

Background: Inflammation and vasculopathy may play a role in the development of interstitial lung disease (ILD) in systemic sclerosis (SSc). Pre-clinical evidence showed possible preventive effects of major vasoactive/vasodilating drugs (VVD), mostly deriving from mouse models of ILD. Similarly, preliminary data from registries indicated a preventive role of some immunosuppressants (IMS).
Objectives: As SSc patients regularly receive such medications to treat other disease manifestations, we aimed to evaluate the association between IMS/VVD use and the development of SSc-ILD.
Methods: Patients from the EUSTAR database were eligible if classified according to the 2013 ACR/EULAR criteria for SSc, with baseline absence of ILD (defined by HRCT negative for lung abnormalities and FVC >70%), at least 1 follow-up CT available at 12±3 months, and information on IMS/VVD exposure. Patients with pulmonary hypertension on right heart catheterization at any timepoint during the observation were excluded. Exposure included the use of any IMS (mycophenolate mofetil, cyclophosphamide, azathioprine, methotrexate, prednisone over 10 mg/day – categorized as conventional IMS ; rituximab or tocilizumab – as biologic IMS ) or any major VVD (bosentan, other endothelin receptor antagonists – grouped as ERAs ; sildenafil, other phosphodiesterase 5 inhibitor – as PDE5i ; iloprost, other prostanoids –as prostanoids ) for at least 3 months during the 1-year interval. New ILD onset on CT scan at 1-year follow-up represented the outcome. Associations with IMS/major VDD were calculated using generalized estimated equation models with backward selection, balancing for multiple 1-year intervals observations per patient. Confounders included risk factors for ILD onset and possible reasons for IMS/major VVD prescription, as well as interaction terms between IMS/major VVD and a possible reason for prescription (i.e., conventional or biologic IMS for diffuse skin involvement or arthritis; ERAs, PDE5i or prostanoids for digital ulcers - DU). Additionally, we performed a sensitivity analysis dividing the observation time in before and after the previous EULAR SSc treatment recommendations were first presented, with index date on 30 June 2015, to test if and how change in practice might influence the results.
Results: In total, 4091 SSc patients were included: 86% female sex, 27% DU, 28% diffuse skin subset (dcSSc), with mean age 55±14, FVC% 100±16, DLco% 74±17. Among 13113 yearly visits over a median 4 (95%CI 2-7) years follow up, ILD onset was detected in 444 instances (11% of patients, 3% visits). Analyzing the preventive effectiveness according to drug classes (conventional/biologic IMS, ERAs, PDE5i, prostanoids), most clinical risk factors were confirmed (Figure 1a), in particular DU (history or presence) significantly associated with ILD development (adjusted OR 1.48, 95%CI 1.17-1.87). PDE5i and prostanoids were significantly associated with ILD onset. While this independent result was seen for the class of prostanoids (OR 0.73, 95%CI 0.54-0.98), a statistically significant interaction emerged for PDE5i only when present in combination with DU. Combining interaction and fixed effects, we observed that exposure to PDE5i in patients with DU was associated to a reduced risk of ILD onset (OR 0.34; 95%CI 0.17-0.49). Although there was a significant association between the interaction of conventional IMS and dcSSc with the risk of ILD onset, this finally translated into a trend towards protection from ILD onset in diffuse SSc patients exposed to conventional IMS (OR 0.80, 95%CI 0.57-1.12). When testing single medications (Figure 2a), we still observed a higher risk of ILD onset for DU and an association of iloprost and sildenafil with ILD onset. Again, this association turned significant for exposure to sildenafil exclusively in combination with DU, determining a protective marginal effect estimate (OR 0.27, 95% CI 0.13-0.57). In line with the results of the rostanoids class, exposure to iloprost was associated with a reduced risk of ILD onset (OR 0.54, 95% CI 0.37-0.78) and this was regardless of DU. None of the single IMS molecules was significantly associated with ILD onset. Observing the data before and after the previous EULAR recommendations for SSc treatment were first presented, new ILD onset was observed in 208/7871 visits of 2519 patients before and 236/5242 visits of 2235 patients after the index date. Comparing the two periods, we noted an increase in the prevalence of “DU ever” from 18.5% to 42.1%, possibly representing better identification following awareness campaigns, as well as a certain survivor bias (disease duration from median 7 to 9 years). In fact, the increase in the use of major VVD was marginal (PDE5i from 5.1% to 8.7%, prostanoids from 18.1% to 23.1%, sildenafil from 4.6% to 6.7%, iloprost from 14.0% to 15.8%). When the models from the overall population were applied to the visits before 2015, we confirmed all protective associations of prostanoids, iloprost, and the interaction between sildenafil and DU (Figures 1b and 2b); after 2015, we confirmed the protective interaction of PDE5i/sildenafil and DU for ILD onset, but not for prostanoids/iloprost (Figures 1c and 2c).
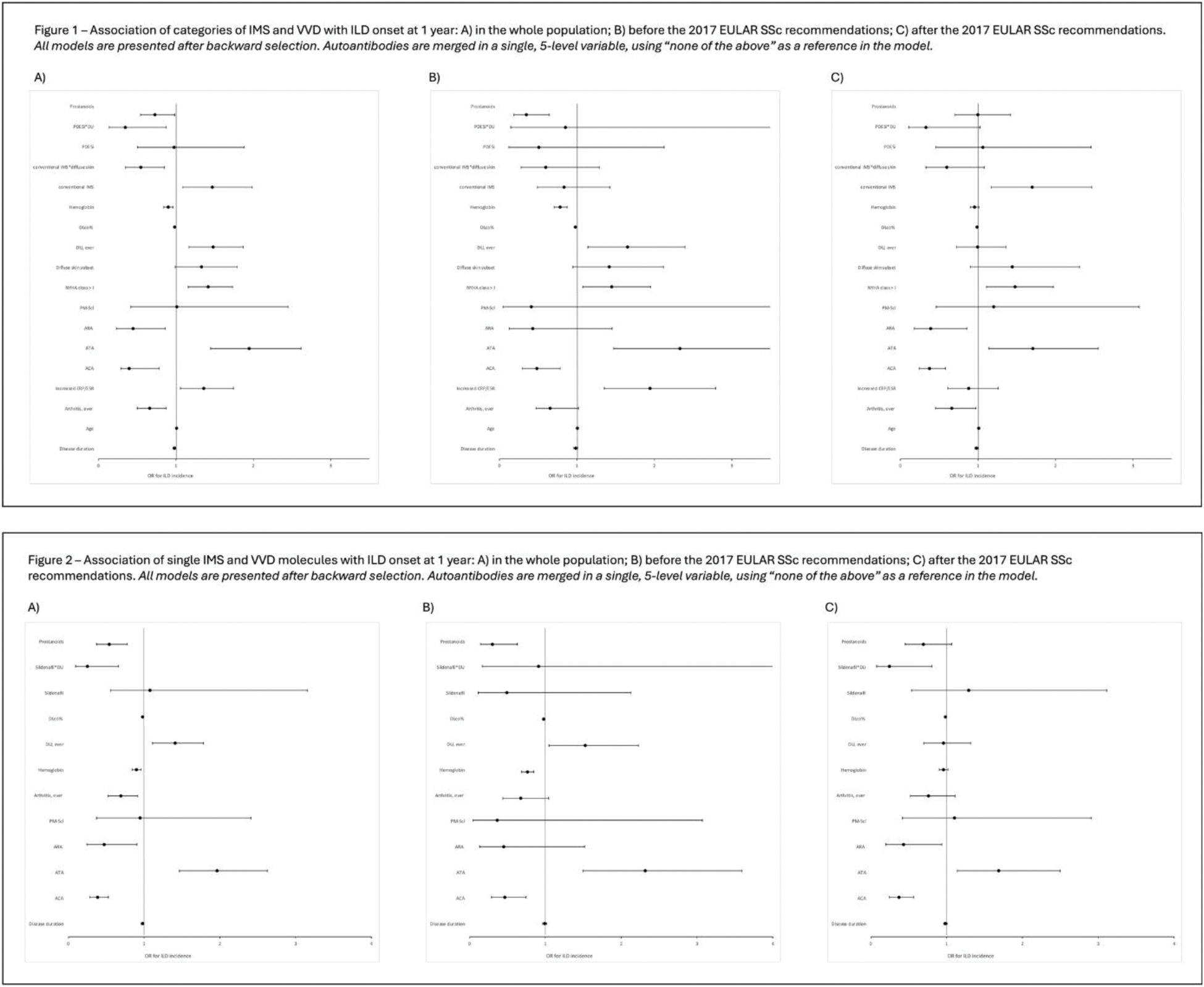
Conclusion: We showed that treatment with major VVDs, specifically sildenafil and iloprost, but not with IMS, might have a protective effect on the development of SSc-ILD within one year. This beneficial effect might vary according to the prevalence of DU, therefore hypothesizing different interventions according to the clinical scenarios. The preventive effect of these medications needs confirmation in randomized controlled trials.
REFERENCES: NIL.
Acknowledgements: On behalf of the EUSTAR collaborators.
Disclosure of Interests: Liubov Petelytska research grant from Swiss National Research Foundation/Scholars at risk, Lorenzo Tofani: None declared, Elise Siegert: None declared, Elisabetta Zanatta: None declared, Gianluca Moroncini: None declared, Christina Bergmann Boehringer-Ingelheim and Janssen, German Research Association, IZKF Erlangen (Clinician Scientist Program), BMBF (iIMMUNE_ACS program), Vanessa Smith: None declared, Gabriela Riemekasten: None declared, Kamal Solanki Boehringer Ingelheim; European Scleroderma Trials and Research (EUSTAR); Apollo Hospitals Education and Research Foundations (AHERF), Fabio Cacciapaglia Abbvie, Janssen, Boehringer-Ingerlheim, Pfizer, Galapagos, Eli Lilly, Abbvie, Boehringer Ingelheim, Janssen, Britta Maurer Boehringer-Ingelheim, GSK, Novartis, Otsuka, Novartis, Boehringer Ingelheim, Janssen-Cilag, AbbVie, Protagen, Novartis Biomedical Research, José Antonio P. da Silva: None declared, Marija Geroldinger-Simic Abbvie, Amgen, AstraZeneca, Boehringer Ingelheim, Janssen, Novartis, Abbvie, Amgen, AstraZeneca, Boehringer Ingelheim, Janssen, Novartis, Vera Bernardino: None declared, Madelon Vonk Boehringer Ingelheim, Bristol-Myers Squibb, GSK, Janssen Pharmaceutical Companies of Johnson & Johnson, MSD, Novartis, and Roche, Boehringer Ingelheim and Janssen Pharmaceutical Companies of Johnson & Johnson, Boehringer Ingelheim, Ferrer, Galapagos and Janssen Pharmaceutical Companies of Johnson & Johnson, Jeska K. de Vries-Bouwstra ABBvie, Janssen, Boehringer-Ingerlheim, ABBvie, Janssen, Boehringer-Ingerlheim, Janssen-Cilag, Galapagos, Roche, Anna-Maria Hoffmann-Vold Boehringer Ingelheim, Janssen, Medscape, Merck Sharp & Dohme, Novartis and Roche, AbbVie, ARXX, Boehringer Ingelheim, Bristol Myers Squibb, Genentech, Janssen, Medscape, Merck Sharp & Dohme, Pliant Therapeutics, Roche and Werfen, Boehringer Ingelheim, Janssen, Oliver Distler 4P-Pharma, Abbvie, Acceleron, Acepodia Biotech, Aera, Alcimed, Altavant, Amgen, AnaMar, Anaveon AG, Argenx, AstraZeneca, Blade, Bayer, Boehringer Ingelheim, Calluna (Arxx), Cantargia AB, Catalyze Capital, Corbus, CSL Behring, Galderma, Galapagos, Glenmark, Gossamer, Horizon, Janssen, Kymera, Lupin, Medscape, MSD Merck, Miltenyi Biotec, Mitsubishi Tanabe, Nkarta Inc., Novartis, Orion, Pilan, Prometheus, Quell, Redxpharma, Roivant, EMD Serono, Topadur and UCB, Patent issued “mir-29 for the treatment of systemic sclerosis” (US8247389, EP2331143). Co-founder of CITUS AG, 4P-Pharma, Abbvie, Acceleron, Acepodia Biotech, Aera, Alcimed, Altavant, Amgen, AnaMar, Anaveon AG, Argenx, AstraZeneca, Blade, Bayer, Boehringer Ingelheim, Calluna (Arxx), Cantargia AB, Catalyze Capital, Corbus, CSL Behring, Galderma, Galapagos, Glenmark, Gossamer, Horizon, Janssen, Kymera, Lupin, Medscape, MSD Merck, Miltenyi Biotec, Mitsubishi Tanabe, Nkarta Inc., Novartis, Orion, Pilan, Prometheus, Quell, Redxpharma, Roivant, EMD Serono, Topadur and UCB, BI, Kymera, Mitsubishi Tanabe, UCB, Cosimo Bruni Boehringer Ingelheim, Research grant from the Iten-Kohaut foundation, the EMDO foundation and the Novartis foundation for medical-biological research.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (