, BASMI, and BASFI.
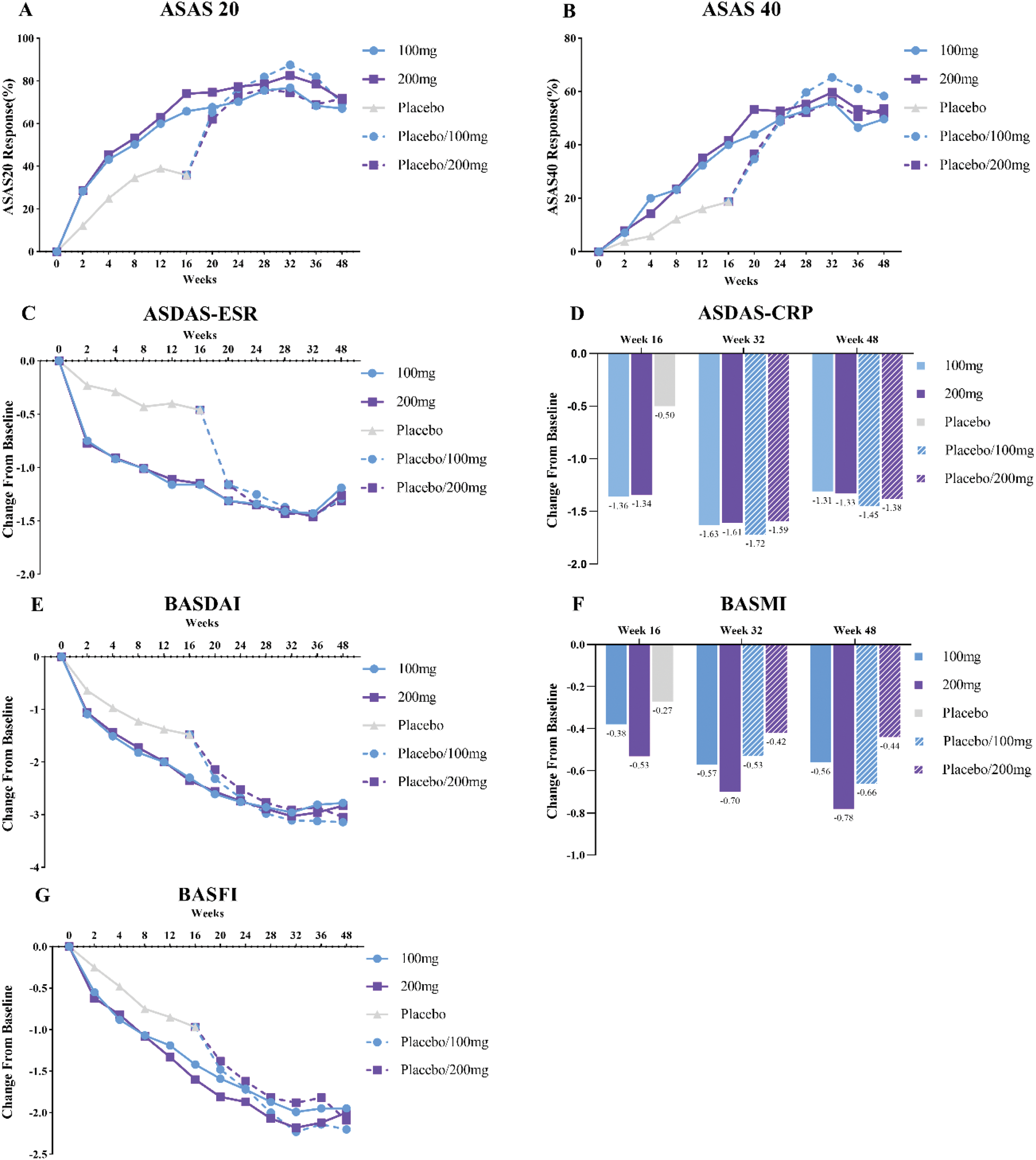
Results: A total of 465 patients were randomized to receive Xeligekimab 100 mg (n=155), 200 mg (n=156), or placebo (n=154) (Figure 1). Of these, 95.3% completed the week 16 core treatment period, and 88.3% completed the 48-week study. At week 16, a significantly higher proportion of patients in the Xeligekimab 200 mg group (n=114, 74.0%; P<0.001) and the Xeligekimab 100 mg group (n=102, 65.8%; P<0.001) achieved an ASAS20 response compared to the placebo group (n=56, 35.9%). Efficacy was sustained through week 48 in the Xeligekimab treatment groups (Figure 2). Significant reductions in disease activity, measured by ASDAS-ESR and ASDAS-CRP, were observed, along with improvements in physical function (BASDAI and BASFI) and spinal mobility (BASMI), with the 200 mg dose showing slightly greater benefits (Figure 2). Additionally, the placebo group demonstrated notable clinical improvement after switching to Xeligekimab at week 16, further confirming the drug’s efficacy in addressing disease activity, inflammation, and functional outcomes. Treatment-emergent adverse events (TEAEs) were comparable across all groups, with serious adverse events (SAEs) reported in 1.6% of patients during the core treatment period in the treatment groups. No unexpected safety concerns were identified up to week 48.
Conclusion: Xeligekimab showed significant efficacy in alleviating the clinical manifestations of active r-axSpA in Chinese patients at week 16, with sustained improvements observed through week 48 and no new safety concerns identified.
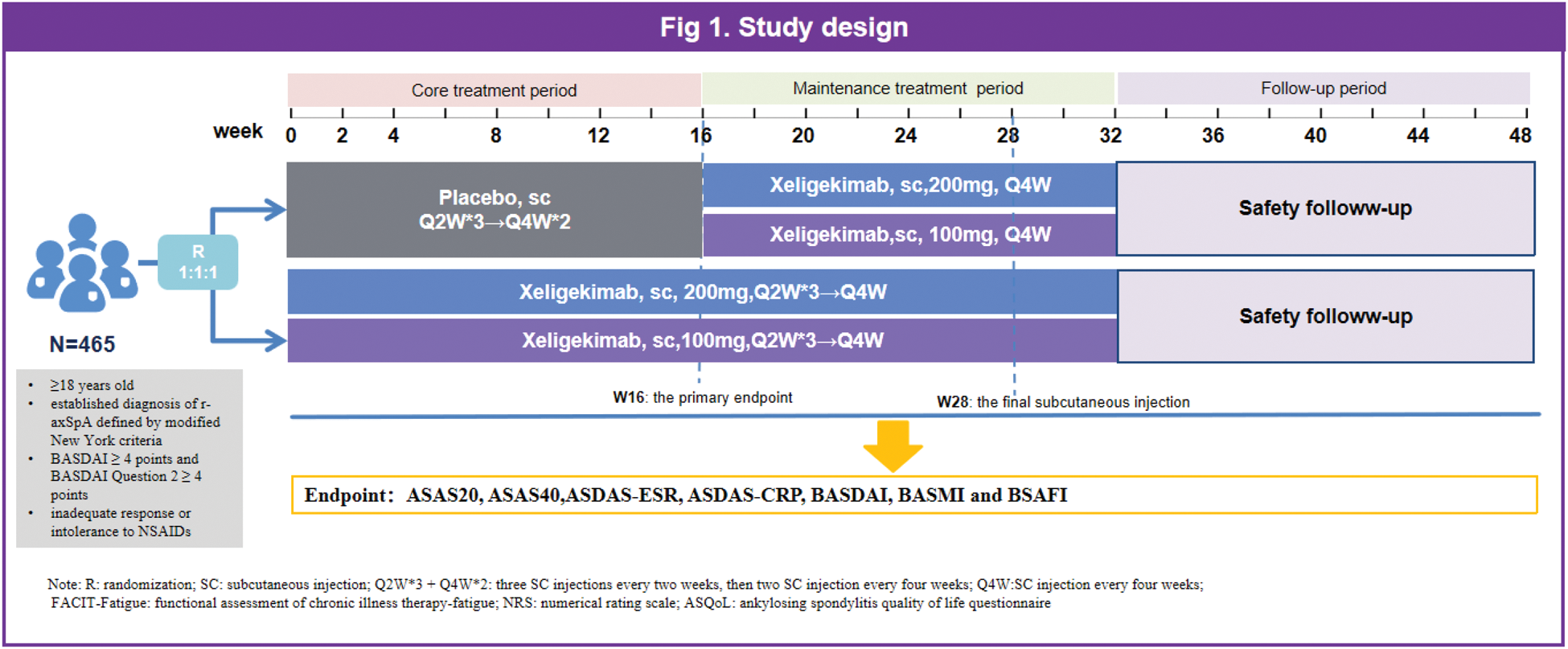
Schematic overview of the study cohort. Screening Phase (Week -4 to Week 0): Patients were screened for eligibility before randomization. Induction Dosing Period (Week 0 to Week 16): Patients were randomized (1:1:1) to receive 200 mg, 100 mg of GR1501, or placebo subcutaneously at Weeks 0, 2, 4, 8, 12, and 16. Maintenance Dosing Period (Week 16 to Week 32): Patients receiving GR1501 continued their respective doses, while placebo patients were re-randomized (1:1) to receive 100 mg or 200 mg of GR1501. Follow-up Period (Week 32 to Week 48): All patients entered a follow-up phase after Week 32.
Clinical efficacy and disease activity outcomes for patients with ankylosing spondylitis (AS) treated with varying doses of Xeligekimab over 48 weeks. The results include ASAS20 (Assessment of Spondyloarthritis International Society 20% improvement criteria) and ASAS40 (40% improvement criteria) response rates, changes in ASDAS-ESR (Ankylosing Spondylitis Disease Activity Score based on Erythrocyte Sedimentation Rate) and ASDAS-CRP (based on C-Reactive Protein), as well as improvements in BASDAI (Bath Ankylosing Spondylitis Disease Activity Index), BASMI (Bath Ankylosing Spondylitis Metrology Index), and BASFI (Bath Ankylosing Spondylitis Functional Index).
REFERENCES: NIL.
Acknowledgements: We sincerely appreciate the participation of the volunteers and the contributions of the staff to this study.
Disclosure of Interests: Shangzhu Zhang: None declared, Dong Xu: None declared, Xiaoxia Wang: None declared, li shujie: None declared, Shengyun Liu: None declared, Fenghong Yuan: None declared, Wei Gou: None declared, Baijie Xu: None declared, Lingyun Sun: None declared, Jieruo Gu: None declared, Dongmei Zhou: None declared, Xiaomei Li: None declared, Ning Kong: None declared, Yi Zhao: None declared, Jie Hao: None declared, Tianwang Li: None declared, Xiaoyun Fan: None declared, Qiang Shu: None declared, Hua Wei: None declared, Tao Jiang: None declared, Jing Yang: None declared, Long Qian: None declared, Hongsheng Sun: None declared, Xiaoyan Cai: None declared, Zhenyu Jiang: None declared, Guohua Yuan: None declared, li Qin: None declared, Min Yang: None declared, Jian Xu: None declared, Wenqiang Fan: None declared, Li Sun: None declared, Hua Zhang: None declared, Chunyan Zhang: None declared, Ning Zhang: None declared, Zhanyun Da: None declared, Jiankang Hu: None declared, Zhiyi Zhang: None declared, Jingchun Jin: None declared, Ju Liu: None declared, Lie Dai: None declared, Lingli Dong: None declared, Wei Wang Conflict of interest: Wei Wang is an employee of Chongqing Genrix Biopharmaceutical Co. Limited, Xiaofeng Zeng: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (