

Background: PRO-SPIRIT is a 24-month, multinational, prospective observational study of patients with psoriatic arthritis (PsA) receiving real-world treatment with 14 biologic and targeted-synthetic disease-modifying anti-rheumatic drugs (b/tsDMARDs), covering a range of mechanisms of actions. The study design, along with baseline characteristics and early (3-month) descriptive and comparative effectiveness, have been previously published [1].
Objectives: To report the 12-month comparative persistence and effectiveness of ixekizumab (IXE) and other b/tsDMARDs in 1,192 patients with PsA. Major endpoints included the achievement of remission defined by the clinical Disease Activity in PsA (cDAPSA) index (cDAPSA REM, score of ≤4), Minimal Disease Activity (MDA), and changes from baseline in affected body surface area (BSA).
Methods: The study included data from a total of 1,192 PsA patients who had initiated or switched to a new interleukin-17A (IL-17A) inhibitor (IL-17Ai), specifically either ixekizumab (IXE, N=343) or secukinumab (SEC, either beginning with 150 mg [SEC 150, n=86] or 300 mg [SEC 300, n=78]), IL-12/23i (N=34), IL-23i (N=56), tumor necrosis factor inhibitor (TNFi, N=437), Janus kinase inhibitor (JAKi, N=124), or phosphodiesterase-4 inhibitor (PDE4i, N=32). To ensure sufficient statistical power for comparative analysis, SEC 150 mg and SEC 300 mg were pooled into ‘SEC TOTAL’ (N=164) and those receiving an IL-12/23i or IL-23i were pooled into ‘IL-12/23i & IL-23i’ (N=90). Data from patients receiving PDE4i are not reported due to low sample size. To control for meaningful between-group differences at baseline [1], our statistical approach utilized the highly robust Frequentist Model Averaging (FMA [2]), which in this case consists of six statistical methods including Inverse Probability of Treatment Weighting (IPTW), stratification, and multiple different regressions. Comparative persistence (time-to-event) is reported as hazard ratios (HR, where values below 1.0 favor IXE over the comparator) with 95% confidence interval (CI). Categorical outcomes of comparative effectiveness analyses are reported as odds ratios (OR, where values above 1.0 favor IXE) with 95% CI, while the continuous outcomes are reported as Least Squares Mean (LSM) differences with 95% CI (i.e., change from baseline, where negative values favor IXE). Missing data were handled using multiple imputation. No multiplicity adjustments were done as part of this analysis.
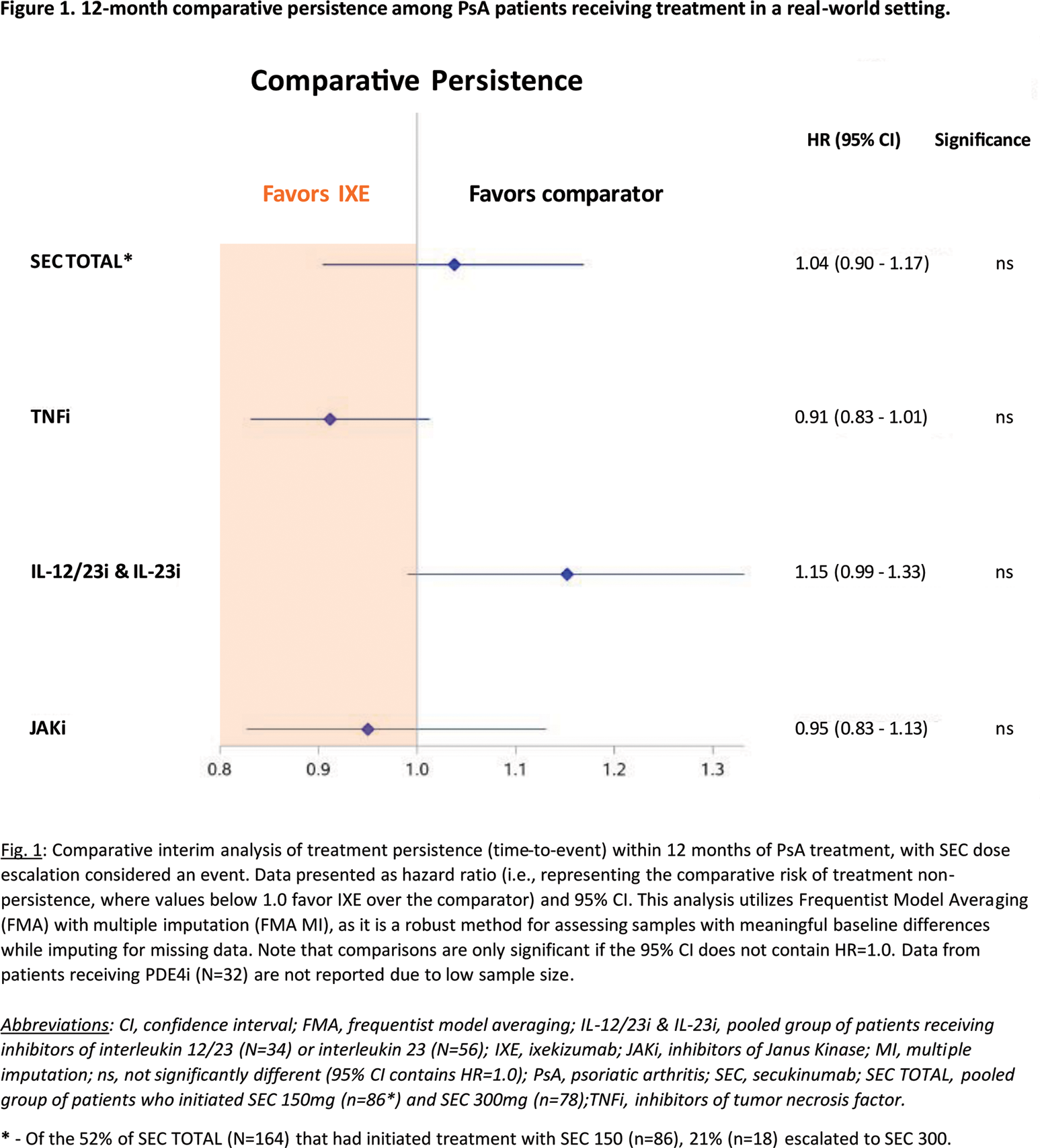
Results: 12-month comparative persistence
Comparative analysis of 12-month persistence revealed no statistical difference between IXE versus SEC TOTAL (HR of 1.04 [95% CI: 0.90 – 1.17]), TNFi (0.91 [0.83 – 1.01]), JAKi (0.95 [0.83 – 1.13]), or the group of IL-12/23i & IL-23i (1.15 [0.99 – 1.33]), respectively (Figure 1). Notably, although 52% of SEC TOTAL (N=164) had initiated treatment with SEC 150 (n=86), 21% (n=18) of these patients escalated to SEC 300, resulting in 59% of SEC TOTAL receiving SEC 300 treatment.
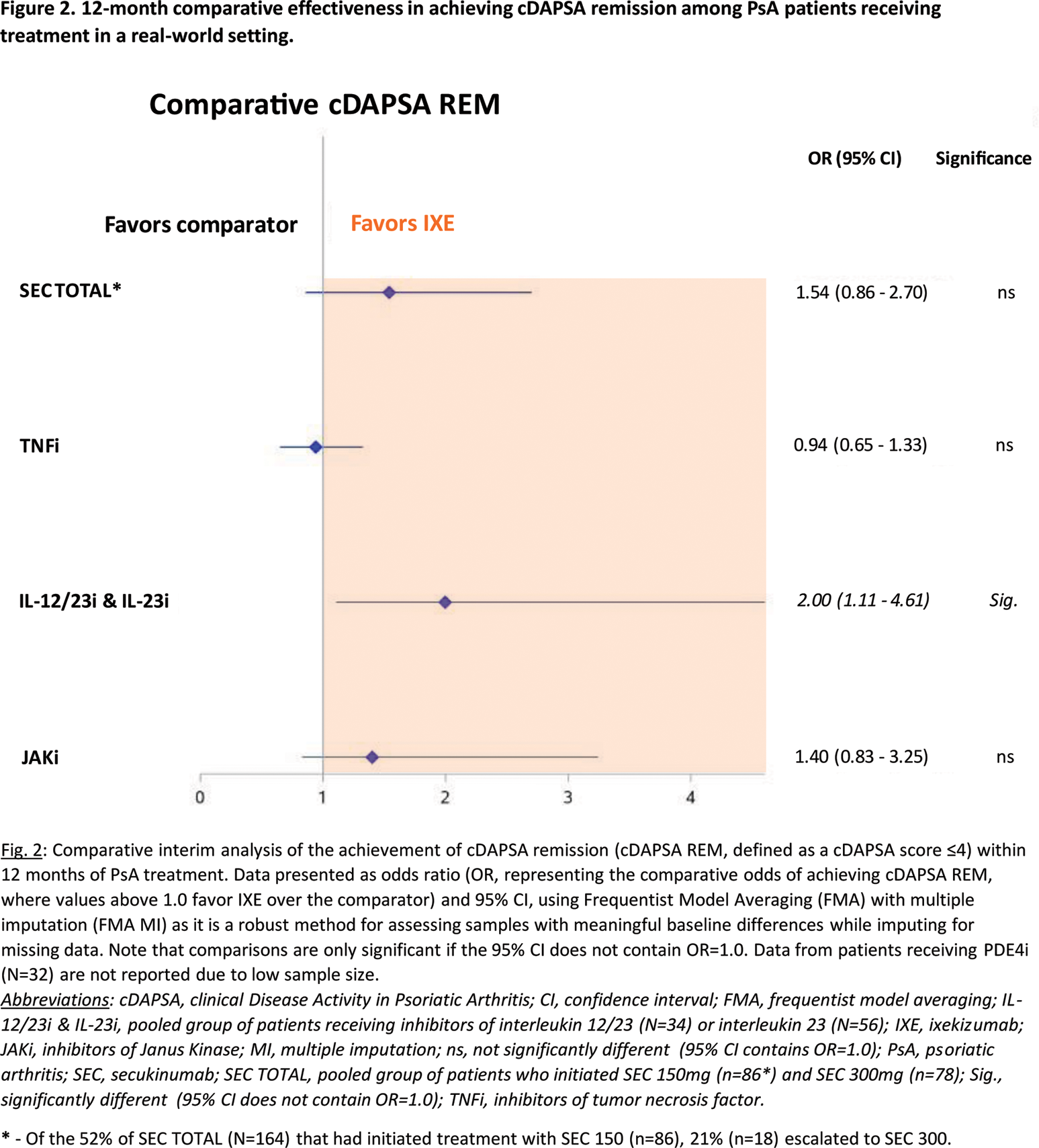
12-month comparative effectiveness
In the comparison of 12-month effectiveness indices, patients treated with IXE were twice as likely to achieve cDAPSA REM by the 12-month timepoint as those in the IL-12/23i & IL-23i group (OR of 2.00 [1.11 – 4.61]). Furthermore, IXE-treated patients tended to have higher (but not statistically different) odds of achieving cDAPSA REM than SEC TOTAL (IXE vs SEC TOTAL OR of 1.54 [0.86 – 2.70]) or JAKi (1.40 [0.83 – 3.25]) patients, while exhibiting similar effectiveness to TNFi (0.94 [0.65 – 1.33]) (Figure 2). When comparing the proportions of patients achieving MDA, despite the notable differences in baseline BSA (higher at baseline for IXE than JAKi or TNFi – see Kristensen et al. [1]), there were no statistical differences in the attainment of 12-month MDA (IXE vs SEC TOTAL: OR of 1.35 [95% CI: 0.66 – 2.47]; IXE vs TNFi: 0.82 [0.59 – 1.17]; IXE vs JAKi: 0.84 [0.40 – 1.78]; IXE vs IL-12/23i & IL-23i: 1.68 [0.75 – 4.36]). Finally, comparative analysis revealed that BSA decreased significantly more by the 12-month timepoint when patients were being treated with IXE than a TNFi (IXE vs TNFi mean change from baseline of -0.87 [95% CI: -1.4 – -0.11]); IXE was otherwise equivalent to other treatment groups in BSA improvement (IXE vs SEC TOTAL: -0.48 [-1.30 – 0.45]; IXE vs JAKi: 0.18 [-0.61 – 0.91]; IXE vs IL-12/23i & IL-23i: -0.28 [-0.98 – 0.47]).
Conclusion: At 12 months, although comparative persistence was not statistically different between treatment groups, comparative effectiveness analyses revealed that patients were twice as likely to attain cDAPSA REM with IXE than IL-12/23i & IL-23i, with similar improvements for both groups in BSA and MDA. IXE-treated patients also exhibited higher, albeit not statistically different, odds of attaining cDAPSA REM than those receiving SEC TOTAL or a JAKi. Relative to TNFi, IXE-treated patients exhibited significantly greater reductions in BSA by month 12, with similar proportions of both treatment groups achieving cDAPSA REM and MDA.
REFERENCES: [1] Kristensen LE, Ng KJ, Ngantcha M, Morel J, Lubrano E, Tillett W, Alten R, Chandran V, Ferrer ÀM, Zhu B, Kennedy D, Holzkämper T, Gullick N, Kronbergs A, Fakhouri W, de la Torre I, McGonagle DG. Comparative early effectiveness across 14 PsA drugs and 5 classes of PsA treatment: 3-month results from the PRO-SPIRIT study. RMD Open . 2024;10(3):e004318.
[2] Zagar A, Kadziola Z, Lipkovich I, Madigan D, Faries D. Evaluating bias control strategies in observational studies using frequentist model averaging. J Biopharm Stat . 2022;32(2):247-76.
Acknowledgements: Eli Lilly and Company would like to thank the investigators as well as the participants and their caregivers, without whom this work would not be possible. A full list of study investigators was published in the Supplemental Appendix of Kristensen et al., 2024.
Disclosure of Interests: Philipp Sewerin received speaker fees from AXIOM Health, AMGEN, AbbVie, Biogen, Bristol-Myers Squibb, Celgene, Chugai Pharma Marketing Ltd./Chugai Europe, Deutscher Psoriasis-Bund, Eli Lilly and Company, Gilead Sciences, Galapagos, Hexal Pharma, Janssen-Cilag, Johnson & Johnson, medi-login, Mediri GmbH, Novartis Pharma, Onkowissen GmbH, Pfizer, Roche, Rheumazentrum Rhein-Ruhr, Sanofi-Genzyme, Spirit Medical Communication, Swedish Orphan Biovitrum, and UCB Pharma, received consulting fees from AXIOM Health, AMGEN, AbbVie, Biogen, Bristol-Myers Squibb, Celgene, Chugai Pharma Marketing Ltd./Chugai Europe, Deutscher Psoriasis-Bund, Eli Lilly and Company, Gilead Sciences, Galapagos, Hexal Pharma, Janssen-Cilag, Johnson & Johnson, medi-login, Mediri GmbH, Novartis Pharma, Onkowissen GmbH, Pfizer, Roche, Rheumazentrum Rhein-Ruhr, Sanofi-Genzyme, Spirit Medical Communication, Swedish Orphan Biovitrum, and UCB Pharma, received grants or research support from AMGEN, AbbVie, Bundesministerium für Bildung und Forschung (BMBF), Bristol Myers Squibb, Celgene, Chugai, Deutsche Forschungsgesellschaft (DFG), Deutscher Psoriasis-Bund, Eli Lilly and Company, Janssen-Cilag, Novartis, Roche, Rheumazentrum Rhein-Ruhr, and UCB Pharma, Helena Marzo-Ortega received speaker fees from AbbVie, Biogen, Eli Lilly and Company, Janssen, Moonlake, Novartis, Pfizer, Takeda, and UCB Pharma, received consulting fees from AbbVie, Biogen, Eli Lilly and Company, Janssen, Moonlake, Novartis, Pfizer, Takeda, and UCB Pharma, received grants or research support from Janssen, Novartis, Pfizer, and UCB Pharma, Carlo Selmi received speaker fees from AbbVie, Amgen, Alfa-Sigma, Biogen, Eli Lilly and Company, EUSA Pharma - Recordati, Galapagos, Janssen, Novartis, Octapharma, Pfizer, Recordati Rare Disease, SOBI, and UCB Pharma, received consulting fees from AbbVie, Amgen, Alfa-Sigma, Biogen, Eli Lilly and Company, EUSA Pharma - Recordati, Galapagos, Janssen, Novartis, Octapharma, Pfizer, Recordati Rare Disease, SOBI, and UCB Pharma, Hagen Russ is a minor shareholder of Eli Lilly and Company, is an employee of Eli Lilly and Company, Khai Jing Ng is a minor shareholder of Eli Lilly and Company, is an employee of Eli Lilly and Company, Inmaculada De La Torre is a minor shareholder of Eli Lilly and Company, is an employee of Eli Lilly and Company, Marcus Ngantcha is a minor shareholder of Eli Lilly and Company, is an employee of Eli Lilly and Company, Dominika Kennedy is a minor shareholder of Eli Lilly and Company, is an employee of Eli Lilly and Company, Adela Gallego: None declared, Laura C. Coates received speaker fees from AbbVie, Amgen, Eli Lilly and Company, Janssen, Novartis, Pfizer, and UCB Pharma, received consulting fees from AbbVie, Amgen, Bristol Myers Squibb, Eli Lilly and Company, Enlivex, Janssen, Moonlake, Novartis, Pfizer, Takeda, and UCB Pharma, received grants or research support from Amgen, Janssen, and UCB Pharma.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (