

Background: Digital ulcers (DUs) affect approximately 50% of SSc patients, causing significant pain and disability. Current management involves both systemic and local therapies. However, the burden in terms of pain and quality of life due to refractory DUs remains heavy. Indeed, over 67% of SSc patients experience >5 DUs in their disease course, reflecting the high burden of this disease domain. While selexipag, an oral selective IP prostacyclin receptor agonist, is approved for the treatment of SSc-associated pulmonary arterial hypertension (PAH), its potential in healing DUs is largely unexplored.
Objectives: We aimed at evaluating the long-term efficacy of selexipag compared to iloprost in treating SSc-DUs.
Methods: This retrospective multicentre study included 96 SSc patients with refractory DUs, 32 treated with selexipag (median dose of 1600 mg/day- IQR 1100 mg) and 64 with intravenous iloprost (0.5–2 ng/kg/min), matched for gender, disease subset, and age at diagnosis. Both groups were concomitantly treated with conventional vascular therapies (i.e. calcium channel blockers, endothelin receptor antagonists and phosphodiesterase 5- inhibitors). DUs number, ischemic pain and Raynaud phenomenon (RP) severity were assessed at baseline, 6, 12, and 24 months. Pain and RP were evaluated using Likert Pain Scale (LPS) and Raynaud Condition Score (RCS), respectively. Additionally, DUs recurrence and new onset were recorded. Healing rates were estimated using Kaplan-Meier analysis. Binary logistic regression was performed separately for both treatment groups to identify predictors of healing outcomes, including the following variables: age at diagnosis, age, sex, SSc subtype, latency between RP onset and the first digital ulcer, age at therapy, latency between ulcer onset and therapy administration, DUs number at baseline, LPS at baseline, and RCS at baseline.
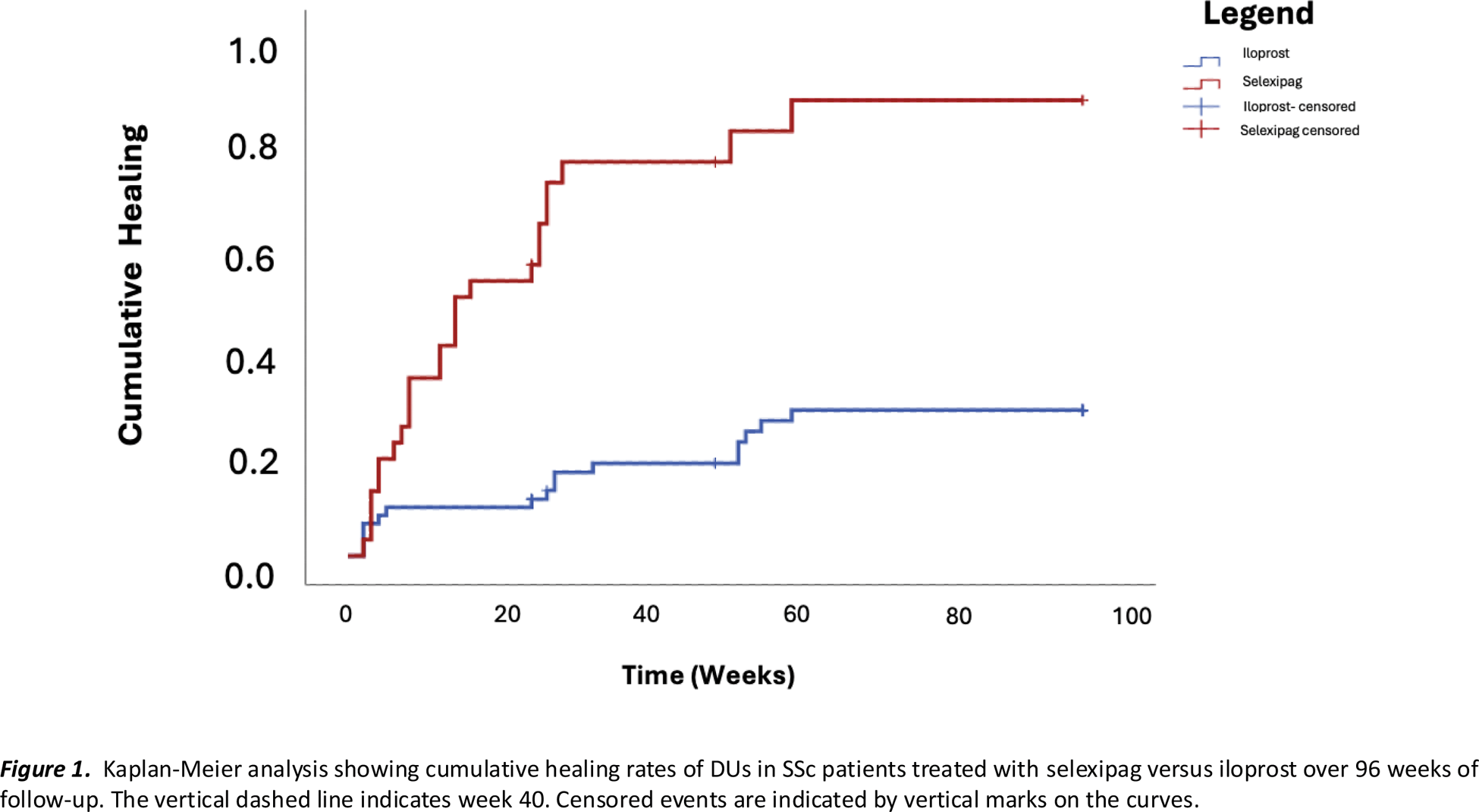
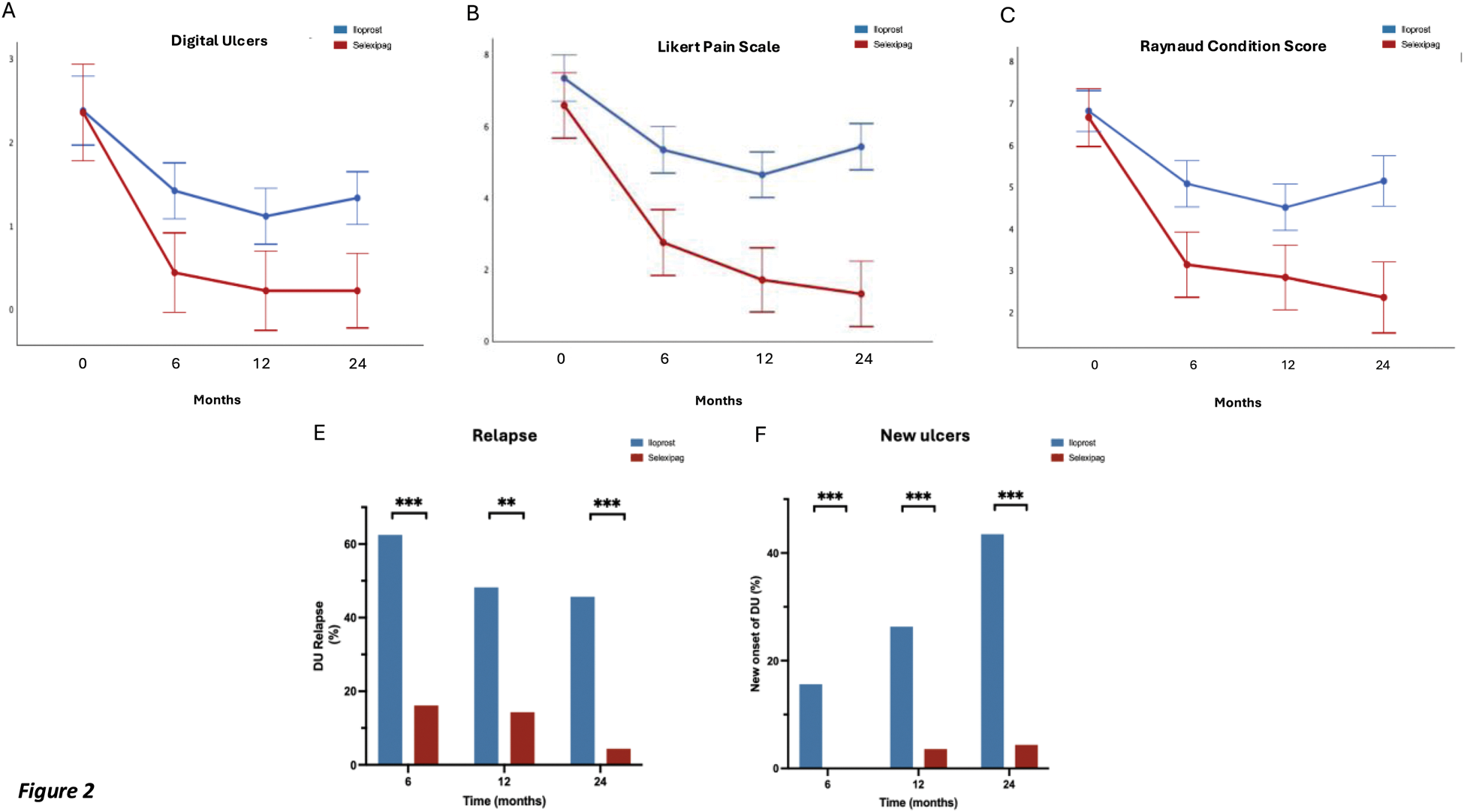
Results: DUs healing rate was significantly higher in selexipag-treated patients in comparison with iloprost-treated ones (87% vs 28%) at 96 weeks, with the formers achieving faster healing (75% vs 18% by week 40) (p<0.001) (Figure 1). DUs number, RCS and LPS scores showed significant improvement in selexipag-treated group compared to iloprost (p<0.001 for all) throughout the 24-month follow-up (Figure 2 A-B-C). Repeated measures analysis demonstrated significant changes over time for all three outcomes. The difference in treatment effect was supported by Mann-Whitney U analyses, showing comparable baseline measures (DUs: p=0.901; RCS: p=0.561; LPS: p=0.708) significant differences emerged by 6 months (DUs: p=0.001; RCS: p<0.001; LPS: p<0.001) and were maintained through 12 and 24 months (all p<0.001). Additionally, selexipag-treated patients experienced significantly lower relapse rates (5% vs 45% at 24 months, p<0.001) (Figure 1D). Consistently, DUs formation remained lower with selexipag (5% vs 40% at 24 months, p<0.001) (Figure 1F).Binary logistic regression revealed that in the iloprost group, baseline ulcer count showed a negative association with healing probability (OR=0.364, 95% CI: 0.147-0.904, p=0.029), while baseline pain score demonstrated a positive association (OR=2.376, 95% CI: 1.198-4.710, p=0.013). In contrast, the selexipag group analysis showed no statistically significant predictors, with baseline ulcer count (OR=0.562, 95% CI: 0.244-1.298, p=0.177) and pain score (OR=1.302, 95% CI: 0.715-2.370, p=0.389) remaining non-significant. Only two patients had to discontinue selexipag for mild intolerance after 12 months of treatment.
Conclusion: Our study shows that SSc patients treated with selexipag have a better sustained outcome of DUs compared to those treated with intravenous iloprost over 24 months. Furthermore, oral administration and good tolerability profile make selexipag a promising alternative option to standard therapy for SSc-related DUs, overcoming challenges such as difficult venous access, high hospitalization costs, and lost work productivity associated with IV administration. Prospective studies are needed to confirm a wider use of selexipag other than PAH.
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (