

Background: Zasocitinib (TAK-279) is an oral, allosteric, highly selective and potent, tyrosine kinase 2 (TYK2) inhibitor in late-stage clinical development for the treatment of immune-mediated inflammatory diseases, including psoriasis. Zasocitinib was developed using an AI-assisted compound design strategy to create a TYK2 inhibitor with next-generation selectivity and > 1 million-fold greater affinity for TYK2 than for Janus kinase (JAK) 1/2/3.
Objectives: To compare the potency and selectivity of zasocitinib for TYK2 versus JAK1/3 with licensed TYK2 and JAK inhibitors.
Methods: The inhibitory constants (K i s) of zasocitinib and deucravacitinib for JAK1 and TYK2 Janus homology 2 (JH2) domains were determined using a homogeneous time-resolved fluorescence assay. Fresh whole-blood cultures from 12–13 healthy volunteers characterised concentration–response (percent inhibition) curves for TYK2- (IL-12/18 IFN-γ) and JAK1/3- (IL-2 pSTAT5) dependent pathways for zasocitinib and licensed TYK2 and JAK inhibitors. Statistical modelling determined the relationship between concentration and percent inhibition, and half-maximal inhibitory concentration (IC 50 ) values were estimated. Drug plasma concentrations at clinically relevant or approved doses were simulated using published population pharmacokinetic models. Time over plasma concentrations > IC 50 and percent daily inhibition were compared between drugs for each pathway.
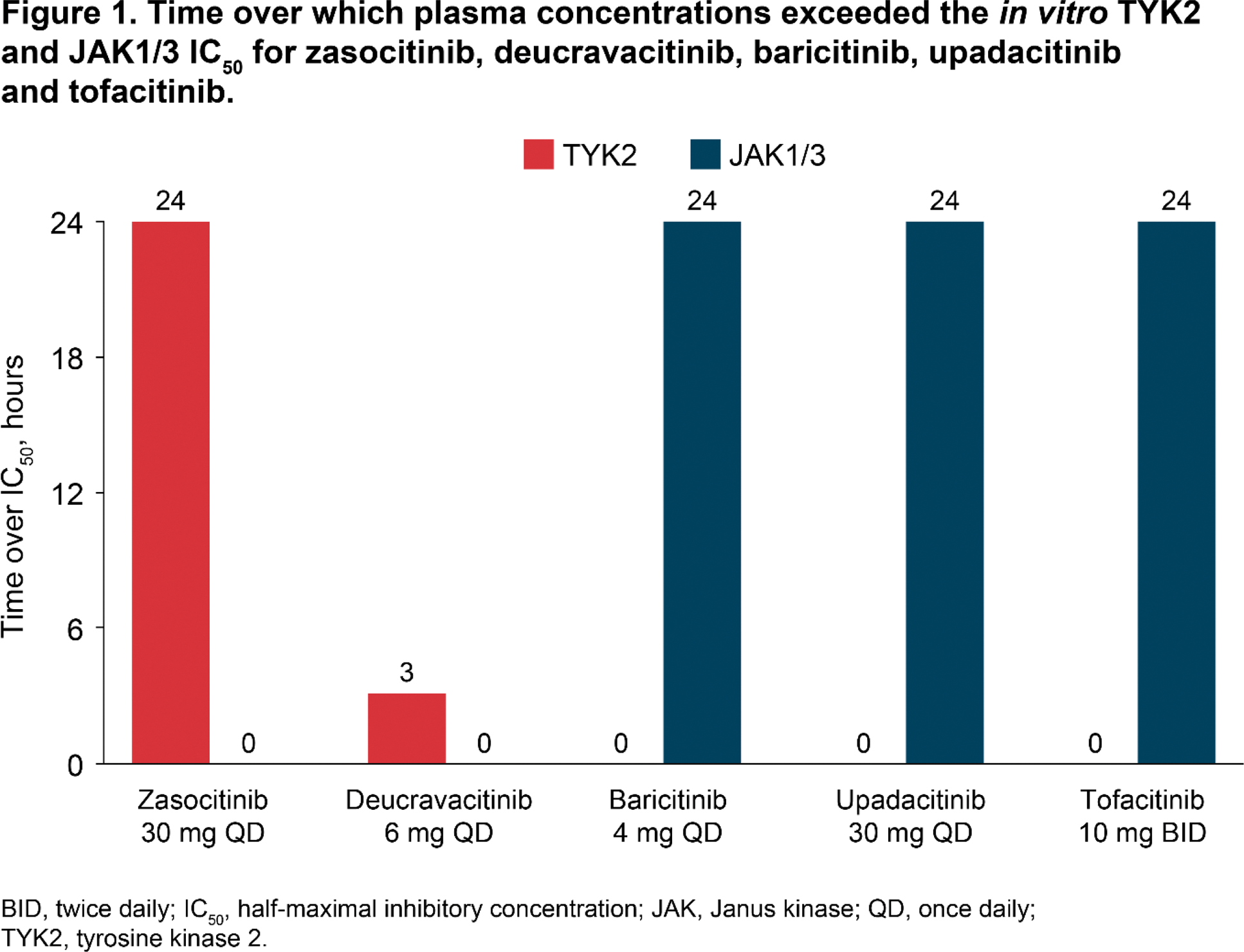
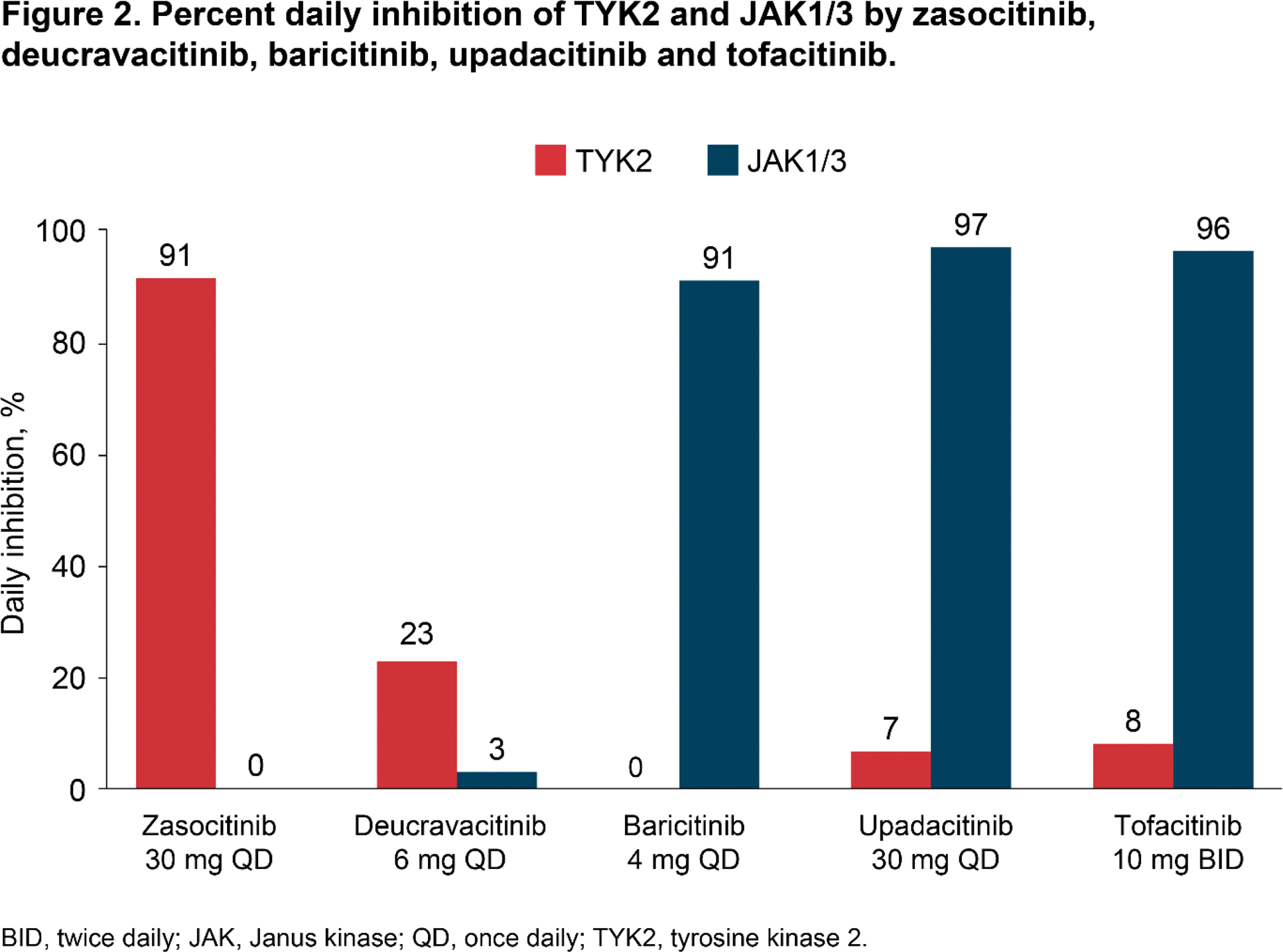
Results: The K i s of zasocitinib and deucravacitinib for the JAK1 JH2 domain were > 15 000 nM and 1 nM, respectively. The K i s of zasocitinib and deucravacitinib for the TYK2 JH2 domain were 0.0087 nM and 0.0115 nM, respectively. Simulated plasma concentrations were above TYK2 IC 50 for 24 hours for zasocitinib 30 mg once daily (QD) versus 3 hours for deucravacitinib 6 mg QD, and 0 hours for baricitinib 4 mg QD, upadacitinib 30 mg QD and tofacitinib 10 mg twice daily (Figure 1). Daily inhibition of TYK2 was 91% for zasocitinib 30 mg and 23% for deucravacitinib 6 mg, and minimal TYK2 inhibition (0–8%) was observed for baricitinib 4 mg, upadacitinib 30 mg and tofacitinib 10 mg (Figure 2). Simulated plasma concentrations of zasocitinib 30 mg and deucravacitinib 6 mg did not reach JAK1/3 IC 50 ; those of baricitinib 4 mg, upadacitinib 30 mg and tofacitinib 10 mg were above JAK1/3 IC 50 for 24 hours. JAK1/3 percent daily inhibition was 0% for zasocitinib 30 mg and 3% for deucravacitinib 6 mg, versus 91–97% for baricitinib 4 mg, upadacitinib 30 mg and tofacitinib 10 mg.
Conclusion: Selective TYK2 inhibition offers a distinct cytokine receptor associated proximal signal kinase inhibition profile. Zasocitinib showed greater and longer selective inhibition of TYK2-mediated signalling versus deucravacitinib at modelled comparative clinical dose, without affecting JAK1/3-mediated signalling.
REFERENCES: NIL.
Acknowledgements: This study was funded by Nimbus Discovery, Inc. and Takeda Development Center Americas, Inc. Writing assistance was provided by Tina Borg, PhD, of Oxford PharmaGenesis and funded by Takeda Development Center Americas, Inc. Nimbus refers to the group of entities including Nimbus Therapeutics LLC, Nimbus Discovery Inc., and Nimbus Lakshmi Inc.
Disclosure of Interests: Shailly Mehrotra is an equity holder and an employee of Takeda, Yasuyo Sano is an equity holder and a previous employee of Takeda, Elizabeth Wilson is an equity holder and an employee of Takeda, Chandra Durairaj is an equity holder and an employee of Takeda, Kok-Fai Kong is an equity holder and a previous employee of Takeda, Guliang Xia is an equity holder and a previous employee of Takeda, Faith Dunbar is an equity holder and an employee of Takeda, Petro Halkowycz is an equity holder and an employee of Takeda, Taylor Spector is an equity holder and an employee of Takeda, Christopher Bunick has received fees and served as an investigator, consultant and speaker for Almirall, Iain B. McInnes has received fees, speaker fees or grants from AstraZeneca, AbbVie, Cabaletta Bio, Causeway Therapeutics, Compugen, Eli Lilly, Evelo Biosciences, GSK, Janssen, Novartis, Pfizer and UCB.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (