

Background: Gout is a common inflammatory arthritis caused by hyperuricemia. Patients initiating urate-lowering therapy (ULT) are at risk of acute gout flares, given that fluctuations in serum uric acid levels can trigger the dissolution of urate crystals in the joints and surrounding soft tissues. Therefore, prophylactic treatment during the initial period of ULT is essential. Inhibition of the pro-inflammatory cytokine interleukin-1β (IL-1β) has been shown to reduce pain and inflammation in patients with gout. Genakumab, an IgG4 monoclonal antibody that selectively binds to and inhibits IL-1β, has demonstrated rapid pain relief and long-term prophylaxis against gout flares in Chinese adults undergoing gout treatment.
Objectives: To investigate the efficacy, safety, and immunogenicity of genakumab subcutaneous injection compared with colchicine for prophylaxis against gout flares in patients with gout initiating ULT.
Methods: This randomized, open label, active-controlled, phase 2 trial (NCT05936281) was conducted across 41 centers in China and included a 1-week screening period followed by a 12-week treatment period. Patients with gout aged 18–75 years were randomized 1:1:1 on Day 1 of the treatment period to receive a single subcutaneous injection of either genakumab 100 mg or 200 mg, or colchicine 0.5 mg/day orally for 12 weeks. ULT was initiated at baseline or 1 week prior to screening and was administered to all randomized patients for 12 weeks. ULT options included oral allopurinol 100 mg/day, febuxostat 40 mg/day, or benzbromarone 50 mg/day. Primary efficacy endpoints were assessed in the full analysis set (FAS; all randomized patients who had received ≥1 dose of the study drug and had ≥1 post-dose efficacy evaluation) and per-protocol set (PPS; patients in the FAS without major protocol deviations that affected efficacy); secondary efficacy endpoints were assessed in the FAS only. The primary efficacy endpoint was the average number of acute gout flares per patient over 12 weeks. Secondary efficacy endpoints included the proportion of patients experiencing ≥1 acute gout flare over 12 weeks, the time from randomization to the first flare, and the mean duration of gout flares per patient for each treatment group. Safety assessments included the incidence of adverse events (AEs), laboratory tests, vital signs, and physical examination. Immunogenicity was assessed by the incidence of anti-drug antibodies (ADAs) and neutralizing antibodies (NAbs).
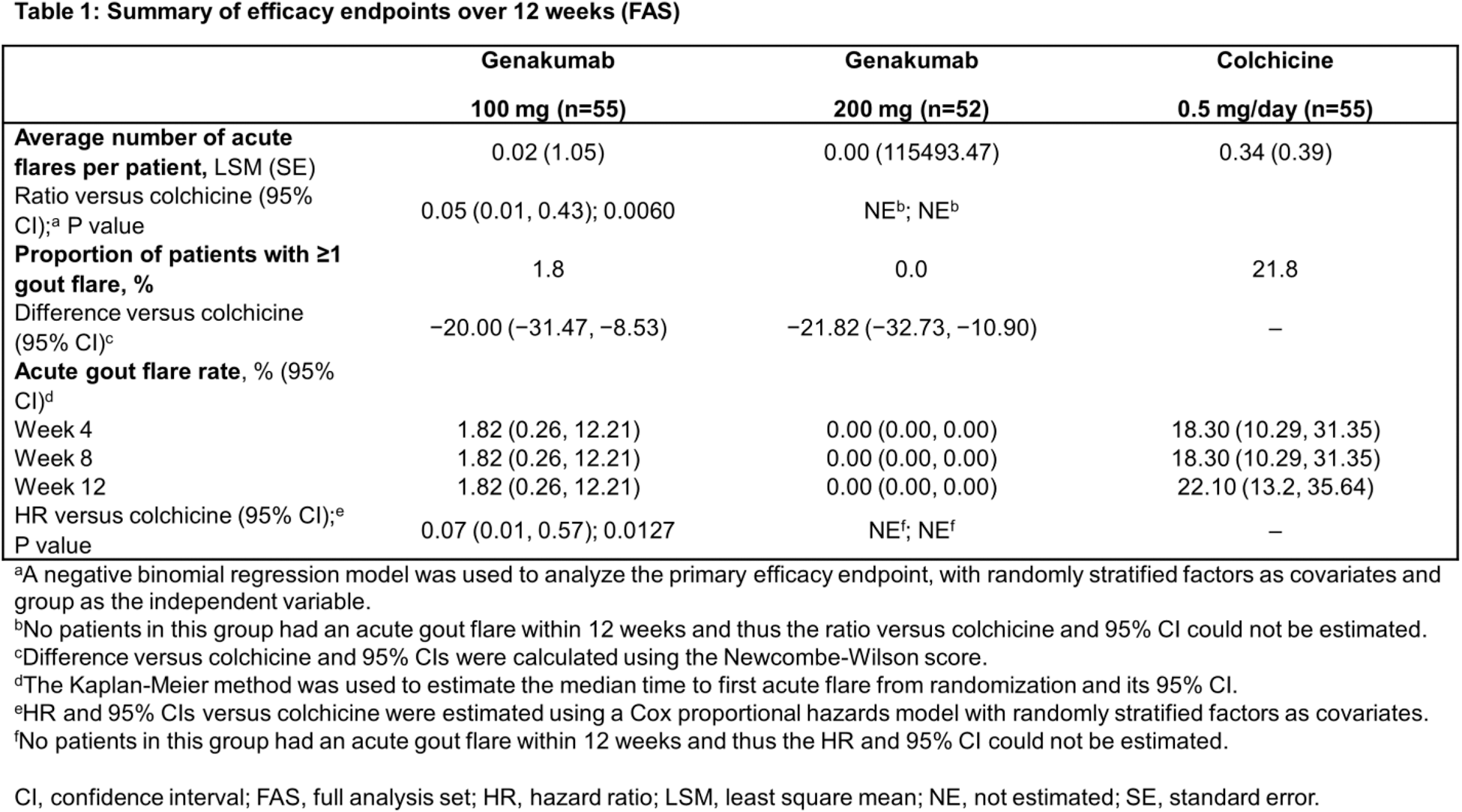
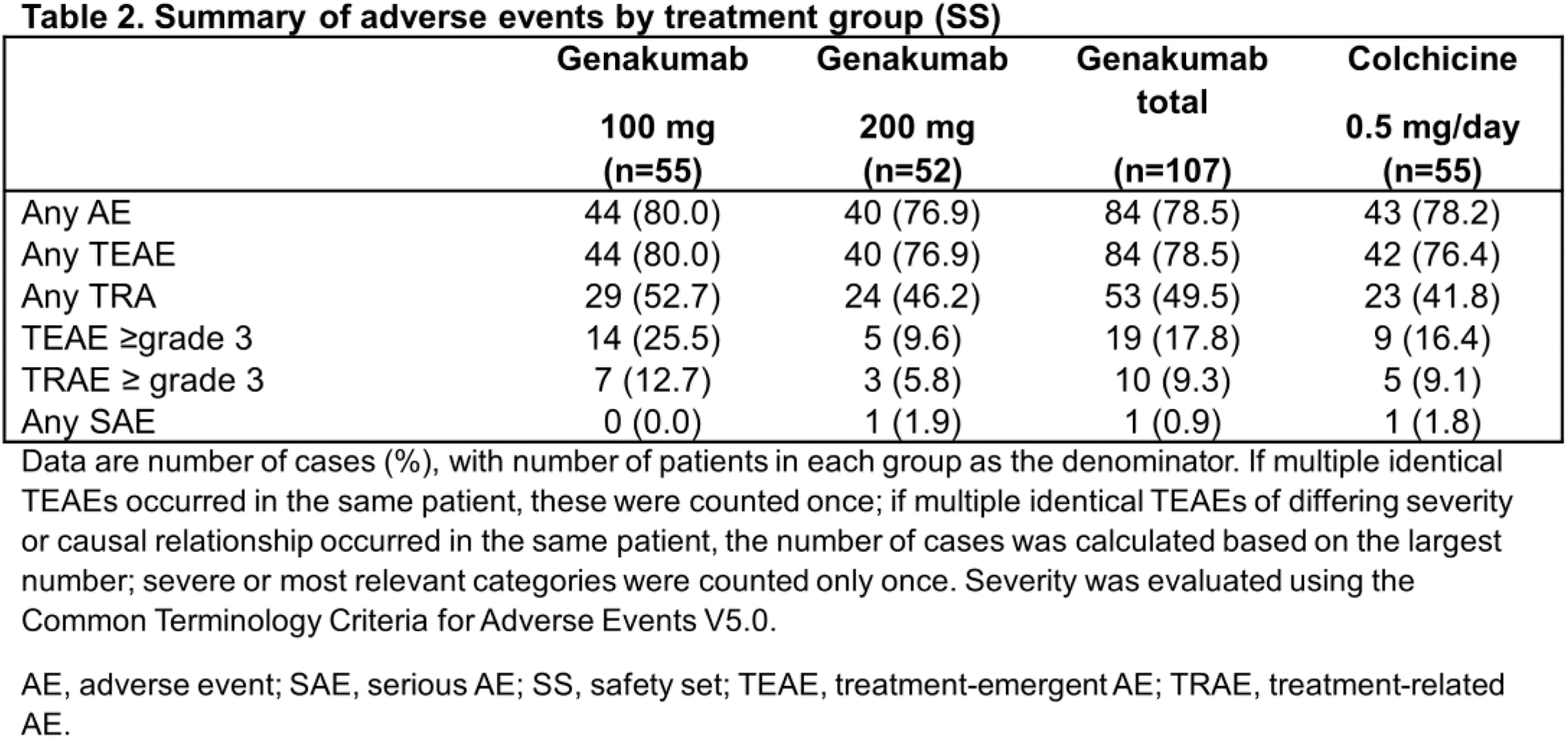
Results: Between July 20, 2023, and January 19, 2024, a total of 165 patients were enrolled in the study, of whom 162 received genakumab 100 mg (n=55), 200 mg (n=52), or colchicine (n=55). All patients included in the study were male, with a mean (standard deviation [SD]) age of 40.8 (11.7) years, body weight of 81.0 (13.7) kg, and BMI of 27.2 (3.8) kg/m 2 . The mean duration of gout history was 66.7 (49.2) months. Overall, baseline demographics and disease characteristics were generally similar between treatment groups. During the 12-week treatment period, only one patient (1.8%) in the genakumab 100 mg group experienced a single acute gout flare lasting 2.0 days and no patients in the genakumab 200 mg group experienced an acute gout flare. In contrast, 12 patients (21.8%) in the colchicine group had ≥1 gout flare, with a mean duration of 11.6 days, of whom 14.5% (n=8), 5.5% (n=3), and 1.8% (n=1) experienced one, two, and five flares, respectively. Based on a negative binomial regression model, a single subcutaneous injection of genakumab 100 mg significantly reduced the average number of acute gout flares per patient occurring over 12 weeks compared with daily colchicine (Table 1). Since no acute gout flares occurred in the genakumab 200 mg group, the ratio of number of flares versus colchicine could not be estimated. In addition, the proportion of patients with ≥1 acute gout flare and the rate of acute gout flare over 12 weeks was numerically reduced in patients receiving genakumab (100 or 200 mg) compared with colchicine (Table 1). Treatment-emergent AEs (TEAEs) were reported in 80.0% (n=44), 76.9% (n=40), and 76.4% (n=42) of patients receiving genakumab 100 mg, 200 mg, and colchicine, respectively (Table 2), with hypertriglyceridemia and hypercholesterolemia the most commonly reported in both genakumab and colchicine groups. No TEAEs leading to study discontinuation, treatment-related serious adverse events (SAEs), or deaths were reported. A low incidence of positivity for ADAs was observed across treatment groups, with no safety events associated with positivity for ADAs and no presence of NAbs.
Conclusion: Genakumab demonstrated efficacy as a prophylaxis against acute gout flares in adults with gout initiating ULT and was generally well-tolerated. A single dose of genakumab 100 mg or 200 mg reduced the average number of acute gout flares per patient and shortened flare duration over 12 weeks, with a safety profile similar to daily colchicine. Notably, in the genakumab 200 mg group, gout flares were not observed for the entire 12-week period, a finding that will be further investigated in larger sample populations.
REFERENCES: NIL.
Acknowledgements: The authors wish to acknowledge the patients and investigators who participated in this study. This study was sponsored by Changchun GeneScience Pharmaceutical Co., Ltd. Medical writing support was provided by Louise Hulme, MSc (Rude Health Consulting Ltd), and was funded by Changchun GeneScience Pharmaceutical Co., Ltd.
Disclosure of Interests: Hejian Zou: None declared, Yu Xue: None declared, Jiankang Hu: None declared, Ning Zhang: None declared, Rongbin Li: None declared, Hongtao Guo: None declared, Long Qian: None declared, Wei Gou: None declared, Jing Yang: None declared, Li Mao: None declared, JING YU: None declared, Shengyun Liu: None declared, Li Qin: None declared, Hua Wei: None declared, Linjie Chen: None declared, Xuebin Wang: None declared, Li Shujie: None declared, Zhengfu Du: None declared, Zhanqing Gu: None declared, Lei Yang: None declared, Rongping Li: None declared, Lihua Duan: None declared, Fenghong Yuan: None declared, Anbin Huang: None declared, Lin Chen: None declared, Xiaofei Shi: None declared, Jie Wu: None declared, Xinwang Duan: None declared, Zhenchun Zhang: None declared, Zili Fu: None declared, Jiangtao Li: None declared, Xiaoping Hong: None declared, Yanlin Li: None declared, Ling Lei: None declared, Zhichun Liu: None declared, Jianzhao Cheng: None declared, Changgui Li: None declared, Qingchu Li: None declared, Jian Xu: None declared, Jiashun Zeng: None declared, Hua Shang: None declared, Juan Li: None declared, Jingyang Li: None declared, Qian Xu Employee of Changchun GeneScience Pharmaceutical Co., Ltd., Tianhong Luo Employee of Changchun GeneScience Pharmaceutical Co., Ltd.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (