

Background: The 1-year double-blind period of the randomised MANDARA trial (NCT04157348) comparing anti-IL-5/R therapies demonstrated non-inferiority of benralizumab to mepolizumab for achieving remission in adults with relapsing/refractory EGPA.
Objectives: Report the combined 2-year data from the double-blind and first year of the ongoing open-label extension (OLE) periods of MANDARA.
Methods: On completion of the double-blind period (n=136/140) comparing benralizumab 1x30mg versus mepolizumab 3x100mg subcutaneously every 4 weeks, patients could enter the OLE, during which they either continued benralizumab (benra/benra) or switched from mepolizumab to benralizumab (mepo/benra). Endpoints included remission (Birmingham Vasculitis Activity Score [BVAS]=0 and oral glucocorticoid [OGC] dose ≤4 mg/day), use of OGC, relapses, blood eosinophil count, 6-item Asthma Control Questionnaire (ACQ-6), pre-bronchodilator forced expiratory volume in 1 second (pre-BD FEV 1 ), BVAS, and safety.
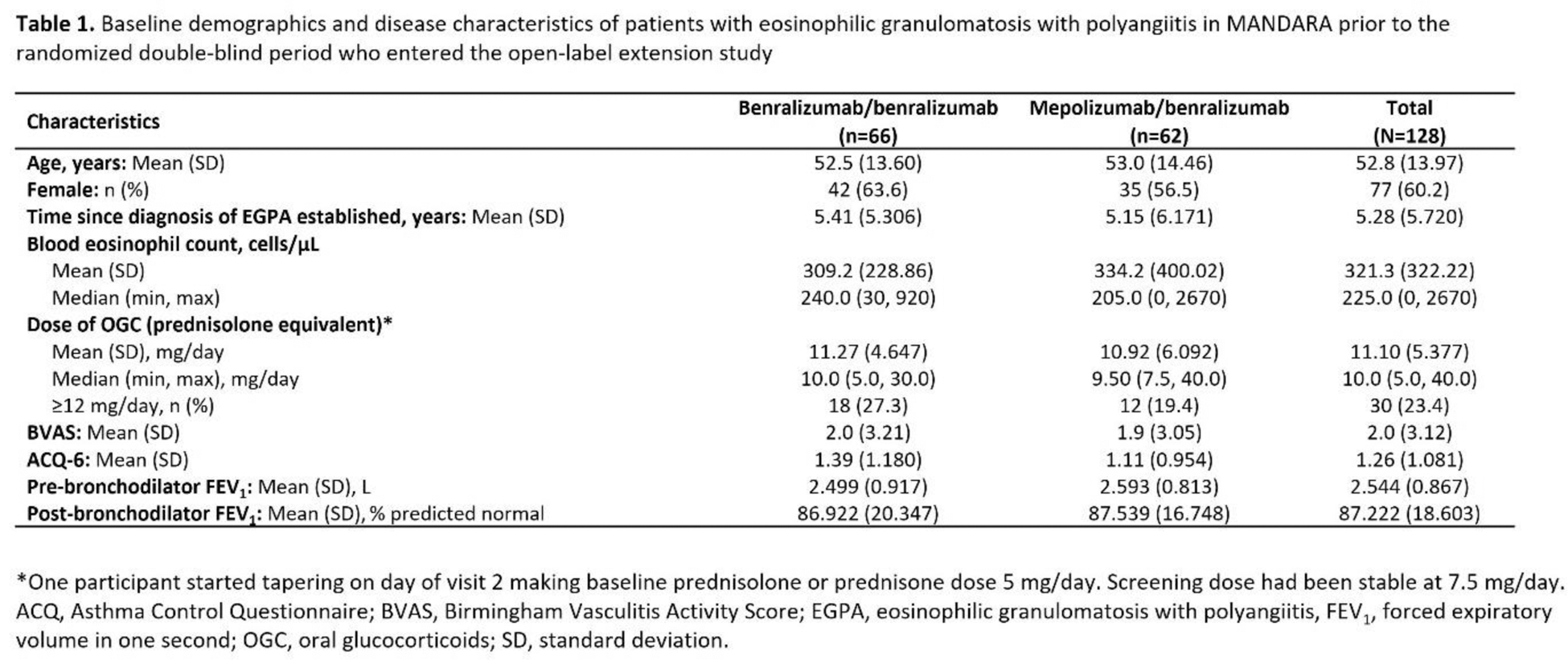
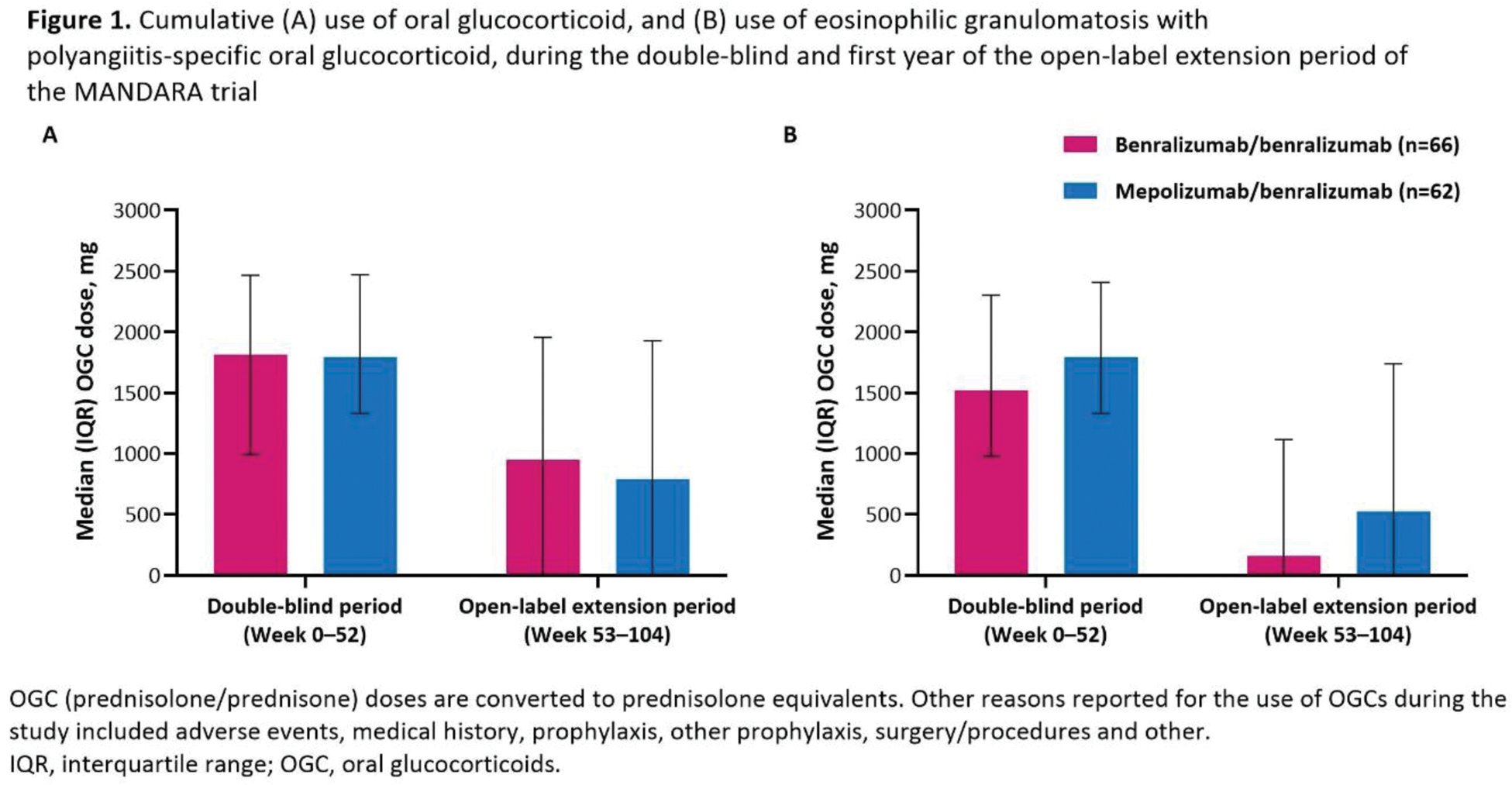
Results: In total, 128 patients entered the OLE (n=66 benra/benra, n=62 mepo/benra; mean age 52.8 years; 60.2% female); 119 completed OLE Year 1. Baseline characteristics of those entering the OLE were similar to those who entered the double-blind period (Table 1). At Week 104, 41 (62.1%) patients in the benra/benra group and 42 (67.7%) in the mepo/benra group achieved remission. During the first year of the OLE, 51 (77.3%) benra/benra, and 42 (67.7%) mepo/benra patients had no relapses. The percentage of patients in the benra/benra group who discontinued OGCs was similar at Weeks 49–52 (27 [40.9%]) and Weeks 101–104 (29 [43.9%]), while among the mepolizumab-treated patients who switched to benralizumab the percentage increased between Weeks 49–52 (16 [25.8%]) and Weeks 101–104 (27 [43.5%]). The median (IQR) OGC dose at Weeks 101–104 was 0.5 (0–5) mg/day in the benra/benra and 1.36 (0–5) mg/day in the mepo/benra group, corresponding to a median change from baseline of –96.7% and –90.5%, respectively. The median (IQR) cumulative use of OGC during the first year of the OLE was 950 (0–1955) mg and 791 (0–1928) mg in benra/benra and mepo/benra recipients respectively, while EGPA-related use of OGC was 159 (0–1114) mg and 523 (0–1739) mg (Figure 1). The median (IQR) blood eosinophil count in benra/benra-treated patients was 20 (10–40) cells/µL at both Weeks 52 and 100. Blood eosinophil count in the mepo/benra group reduced from 70 (40–90) cells/µL at Week 52 to 20 (10–50) cells/µL at Week 56, the first measurement after switching from mepolizumab to benralizumab. Mean (SD) change from study baseline in ACQ-6 scores were –0.78 (1.02) in the benra/benra group and –0.48 (0.96) in the mepo/benra group at Week 104. Mean (SD) change from study baseline in pre-BD FEV 1 was 0.03 (0.51) L and 0.02 (0.51) L in benra/benra- and mepo/benra-treated patients at Week 100, respectively. Mean (SD) change from study baseline in BVAS at Week 104 was similar between the benra/benra (–1.5 [4.16]) and mepo/benra (–1.6 [2.61]) patients. The most common adverse events during the double-blind and OLE periods were COVID-19, nasopharyngitis, and sinusitis. The safety of benralizumab was consistent with its known profile.
Conclusion: In patients with EGPA receiving benralizumab, remission rates, discontinuation of OGC, and blood eosinophil count depletion were durable over 104 weeks with a low rate of relapse, low EGPA disease activity, and without loss of control of asthma or decline of lung function. Additional depletion of blood eosinophil count and OGC-sparing effects were observed in patients switching from mepolizumab to benralizumab. These data suggest that anti-IL-5/R therapies have enduring benefits in patients with relapsing/refractory EGPA.
REFERENCES: NIL.
Acknowledgements: Medical writing support was provided by Caroline Ridley and Anna Mett of inScience Communications, Springer Healthcare Ltd, UK, which was funded by AstraZeneca. We thank the patients and their carers, and the investigators and site staff who participated in this study.
Disclosure of Interests: Peter A Merkel consulting and stock options in Kyverna, Q32 and Sparrow, consulting fees and research support from AbbVie, Amgen, AstraZeneca, Boehringer Ingelheim, Bristol Myers Squibb, GSK, InflaRx and Takeda; consulting fees only from argenx, Cabaletta, CSL Behring, Dynacure, HiBio, Janssen, Novartis, NS Pharma, Regeneron Pharmaceuticals and Visterra, research support only from Eicos, Electra, Forbius, Genentech/Roche, Neutrolis and Sanofi/Genzyme, Parameswaran Nair honoraria from Arrowhead, AstraZeneca, CSL Behring, GSK and Sanofi, institution received grant support from AstraZeneca, Cyclomedica, Equillium, Foresee, Genentech, Sanofi and Teva, Nader Khalidi consulting fees and research support from AbbVie, Bristol Myers Squibb and Sanofi; consulting fees only from GSK, Mallinckrodt Pharmaceuticals, Otsuka Pharmaceuticals and Roche, Benjamin Terrier consulting fees from AstraZeneca, GSK, Novartis and Vifor Pharma, Bernhard Hellmich speaker fees and/or consultancies from AbbVie, Amgen, AstraZeneca, Boehringer Ingelheim, Bristol Myers Squibb, Chugai, GSK, InflaRx, Janssen, MSD, Novartis, Pfizer, Phadia, Roche and CSL Vifor, Arnaud Bourdin consultancy fees and speaker fees from Amgen, AstraZeneca, Boehringer Ingelheim, Chiesi, GSK, Novartis and Sanofi Regeneron, research grants from AstraZeneca, Boehringer Ingelheim, and GSK, David R. W. Jayne speaker fees and/or consultancies from Amgen, AstraZeneca, Bristol Myers Squibb, Boehringer Ingelheim, ChemoCentryx, Chugai, GSK, Novartis, Roche, Takeda and Vifor Pharma, advisory board member for Aurinia, Chinook, GSK and Takeda, David J. Jackson consultancy fees and speakers’ fees from AstraZeneca, Boehringer Ingelheim, Chiesi, GSK, and Sanofi Regeneron, research grants from AstraZeneca, Florence Roufosse consulting fees from AstraZeneca, GSK, and Merck, Christian Pagnoux grants and personal speakers or advisory board fees from Roche, consulting and speaker fees from GSK, Otsuka, Pfizer and Roche; advisory board from AstraZeneca, GSK and Otsuka, educational grants from GSK, Otsuka and Pfizer, Ulrich Specks consulting fees from Amgen, argenx, AstraZeneca, Boehringer Ingelheim and CSL Vifor, research grants from Amgen, AstraZeneca, Bristol Myers Squibb, Genentech, GSK, NorthStar Medical Radioisotopes, NS Pharma and Novartis, Lena Börjesson Sjö employee of AstraZeneca and may own stock/stock options, Calvin N. Ho employee of AstraZeneca and may own stock/stock options, Maria Jison employee of AstraZeneca and may own stock/stock options, Christopher McCrae employee of AstraZeneca and may own stock/stock options, Sofia Necander employee of AstraZeneca and may own stock/stock options, Eva Rodríguez-Suárez employee of AstraZeneca and may own stock/stock options, Anat Shavit employee of AstraZeneca and may own stock/stock options, Claire Walton employee of AstraZeneca and may own stock/stock options, Michael Wechsler consulting, advisory, or speaking honoraria from the following: Allakos, Amgen, Areteia Therapeutics, Arrowhead Pharmaceutical, AstraZeneca, Avalo Therapeutics, Celldex, Connect Biopharma, Eli Lilly, Equillium, GSK, Incyte, Kinaset, Kymera, Merck, Phylaxis, Pulmatrix, Rapt Therapeutics, Recludix Pharma, Regeneron Pharmaceuticals, Roche/Genentech, Sanofi/Genzyme, Sentien, Sound Biologics, Tetherex Pharmaceuticals, Uniquity Bio, Upstream Bio, Verona Pharma and Zurabio.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (