

Background: The Anifrolumab Study of Treatment Effectiveness in the Real World (ASTER; NCT05637112), is a multi-national, prospective, observational study assessing the effectiveness of anifrolumab in patients with systemic lupus erythematosus (SLE) in routine clinical practice. Post-hoc analyses of the phase 3 TULIP-1/-2 and long-term extension (LTE) trials showed that, compared with placebo, remission according to the Definition of Remission in SLE (DORIS) and Lupus Low Disease Activity State (LLDAS), is more attainable and sustainable with anifrolumab treatment in addition to standard therapy in patients with SLE, with shorter time to first attainment and longer cumulative time in these states.[1,2] Here, we describe an interim analysis of DORIS remission and LLDAS attainment rates after 6 months of anifrolumab treatment in patients who participated in ASTER.
Objectives: To evaluate the effects of anifrolumab treatment on DORIS remission and LLDAS attainment in patients with SLE from the first 6 months of the ASTER study.
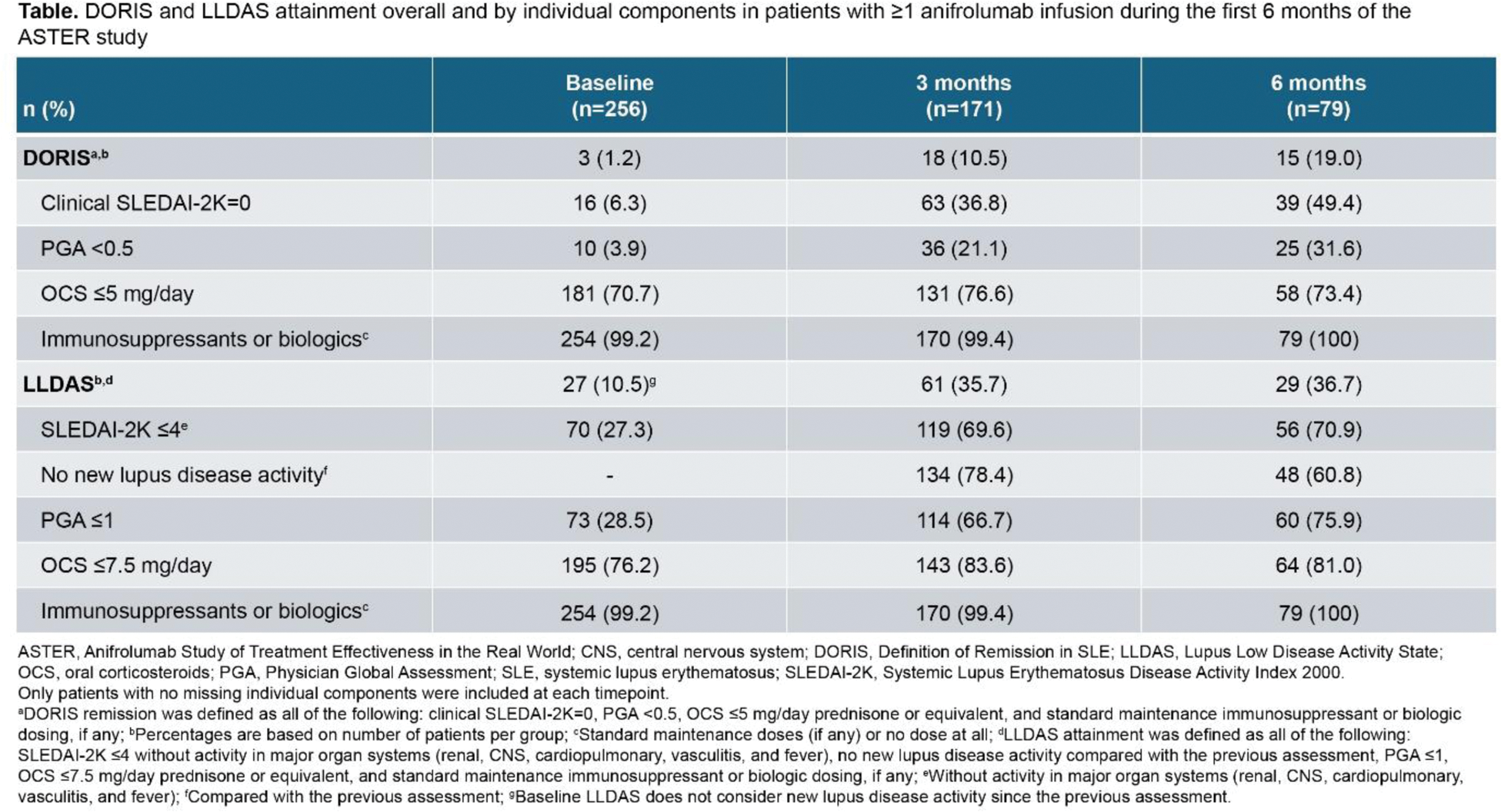
Methods: Eligible adults with SLE were enrolled at routine clinical visits before the first anifrolumab infusion date (index) and started anifrolumab treatment per the approved country-specific label at study sites. Baseline disease characteristics were collected retrospectively for 1 year prior to index. Data are reported as mean (standard deviation, SD) for time from SLE diagnosis, SLE Disease Activity Index 2000 (SLEDAI-2K), Physician Global Assessment (PGA), and Cutaneous Lupus Erythematosus Disease Area and Severity Index (CLASI) scores, and oral corticosteroid (OCS) daily dosage. Proportions of patients in DORIS remission, LLDAS, and the individual components of each were evaluated during the observed period at baseline and every 3 months up to 6 months in patients with ≥1 anifrolumab infusion; only patients with no missing individual components were included at each timepoint. The observed period was the time from the first anifrolumab infusion to study discontinuation or loss to follow-up. DORIS remission was defined by clinical SLEDAI-2K=0, PGA <0.5, OCS ≤5 mg/day prednisone or equivalent, and standard maintenance immunosuppressant or biologic dosing, if any. LLDAS was defined by all of the following criteria: SLEDAI-2K ≤4 with no activity in major organ systems, no new lupus disease activity compared with the previous assessment, PGA ≤1, OCS ≤7.5 mg/day prednisone or equivalent, and standard maintenance immunosuppressant or biologic dosing, if any.
Results: Among the 266 patients in the interim ASTER cohort, mean (SD) time from SLE diagnosis to index was 12.3 (9.6) years. At baseline, mean (SD) scores were SLEDAI-2K=7.4 (4.1), PGA=1.5 (0.6), CLASI activity=6.4 (7.2), and CLASI damage=3.3 (6.4). Among patients with available SDI data, about half (53.0%, n/N=133/251) had a baseline SDI score ≥1. Of the 260 patients with available SLEDAI-2K data, most patients (74.2%, 193/260) had a baseline SLEDAI-2K score <10. Overall, mean (SD) OCS dosage at baseline was 5.7 (8.6) mg/day (>5 mg/day: 28.9%, 77/266; >7.5 mg/day: 23.7%, 63/266); among patients with baseline OCS dosage >0 mg/day (n=144), mean dosage was 10.5 (9.2) mg/day. At baseline, 23.3% of patients (62/266) had prior use of a biologic, 13.2% (35/266) an immunosuppressant, and 30.8% (82/266) received at least one of these medications. Among the patients with no missing data for any DORIS remission components, rates of DORIS remission increased from 1.2% (3/256) at baseline to 10.5% (18/171) at 3 months and 19.0% (15/79) at 6 months after the first anifrolumab infusion (Table 1). Similarly, rates of LLDAS attainment increased from 10.5% (27/256) at baseline to 35.7% (61/171) at 3 months and 36.7% (29/79) at 6 months after the first anifrolumab infusion. Over 6 months of follow-up, the greatest increase in the proportion of patients achieving an individual DORIS component was in the clinical SLEDAI-2K=0 component (baseline: 6.3%, 16/256; 3 months: 36.8%, 63/171; and 6 months: 49.4%, 39/79); the greatest increase among LLDAS components was in the PGA ≤1 component (baseline: 28.5%, 73/256; 3 months: 66.7%, 114/171; and 6 months: 75.9%, 60/79) (Table 1).
Conclusion: In ASTER at baseline, rates of DORIS remission and LLDAS were low among patients receiving routine SLE care. Within 6 months of the first anifrolumab infusion, attainment rates for both states substantially increased. Though follow-up data beyond 6 months are needed to determine if these effects can be sustained, these findings suggest that both targets are achievable with anifrolumab treatment in a real-world setting.
REFERENCES: [1] Morand EF, et al. Ann Rheum Dis. 2023;82(5):639–45.
[2] Morand EF, et al. Ann Rheum Dis. Accepted for publication, November 2024.
Acknowledgements: This study was sponsored by AstraZeneca. Writing assistance was provided by Sheila Longo, PhD, of JK Associates Inc., part of Avalere Health, and funded by AstraZeneca.
Disclosure of Interests: Marta Mosca Speaker - Abbvie; AstraZeneca; GSK; Lilly; Otsuka; UCB, Consultant - AstraZeneca; Abbvie; GSK; BMS; Novartis; Biogen; Idorsia; UCB; Otsuka, Grant/Research Support - GSK, Zahir Amoura Consultant - Roche; GSK; AstraZeneca; Merck; Novartis, Grant/Research Support - Roche; GSK; AstraZeneca; Novartis, Julia Weinmann-Menke Speaker - Otsuka; Boehringer Ingelheim; Novartis; AstraZeneca; Vifor Pharma; Alexion, Consultant - Otsuka; Boehringer Ingelheim; Novartis; Astra-Zeneca; Vifor Pharma, Grant/Research Support - Grants paid to my institution to perform clinical trials and IITs, Mark Matsos Speaker - AstraZeneca, Consultant - AstraZeneca, Ioannis Parodis Speaker - Amgen; AstraZeneca; Aurinia Pharmaceuticals; Elly Lilly and Company; Gilead Sciences; GSK; Janssen Pharmaceuticals; Novartis; Otsuka Pharmaceutical; F. Hoffmann-La Roche AG, Grant/Research Support - Amgen; AstraZeneca; Aurinia Pharmaceuticals; Elly Lilly and Company; Gilead Sciences; GSK; Janssen Pharmaceuticals; Novartis; Otsuka Pharmaceutical; F. Hoffmann-La Roche AG, Danuta Kielar Shares/Stock - AstraZeneca, Current Employee - AstraZeneca, Jonatan Nåtman Shares/Stock - AstraZeneca, Current Employee - AstraZeneca, Stephanie Chen Shares/Stock - AstraZeneca, Current Employee - AstraZeneca, Cathy Emmas Shares/Stock - AstraZeneca, Current Employee - AstraZeneca, Miina Waratani Shares/Stock - AstraZeneca, Current Employee – AstraZeneca.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (