

Background: Over 500 million people burdened with osteoarthritis (OA) are without approved disease-modifying treatments. Interventional trials have previously failed to meet their primary endpoints, which can partly be attributed to the considerable heterogeneity of OA. It has been hypothesized that targeting patient subgroups, or endotypes, driven by pathways aligning with a drug’s mode-of-action, could enhance treatment responses. Developing a practical clinical approach to predict such treatable endotypes is urgently needed to enrich trials for the right OA subgroups and thereby maximize the likelihood of achieving meaningful therapeutic outcomes in OA. Three biomarker-based endotypes driven by structural damage, inflammation, and low tissue turnover have been described in 295 knee OA (KOA) participants from the IMI-APPROACH cohort. We recently showed that over half of the participants exhibited longitudinal endotype stability. However, the clinical relevance of these endotypes has yet to be established through their ability to predict differential responses to treatment.
Objectives: The purpose of this study was to: i) develop an actionable clinical tool with which to predict OA endotypes, and ii) assess in a post-hoc subgroup analysis their ability to predict differential treatment responses in three interventional trials.
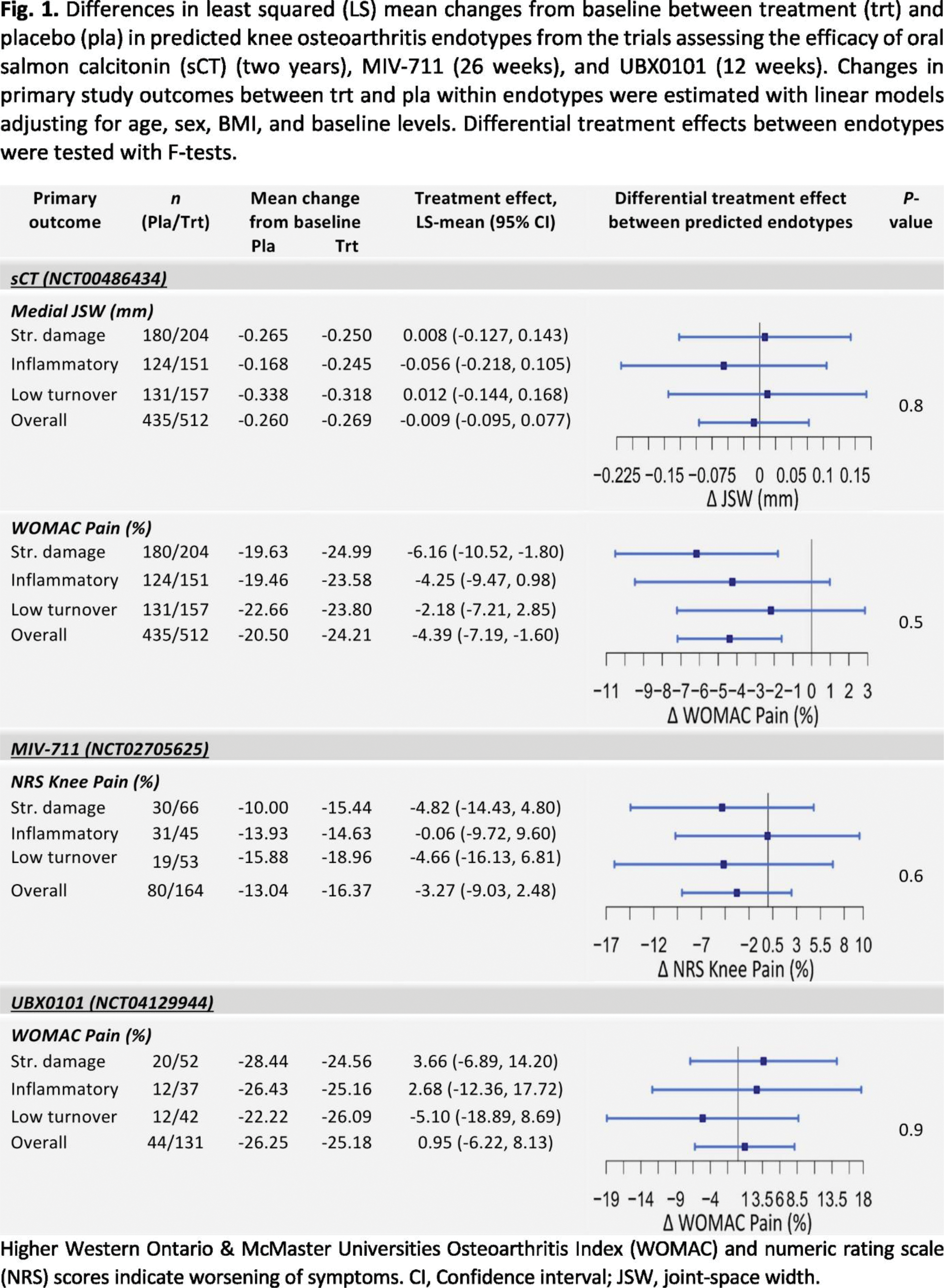
Methods: Longitudinal measures of 14 serum markers of KOA participants from the IMI-APPROACH study were included to develop a multi-class, logistic regression prediction model of structural damage, inflammation, and low turnover endotypes. Biomarkers were log-transformed, and z-score scaled. 75% of all participants were included for training and 25% of participants with longitudinally stable endotypes were used to assess predictions. To determine the optimal panel of six biomarkers, all marker combinations were assessed (3003 models). The model with the highest sensitivity across all endotypes (90%) included age, sex, BMI, and the biomarker panel; C2M, C3M, sCTX-I, N-MID, PRO-C2, and PRO-C4. Differential treatment response based on the endotypes was assessed in participants from the trials testing the efficacy of MIV-711 ( n = 244) (NCT02705625, cathepsin K inhibitor), oral salmon calcitonin (sCT) ( n = 947) (NCT00486434, bone anti-resorptive), and UBX0101 ( n = 175) (NCT04129944, senolytic inhibiting p53-MDM2). Multiple dosage groups were considered as one. Missing biomarker data at baseline (0.1% - 7.0%) were imputed with random forest models. Endotypes were predicted for each trial based on the model developed in IMI-APPROACH, using log-transformed, z-score scaled data at baseline. Changes from baseline of primary endpoints were considered, and mean differences in clinical outcomes of treatment versus placebo within each endotype were estimated with linear regression models adjusting for age, sex, BMI, and baseline levels. Differential treatment effects between endotypes were tested with F-tests.
Results: The structural damage, inflammatory, and low tissue turnover endotypes were predicted at baseline in the MIV-711, sCT, and UBX0101 trials using a multi-class logistic regression model based on a panel of six biomarkers (Figure 1). Overall, the trials included did not provide strong evidence for differential treatment effect between the endotypes. A number of potential signals were observed. For MIV-711, the predicted structural damage and low tissue turnover endotypes showed the highest numerical 26-week reduction of numeric rating scale (NRS) knee pain for treatment compared to placebo. For sCT, only the predicted inflammatory endotype was associated with a numerically increased two-year loss of radiographic joint-space width for treatment compared to placebo, while the structural damage endotype was associated with a significant reduction of WOMAC pain for treatment compared to placebo. For UBX0101, only the predicted low tissue turnover endotype exhibited a numerical 12-week reduction of WOMAC pain in response to UBX0101 treatment.
Conclusion: This study demonstrates the potential to predict OA endotypes using a small, targeted panel of biomarkers on trial data, offering a clinically practical tool to enhance trial recruitment. The observed responses to anti-resorptive treatments within OA subgroups highlight the potential benefits of aligning therapies with endotypes defined by the drug’s mode of action. Further investigation is needed to refine strategies for recruiting the right subgroups at the optimal time. Biomarker-based endotyping represents a promising approach to increasing the likelihood of successful trial outcomes.
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: Monica Toft Hannani: None declared, Peder Frederiksen: None declared, Morten Karsdal: None declared, Asger R. Bihlet: None declared, Karin Tunblad Shareholder of Medivir AB, Employed by Medivir AB, Fredrik Öberg Shareholder of Medivir AB, Employed by Medivir AB, Jamie Collins: None declared, Virginia B Kraus: None declared, David Hunter Provides consulting advice on scientific advisory boards for Haleon, TLCBio, Novartis, Tissuegene, Biobone, Sanofi, Enlivex, Jaume Bacardit: None declared, Anne-Christine Bay-Jensen: None declared, Christian Thudium: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (