

Background: Zasocitinib (TAK-279) is a highly selective and potent, oral, allosteric tyrosine kinase 2 (TYK2) inhibitor. In a phase 2b trial in patients with active psoriatic arthritis (PsA; NCT05153148), significantly more patients treated with zasocitinib achieved American College of Rheumatology (ACR)20 responses at Week 12 than with placebo (15 mg [53.3%], 30 mg [54.2%], placebo [29.2%]; p = 0.002) [1].
Objectives: To present additional data on skin and overall disease activity from the phase 2b study.
Methods: This was a randomised, multicentre, double-blind, placebo-controlled study. Eligible patients were aged ≥ 18 years, with PsA symptoms for ≥ 6 months prior to screening, met CASPAR criteria, and had ≥ 3 tender and ≥ 3 swollen joint counts at enrolment despite prior treatment with biologics, disease-modifying antirheumatic drugs or non-steroidal anti-inflammatory drugs. If receiving concurrent conventional PsA treatments (permitted therapies were pre-defined in the study protocol), patients remained on stable doses throughout the study. Patients were randomised (1:1:1:1) to oral zasocitinib (5, 15 or 30 mg) or placebo, once daily for 12 weeks. Outcomes included achievement of minimal disease activity (MDA), 75/90/100% improvements from baseline in psoriasis area and severity index (PASI 75/90/100), change from baseline in PASI in patients with ≥ 3% psoriasis (PsO) body surface area (BSA) at baseline and Physician Global Assessment of PsO (PGA-PsO) response (0/1 and a ≥ 2-point improvement from baseline in patients with a PGA-PsO score ≥ 2 at baseline). Differences were assessed using a Mantel–Haenszel test (binary endpoints) and a mixed model for repeated measures (continuous endpoints); p values were nominal.
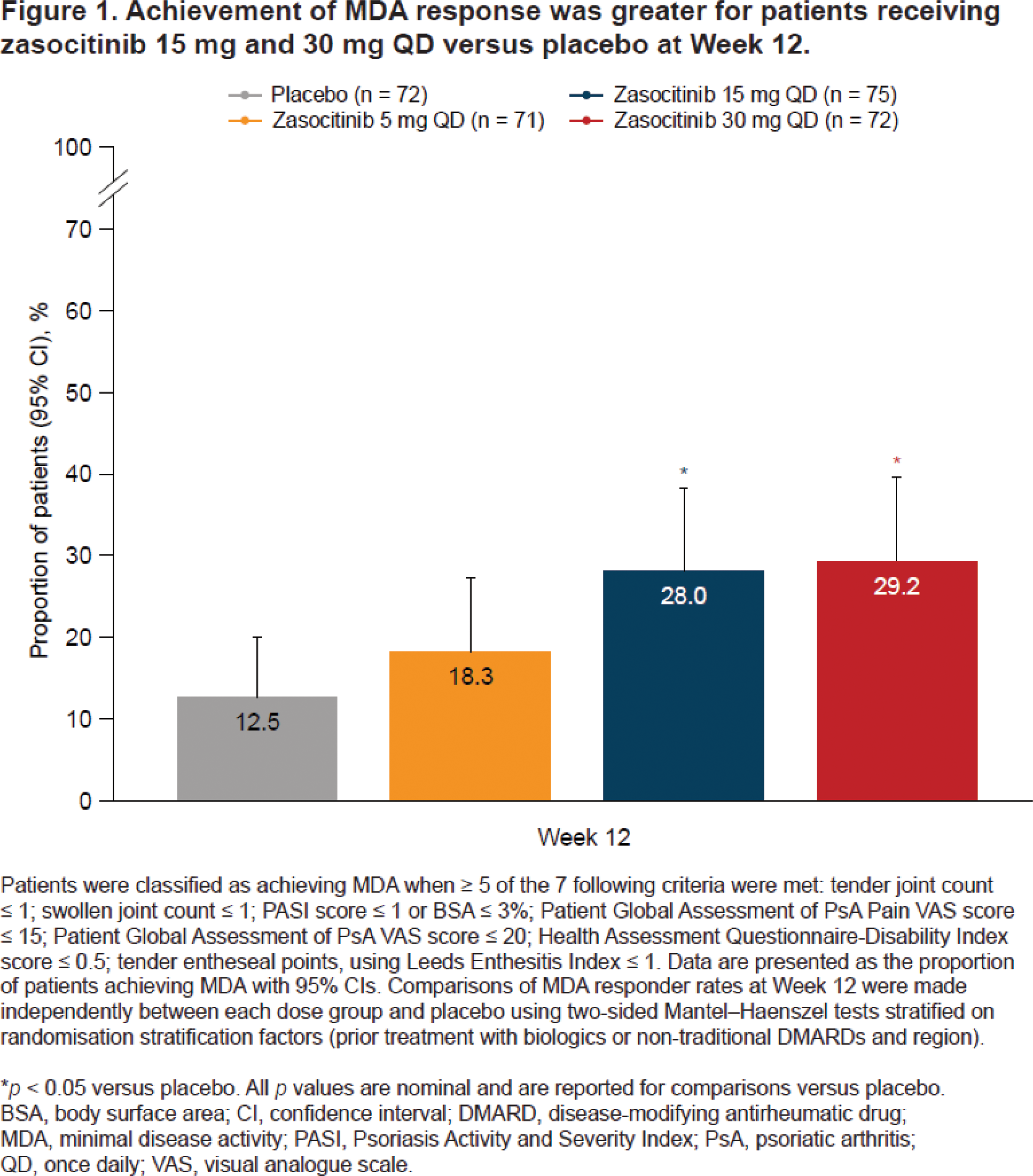
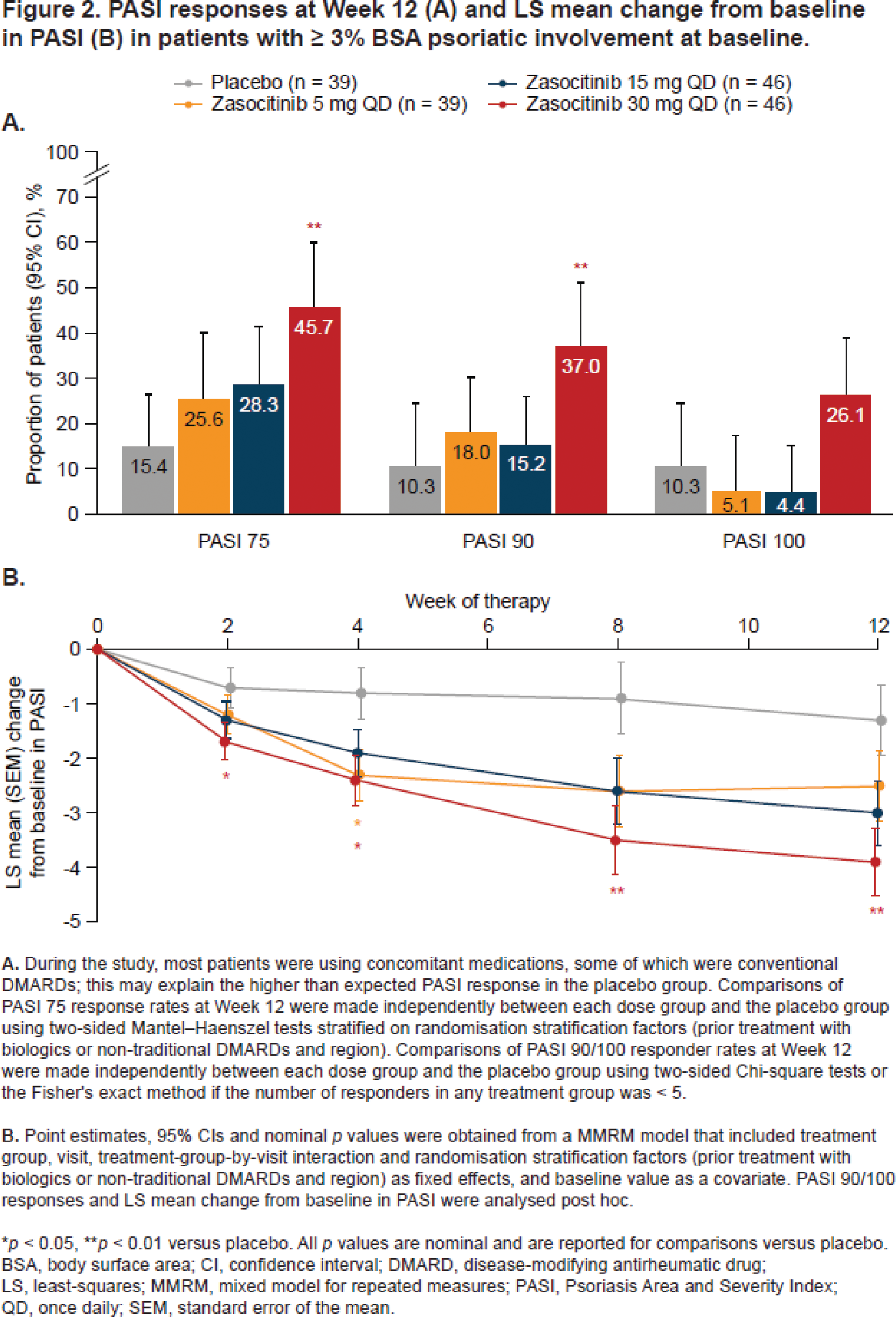
Results: Overall, 290 patients were randomised. Baseline demographics and disease characteristics were similar between groups; 58.6% had PsO BSA ≥ 3% (mean [standard deviation] baseline PASI, 6.2 [5.5]). At baseline, 59.7, 63.4, 61.3 and 65.3% of patients were receiving concomitant traditional DMARDs in the placebo and zasocitinib 5, 15 and 30 mg groups, respectively. Higher doses of zasocitinib resulted in higher rates of MDA at Week 12 compared with placebo (15 mg [28.0%], 30 mg [29.2%], placebo [12.5%]; both p < 0.05; Figure 1). In the 30 mg group, Week 12 PASI 75/90 responses were substantially higher than placebo (both p < 0.01) and 1 in 4 patients achieved PASI 100 (Figure 2A). Reductions in least-squares mean change from baseline in PASI were greater for zasocitinib 30 mg than placebo from Week 2 (–1.1 vs placebo; 95% CI: –2.0, –0.2; p < 0.05) and were maintained through Week 12 (–2.5 vs placebo; 95% CI: –4.3, –0.8; p < 0.01, Figure 2B). A substantially higher proportion of patients also achieved a PGA-PsO response with zasocitinib 30 mg than with placebo at Week 12 (32.8% vs 15.8%; p < 0.05). Zasocitinib was well tolerated with a safety profile consistent with that seen in the phase 2b PsO study [2].
Conclusion: At Week 12, treatment with zasocitinib 30 mg led to substantially higher responses versus placebo across all outcomes assessed, with exception to PASI 100. Higher doses of zasocitinib (15 and 30 mg) achieved higher rates of MDA and robust efficacy across all skin endpoints compared with placebo by Week 12, indicating meaningful improvement in core PsA domains. Skin responses were evident from Week 2 onwards, and more patients treated with zasocitinib 30 mg achieved total or near-total clearance of PsO lesions at Week 12 versus placebo. Zasocitinib was well tolerated in patients with PsA.
REFERENCES: [1] Kivitz A et al. Arthritis Rheumatol 2023;75(Suppl 9).
[2] Armstrong A et al. JAMA Dermatol 2024;160:1066–74.
Acknowledgements: This study was funded by Nimbus Discovery, Inc. and Takeda Development Center Americas, Inc. Writing assistance was provided by Tina Borg, PhD, of Oxford PharmaGenesis and funded by Takeda Development Center Americas, Inc. Nimbus refers to the group of entities including Nimbus Therapeutics LLC, Nimbus Discovery Inc. and Nimbus Lakshmi Inc.
Disclosure of Interests: Alice B Gottlieb has received honoraria as an advisory board member and consultant for Amgen, AnaptysBio, Avotres, Boehringer Ingelheim, Bristol Myers Squibb, Dice Therapeutics, Eli Lilly, Highlight Therapeutics, Janssen, Novartis, Sanofi, Teva Pharmaceuticals, UCB and XBiotech (stock options for rheumatoid arthritis), and has received research/educational grants from Highlight Therapeutics, Bristol Myers Squibb, Janssen and UCB (all paid to Mount Sinai School of Medicine), Elena Tomaselli Muensterman is an equity holder and employee of Takeda, Alan Kivitz has stocks or stock options in Amgen, GSK, Gilead, Novartis and Pfizer, has received consulting fees from Fresenius Kabi, Genzyme, Gilead, Grünenthal, GSK, Horizon, Janssen, Pfizer, Selecta, SynAct Pharma and Takeda, has received payment or fees for lectures, presentations, speakers’ bureaus, manuscript writing or educational events from AbbVie, Amgen, Eli Lilly, GSK, Pfizer and UCB, and has been a board or advisory board member for ChemoCentryx, Horizon, Janssen, Novartis, Princeton Biopartners and UCB, Eva Dokoupilova has received grant/research support from AbbVie, Eli Lilly, Gilead, Janssen-Cilag, Nimbus, Novartis and UCB Biopharma SPRL, Apinya Lertratanakul is an equity holder and employee of Takeda, Ting Hong is an equity holder and employee of Takeda, Jingjing Chen is an equity holder and employee of Takeda, Xenofon Baraliakos has received consulting fees/honoraria from, or been involved in speakers’ bureaus and scientific advisory boards for AbbVie, Amgen, Bristol Myers Squibb, Chugai, Eli Lilly, Galapagos, MSD, Novartis, Pfizer, Roche, Sandoz and UCB.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (