

Background: A 6-month intervention with abatacept in the ARIAA trial (EUDRA-CT 2014–000555–93) significantly retarded the development of RA compared to placebo in ACPA-positive individuals with joint pain and subclinical inflammation on hand MRI. This inhibitory effect peaked at the end of the 6-month treatment period and was still discernible after 1-year of follow-up without any intervention. The durability of the inhibitory effect on development of RA is not known.
Objectives: To assess the durability of the inhibitory effect of abatacept to develop RA observed in the ARIAA trial.
Methods: The ARIAA trial recruited individuals with positive ACPA antibodies, joint pain and findings of synovitis, tenosynovitis or bone marrow edema on the MRI of the dominant hand. Subjectswere randomized to receive a 6-month course of abatacept 125 mg sc weekly or placebo and followed without any treatment for additional 12 months. Individuals who did not develop RA during the study period of December 2014 and June 2021 were contacted up to November 2024 to ascertain their latest status for RA diagnosis based on the discretion of their primary physician. We used survival analysis methods and calculated restricted mean survival time difference, net average RA-free time gained due to treatment over time to evaluate the durability of treatment effect and compared the characteristics of participants who developed RA during study to those who developed RA later and who never developed RA.
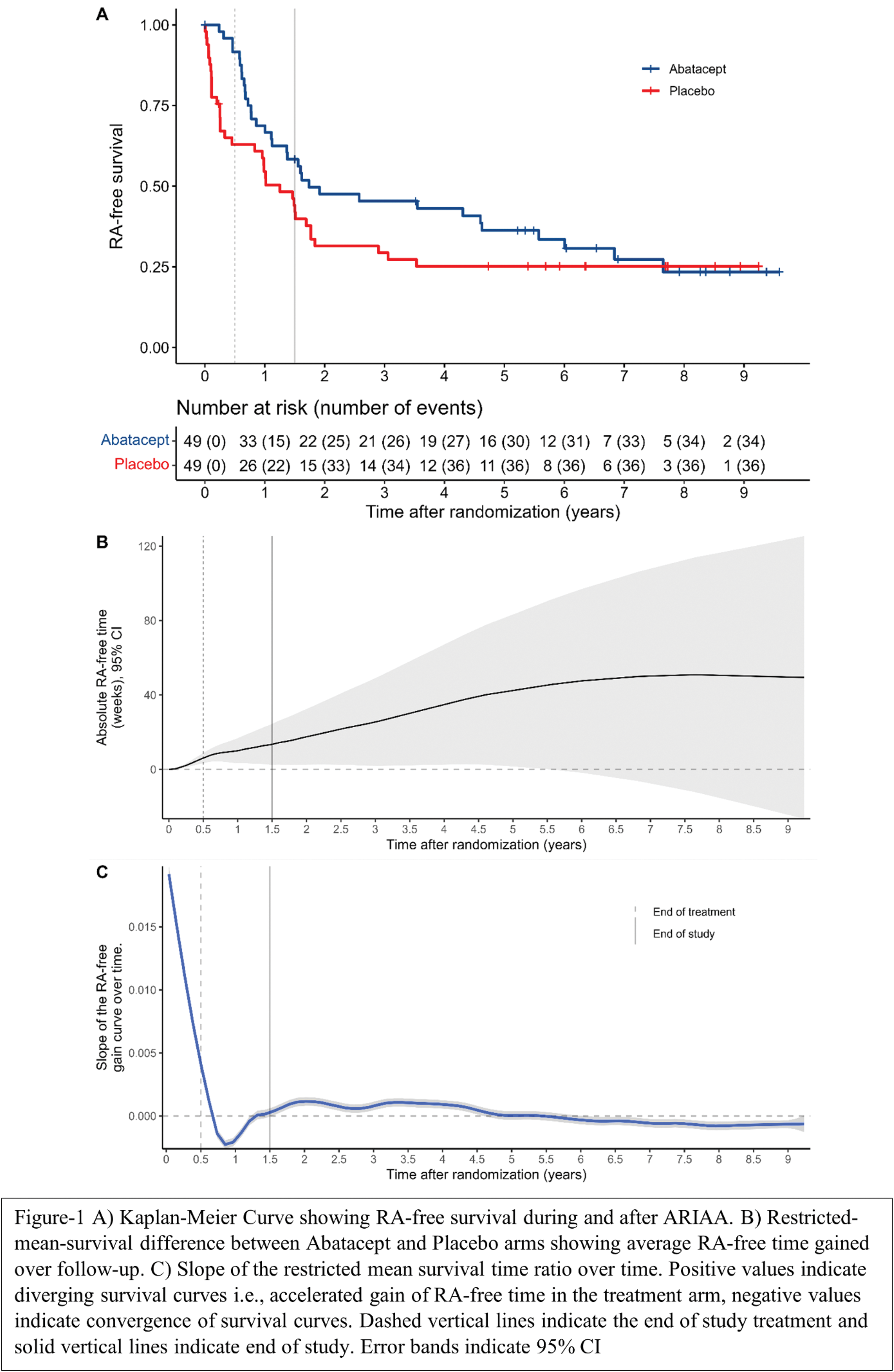
Results: 98 individuals with a positive ACPA were randomized to abatacept (49) or placebo (49). At the end of treatment (6 months) and observation (18 months) periods, RA developed in 4 (8%) and 17 (35%) participants in the abatacept group as opposed to 17 (35%) and 28 (57%) participants in the placebo group resulting in 15.3 weeks (95%CI, 4.3 to 26.3) of RA-free time gain on average in the abatacept group compared to placebo by the end of the study. After the closure of the study, 17 participants in the abatacept group and 8 participants in the placebo group developed RA during a median follow-up duration of 5.3 years after randomization (IQR, 1.9 to 7.2 years) which cumulated to 34/49 (69%) in the abatacept group and 36/49 (73%) in the placebo group. During this period the RA-free survival gain in the Abatacept group persisted for up to 5 years (Figure 1A) increasing to ~40 weeks (Figure 1B). The first derivative (Figure 1C) of the RMST difference curve indicates that the inhibitory effect (a positive slope for RA-free time gain) of abatacept during the treatment-period is offset initially during the first year after treatment cessation but then persisted for up-to five years after randomization and waned afterwards resulting in an ultimate catch-up of the placebo arm. A descriptive comparison of participants who never developed RA indicate that these had a lower ESR, lower RF levels, lower pain scores and better functional status at baseline (Table 1).
Conclusion: Abatacept showed a delayed inhibitory effect against the development of RA lasting for up-to 5 years after the initial 6 months intervention. Systemic inflammation, autoimmunity, pain, and function could help improve risk stratification while selecting individuals at risk of RA for a preventive intervention.
Characteristics of the study population by RA status at long-term follow-up.
| Characteristic | Overall
| No RA
| RA after study
| RA during study
| p-value2 |
|---|---|---|---|---|---|
| Age | 49 (42, 59) | 49 (46, 62) | 52 (41, 59) | 49 (42, 56) | 0.5 |
| Female gender | 70 (71%) | 22 (79%) | 15 (60%) | 33 (73%) | 0.3 |
| Abatacept use | 49 (50%) | 15 (54%) | 17 (68%) | 17 (38%) | 0.048 |
| RF | 33 (20, 112) | 20 (13, 23) | 55 (22, 111) | 62 (25, 281) | <0.001 |
| ACPA above median | 49 (50%) | 13 (46%) | 12 (48%) | 24 (53%) | 0.8 |
| Never smoker | 40 (41%) | 14 (52%) | 12 (48%) | 14 (31%) | 0.2 |
| MRI synovitis score | 6.0 (4.0, 9.0) | 5.0 (4.0, 9.0) | 9.0 (5.0, 11.0) | 6.0 (3.0, 8.0) | 0.084 |
| MRI tenosynovitis score | 4.0 (2.0, 7.0) | 4.0 (2.0, 5.0) | 4.0 (3.0, 8.0) | 4.5 (2.0, 7.5) | 0.4 |
| MRI osteitis score | 1.00 (0.00, 2.00) | 0.00 (0.00, 1.00) | 0.00 (0.00, 2.00) | 1.00 (0.00, 3.00) | 0.13 |
| ESR | 13 (8, 20) | 9 (5, 14) | 13 (8, 24) | 16 (9, 26) | 0.024 |
| Painful joints | 3.0 (0.0, 6.0) | 0.5 (0.0, 2.5) | 3.0 (0.0, 6.0) | 4.0 (2.0, 8.0) | 0.001 |
| DAS-28 ESR | 2.92 (2.28, 3.97) | 2.30 (1.49, 2.79) | 3.19 (2.29, 4.06) | 3.39 (2.88, 4.41) | <0.001 |
| Pain VAS, mm | 45 (13, 69) | 15 (3, 46) | 45 (22, 65) | 55 (21, 77) | 0.001 |
| Patient global VAS | 44 (10, 72) | 13 (3, 35) | 51 (21, 70) | 56 (20, 79) | <0.001 |
| Physician global VAS | 18 (4, 36) | 5 (1, 18) | 18 (10, 40) | 30 (12, 44) | <0.001 |
| RAID | 3.87 (1.87, 5.68) | 2.02 (0.58, 3.95) | 4.90 (2.48, 6.09) | 5.15 (2.61, 6.53) | <0.001 |
| SF-36 PCS | 41 (31, 47) | 46 (39, 50) | 41 (31, 50) | 35 (29, 43) | 0.005 |
RF rheumatoid factor, ACPA anti-citrullinated protein antibodies, MRI magnetic resonance imaging, ESR erythrocyte sedimentation rate, CRP c-reactive protein, VAS visual analog scale, RAID, rheumatoid arthritis impact of disease questionnaire, SF-36 short form 36 items, PCS physical component summary, MCS mental component summary
1 Median (Q1, Q3); n (%)
2 Kruskal-Wallis rank sum test; Pearson’s Chi-squared test
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: Koray Tascilar Novartis, Mikkel Østergaard: None declared, Verena Schönau: None declared, Uta Kiltz: None declared, Xenofon Baraliakos: None declared, Martin Fleck: None declared, David M Kofler: None declared, Martin Feuchtenberger: None declared, Ulrike Steffen (née Harre): None declared, Mario M. Zaiss: None declared, Michael Zaenker: None declared, Reinhard Voll: None declared, Cornelia Glaser: None declared, Eugen Feist: None declared, Gerd R. Burmester: None declared, Jan Zernicke: None declared, Michaela Koehm: None declared, Frank Behrens: None declared, Kirsten Karberg: None declared, Johannes Strunk: None declared, Juan de Dios Cañete Crespillo: None declared, Esperanza Naredo: None declared, Mária Filková: None declared, Ladislav Šenolt: None declared, Georg Schett: None declared, Jürgen Rech: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (