

Background: The treatment goals for idiopathic inflammatory myopathy (IIM) are to reduce inflammation and disease activity and decrease long-term morbidity and mortality. Therapies providing durable clinical responses without requiring chronic immunomodulatory drugs are lacking. CD19-targeting chimeric antigen receptor (CAR) T cells have achieved durable drug-free responses in IIM patients in an academic program. Rese-cel (formerly referred to as CABA-201), is a fully human, autologous 4-1BB anti-CD19-CAR T cell therapy, designed to deeply and transiently deplete CD19 positive cells following a weight-based, one-time infusion. This approach may enable an “immune system reset” with the potential for durable response without chronic immunosuppression. RESET-Myositis TM (NCT06154252) is an ongoing Phase 1/2 trial evaluating rese-cel in 3 independent cohorts of adults with IIM (immune mediated necrotizing myopathy [IMNM], dermatomyositis [DM], and antisynthetase syndrome [ASyS]), as well as a fourth cohort of patients with juvenile IIM (JIIM).
Objectives: The primary objective is safety and tolerability within 28 days of infusion. Secondary objectives include clinical outcomes (ie., total improvement score [TIS], Cutaneous Dermatomyositis Disease Area and Severity Index Activity Score [CDASI-A], and use of immunomodulatory agents) and translational assessments (ie., CAR T cell pharmacokinetics and impact on peripheral B-cell populations).
Methods: Eligible patients in the adult cohorts are ≥18 to ≤75 years with IIM, positive myositis specific autoantibody (MSA), manual muscle test (MMT-8) ≤142 and at least moderate disease activity on 2 or more core set measures. A single weight-based infusion of 1x10 6 CAR T cells/kg is administered following a preconditioning regimen (fludarabine 25 mg/m 2 /d on Days -5, -4 and -3, and cyclophosphamide 1,000 mg/m 2 on Day -3). All non-glucocorticoid immunomodulatory agents are discontinued by Day –5.
Results: 3 patients have been treated with rese-cel and have completed at least 1 month of follow-up in the RESET-Myositis TM trial, IMNM-1, IMNM-2 and DM-1 as of 16 December 2024. A fourth patient (IMNM-3) was recently dosed with rese-cel and others are anticipated in early 2025 (Table 1).
Table 1.
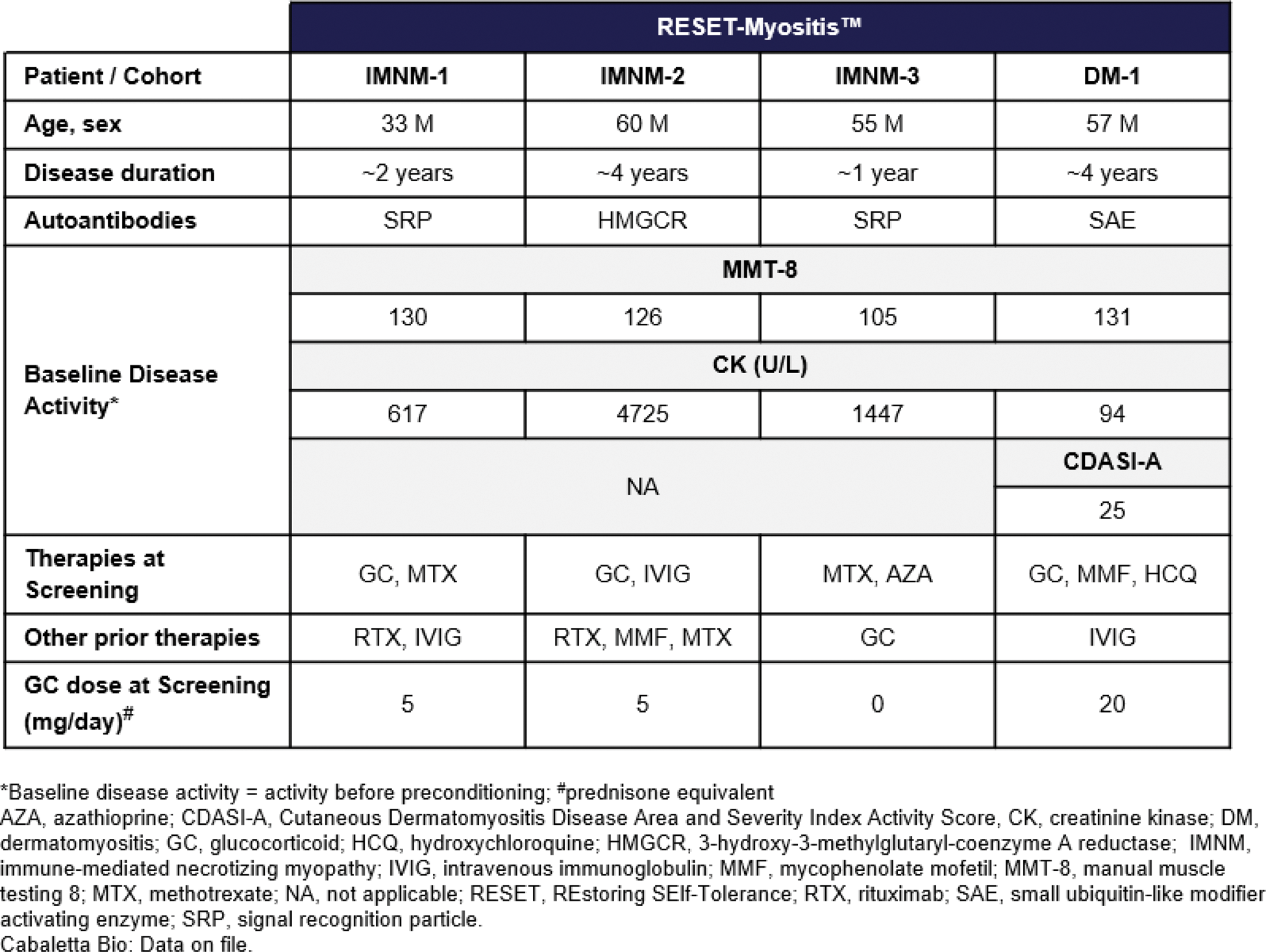
Rese-cel has been well-tolerated with no dose-limiting toxicity, cytokine release syndrome (CRS) or immune effector cell-associated neurotoxicity syndrome (ICANS) in the first 3 IIM patients with at least 1 month of follow-up. IMNM-2 who had multiple genetic and clinical risk factors for thromboembolism experienced a pulmonary embolism (PE) on Day 38, which was assessed as unrelated to rese-cel. DM-1 demonstrated a rapid and robust response following rese-cel infusion, achieving major TIS response at Day 29, maintained up to Week 12 while remaining off all therapies and tapering steroids. CDASI-A improved from 25 to 8 by Week 12. IMNM-1, who discontinued steroids shortly after rese-cel, achieved clinically meaningful TIS by Week 12 and moderate TIS by Week 24, and remains off all therapies through Week 40. IMNM-2 showed downward trending CK levels at Day 29 then developed a PE on Day 38 with a prolonged hospitalization and recovery, potentially confounding subsequent evaluations of treatment response. Following infusion of rese-cel, the first 3 patients exhibited CAR T cell expansion that peaked between day 8 and day 15 (C max ). CAR T cells were undetectable peripherally by day 29 for IMNM-1 and IMNM-2 and remained detectable for up to 12 weeks for DM-1. Serum IFN-γ levels increased prior to rese-cel C max in IMNM-1 and DM-1 and CAR T cells exhibited a predominantly CD8+ phenotype at C max . In IMNM-1, IMNM-2 and DM-1, B cells were rapidly reduced to undetectable levels by 15 days post rese-cel infusion and remained undetectable through the first month. Repopulation with transitional naïve B cells occurred at 8 weeks in IMNM-1 and DM-1. B cells remained undetectable in IMNM-2 eight weeks following infusion.
Conclusion: Data from IIM patients dosed with rese-cel demonstrate early immunomodulatory-free clinical responses and a favorable safety profile in the setting of CAR T cell expansion and peripheral B cell depletion. These initial data suggest the potential for rese-cel to reset the immune system in IIM, allowing patients to achieve meaningful clinical responses while off all immunomodulatory therapies and tapering glucocorticoids.
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: Tahseen Mozaffar Sanofi, Amicus; AnnJi; Argenx; Astellas Gene Therapy; AstraZeneca; Janssen; Sanofi; Spark Therapeutics; UCB, Arvinas; AskBio; Horizon Therapeutics; Maze Therapeutics; Sarepta, Fate Therapeutics, Amicus; AnnJi; Argenx; Astellas Gene Therapy; AstraZeneca; Janssen; Sanofi; Spark Therapeutics; UCB, Bristol-Myers Squibb (BMS); Cartesian Therapeutics; Grifols; ML-Bio; Valerion, Elie Naddaf Johnson and Johnson, Nizar Chahin: None declared, Erin Wilfong Astra Zeneca, Boehringer Ingelheim, Boehringer Ingelheim, Courtney Little Cabaletta Bio, Carl DiCasoli Cabaletta Bio, Claire Miller Cabaletta Bio, Jenell Volkov Cabaletta Bio, Daniel Nunez Cabaletta Bio, Thomas Furmanak Cabaletta Bio, Jason Stadanlick Cabaletta Bio, Larissa Ishikawa Cabaletta Bio, Zachary Vorndran Cabaletta Bio, Alexandra Ellis Cabaletta Bio, Jazmean Williams Cabaletta Bio, Steve Flanagan Cabaletta Bio, Quynh Lam Cabaletta Bio, Fatemeh Hadi Nezhad Cabaletta Bio, Raj Tummala Cabaletta Bio, Samik Basu Cabaletta Bio, David Chang Cabaletta Bio.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (