

Background: Eosinophilic granulomatosis with polyangiitis (EGPA) was first described in 1951 as a form of disseminated necrotizing vasculitis characterized by extravascular granulomas in patients with asthma and tissue hypereosinophilia. Serum anti-neutrophil cytoplasmic antibodies (ANCA), typically directed against myeloperoxidase (MPO), and rheumatoid factor (RF) are frequently detected, strongly suggesting an autoimmune component to the disease. Eosinophilia is a hallmark feature of EGPA, and recently, mepolizumab (MPZ)—a monoclonal antibody targeting interleukin (IL)-5, which regulates eosinophil progenitor cell proliferation and activation—has been approved as a treatment option. While Th2 responses are recognized as central to the pathogenesis of EGPA, the roles of Th1 and Th17 responses remain less well understood. Additionally, cytokines upstream of IL-5, including “alarmins” such as IL-25, IL-33, and thymic stromal lymphopoietin (TSLP), have gained interest in EGPA research.
Objectives: To elucidate the pathogenesis of EGPA through (1) a cross-sectional analysis to quantify multiple humoral factors in the serum of untreated patients with EGPA and explore their interrelationships and (2) a longitudinal analysis to assess the behavior of these factors before and after the initial treatment, as well as after MPZ treatment.
Methods: We included 27 patients with EGPA (15 newly diagnosed) diagnosed at the University Hospital between May 2020 and April 2024. For comparison, 26 newly diagnosed patients with SLE or those with exacerbated SLE served as the control group. EGPA cases were classified according to the 1990 American College of Rheumatology (ACR) classification criteria. SLE cases were classified according to the 2019 European Alliance of Associations for Rheumatology/ACR classification criteria. Serum levels of eotaxin, fibroblast growth factor basic, granulocyte colony-stimulating factor, granulocyte-macrophage colony-stimulating factor, interferon gamma (IFN-γ), IL-1β, IL-1Ra, IL-2, IL-4, IL-5, IL-6, IL-7, IL-8, IL-9, IL-10, IL-12, IL-13, IL-15, IL-17, IFN-γ-inducible protein (IP)-10, monocyte chemotactic protein (MCP)-1, macrophage inflammatory protein (MIP)-1 alpha, MIP-1β, platelet-derived growth factor-BB, tumor necrosis factor alpha, vascular endothelial growth factor (VEGF), B-cell activating factor (BAFF), and regulated upon activation, normal T cell expressed and secreted (RANTES) were determined using bead array assays. ELISA was used to measure the IL-5, IL-25, IL-33, and TSLP levels. Sera were analyzed before and 4 ± 2 weeks after initial treatment (mainly high-dose glucocorticoid with or without cyclophosphamide pulse therapy; n = 15), as well as before and 12 ± 4 weeks after MPZ treatment (n = 18). Peripheral blood mononuclear cells (PBMCs) were isolated from three untreated patients with EGPA using density gradient centrifugation. Erythrocytes in the remaining cell population were lysed, and eosinophils were isolated using a magnetic bead-based eosinophil isolation kit (>90% purity). The remaining cells were considered neutrophils. Cell suspensions (10 6 cells/mL each) were incubated with or without 10 ng/mL phorbol 12-myristate 13-acetate (PMA) for 15 h. Culture supernatants were then collected, and IL-1Ra and galectin-10 concentrations were measured using bead array and ELISA, respectively.
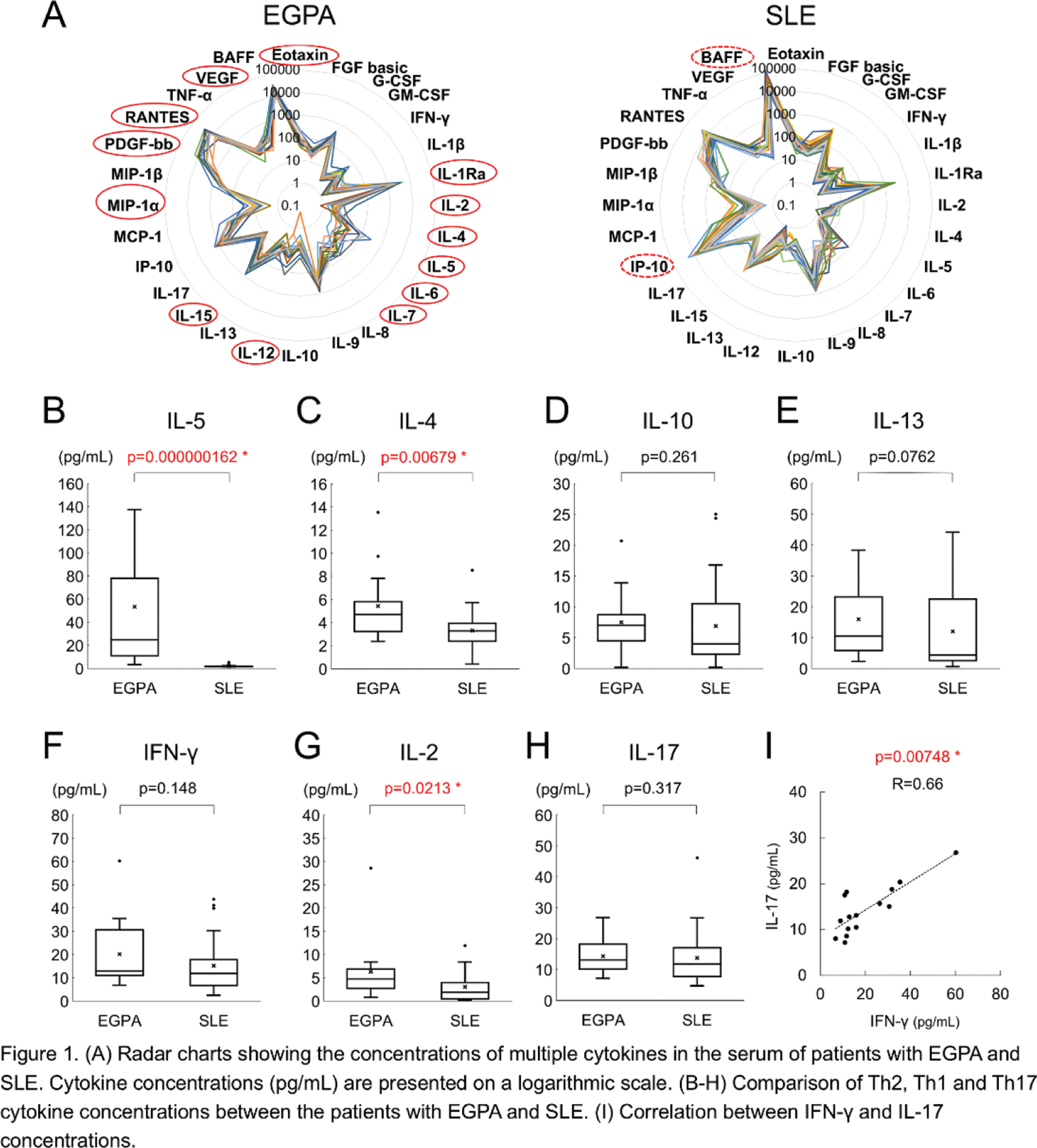
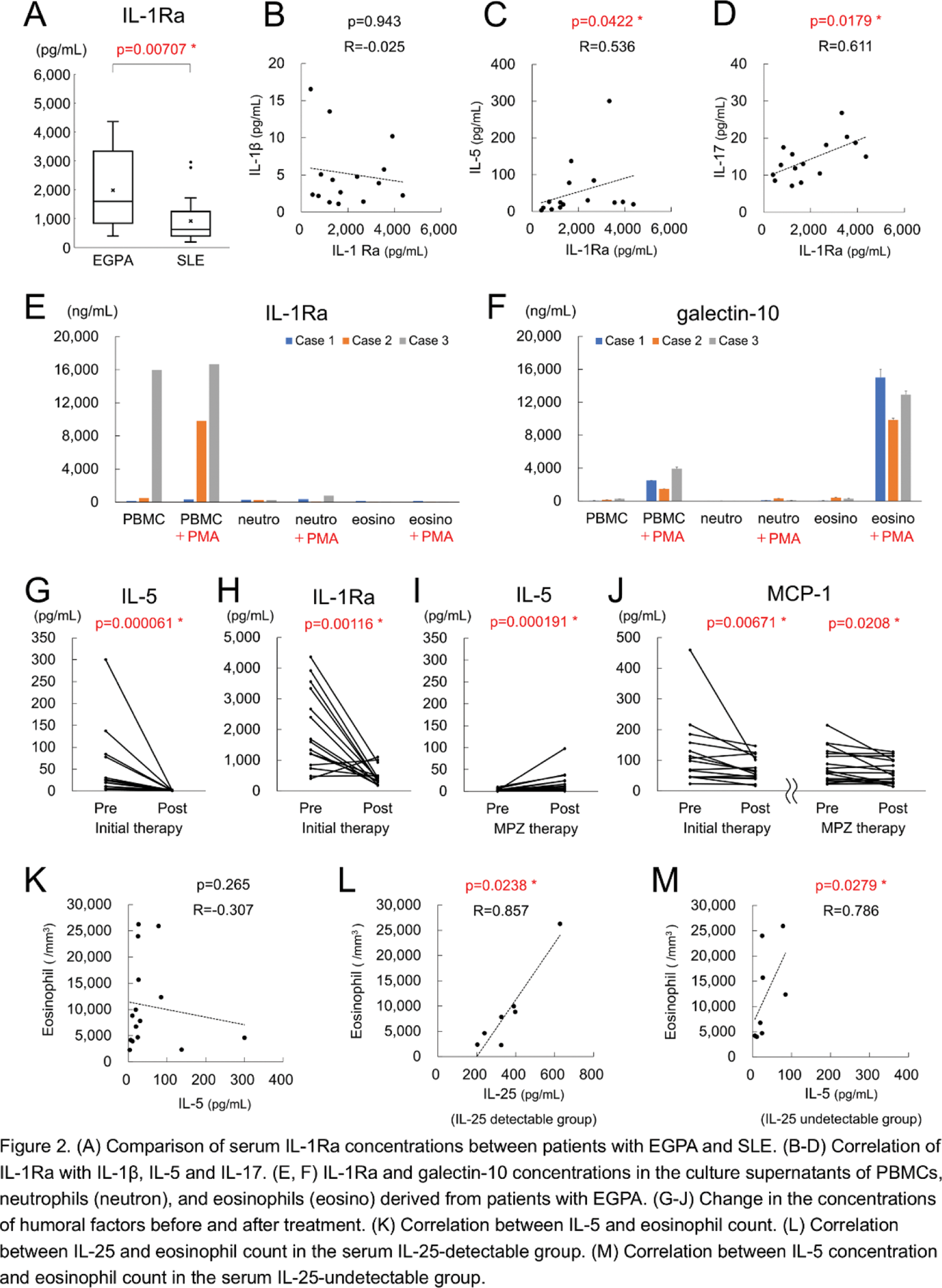
Results: Humoral factors were more elevated in patients with EGPA than in those with SLE (Figure 1A). Conversely, IP-10 and BAFF levels, both of which are reported to be induced by type I IFN, were higher in patients with SLE. Furthermore, IL-5 levels were significantly elevated in the EGPA group than in the SLE group, whereas IL-10 and IL-13 levels remained similar in both groups (Figure 1B–E). Th1 cytokines IFN-γ and IL-2 were slightly elevated in patients with EGPA than in patients with SLE, whereas IL-17, a Th17 cytokine, exhibited no difference (Figure 1F-H). A strong correlation was observed between IFN-γ and IL-17 (Figure 1I). IL-1Ra levels tended to be higher in patients with EGPA and were positively correlated with IL-5 and IL-17 levels but not IL-1β (Figure 2A–D). Notably, eosinophils in the gut have been reported to produce IL-1Ra, which suppresses Th17 differentiation. However, eosinophils derived from the peripheral blood of three patients with EGPA did not secrete IL-1Ra upon stimulation with PMA, indicating that eosinophils in the peripheral blood are not the source of IL-1Ra. In contrast, galectin-10 was highly detected (Figure 2E–F). Serum IL-5 and IL-1Ra levels significantly decreased after initial treatment (Figure 2G, H), whereas IL-5 levels increased after MPZ treatment (Figure 2I). MCP-1 levels decreased substantially after both initial and MPZ treatments (Figure 2J). Moreover, IL-25, IL-33, and TSLP, which are supposed to regulate IL-5 production, exhibited no consistent correlation with IL-5 or the eosinophil count. Additionally, no correlation was observed between IL-5 levels and the eosinophil count (Figure 2K). Interestingly, subgroup analysis revealed a strong correlation between IL-25 and eosinophil count in IL-25-detectable cases and between IL-5 and eosinophil count in IL-25-undetectable cases (Figure 2L-M).
Conclusion: Patients with EGPA did not exhibit a pronounced Th2 response compared with patients with SLE, except for IL-5. Th1 and Th17 responses were comparable between the patients with these diseases and strong correlations were observed between Th1 and Th17 cytokines. Quantification of various alarmins suggested that EGPA can be categorized into two subgroups—one in which eosinophil count is regulated by IL-25 and the other in which IL-25 is undetectable and eosinophil count is regulated by IL-5.
REFERENCES: NIL.
Acknowledgements: This research was supported by the Grants-in-Aid for Scientific Research from the Japan Society for the Promotion of Science (24K11602). The authors would like to thank all the patients who participated in this study. We would also like to thank Ms. Chiyomi Hayashi, Ms. Sachiko Mamada, and Ms. Chisato Udagawa for their technical assistance.
Disclosure of Interests: Kojiro Sato Ono Pharma, GlaxoSmithKline, Chugai, Asahi Kasei Pharma, Yasuyuki Kamata: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (