

Background: Polymyalgia rheumatica (PMR) primarily affects adults over 50 years old. Glucocorticoids (GCs) are the cornerstone of treatment, starting with a moderate initial dose followed by gradual tapering. However, cumulative GC dosages can become substantial in cases of relapse or corticosteroid dependence (50% of patients), leading to significant side effects. In this context, GC-sparing treatments have recently emerged as promising therapeutic options in cases of GC resistance [1, 2], but also without GC [3] in early onset PMR at high risk of serious GC-related adverse events (osteoporosis, diabetes, hypertension, obesity, infections, etc.).
Objectives: The objective of this study was to show the cumulative glucocorticoid dosages administered to different profiles of PMR patients and to identify thresholds at which GC-sparing agents should be considered.
Methods: We developed prescription models based on international recommendations [4, 5] for various patient profiles (50 kg, 70 kg, 100 kg, with or without relapse) to build curves showing the cumulative dosage of steroid over the time. Then, for each patient profile, we calculated the cumulative GC dosages over the treatment course. To compare our results to the dosage used in clinical trial, we reported the cumulative dosage of steroid ordered in placebo groups from studies evaluating biologics in PMR. Then, we shared the results with a panel of experts of the French Society of Rheumatology, who were asked to provide feedback on indications for GC-sparing agents.
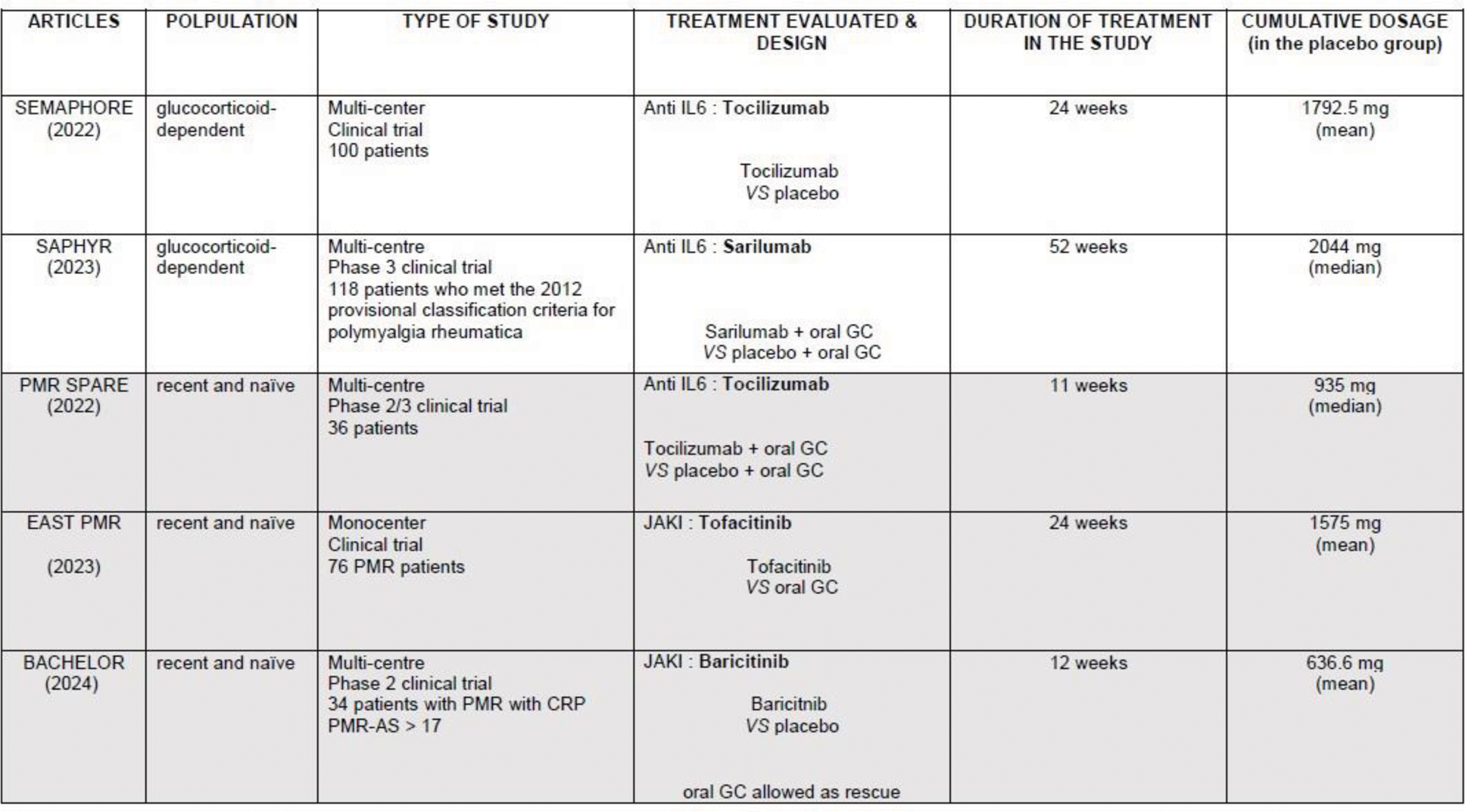
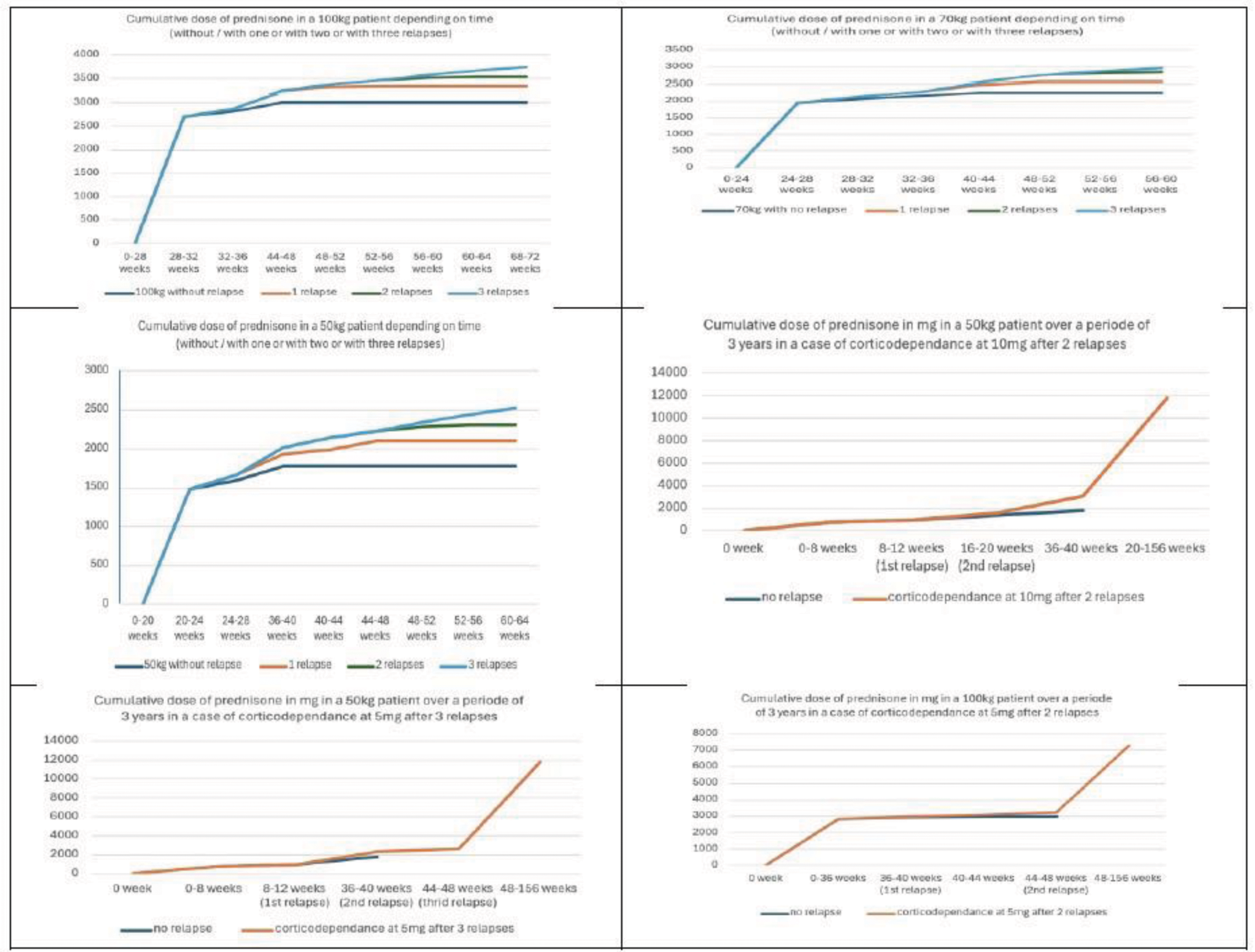
Results: By modeling GC use in different patient scenarios, we found that cumulative GC doses in recent-onset PMR ranged from 1.8 g (50 kg, no relapse) to 12.4 g (100 kg, GC-dependance at 10 mg, 3-year treatment duration) (Table 1). The majority of the cumulative dose was administered during the initial treatment phase, with relapses having minimal impact on final cumulative dosages (Figure 1). Body weight does not appear to be a major determinant of cumulative GC dose inflation. In cases of GC resistance, cumulative dosages ranged 6.6 g to 7.3 g for resistance at 5 mg and 11.8 g to 12.4 g for resistance at 10 mg, over a 3-year duration. In clinical trials, cumulative dosage in the placebo group is between 0.5 and 1 g at 12 weeks, 1 and 2 g at 24 weeks, and >2 g at 52 weeks (Table 2). 13/17 of the panel of experts considered that a cost-effective targeted treatment of PMR which cure PMR in 6 months should be ordered at all patients at inclusion, all 17 that targeted treatment is justified in case of risk factor of GC adverse event at inclusion and 8/17 that it would be justified in case of corticodependance for a cumulative dosage of GC>3 g.
Conclusion: Most experts consider that a cost-effective targeted treatment of PMR which cure PMR in 6 months could be ordered at all patients at inclusion and that it would be justified in case of corticodependance for a cumulative dosage of GC>3 grams. Future studies are needed to better define specific indications for these treatments in PMR.
REFERENCES: [1] Devauchelle-Pensec V, et al. Effect of Tocilizumab on Disease Activity in Patients With Active Polymyalgia Rheumatica Receiving Glucocorticoid Therapy: A Randomized Clinical Trial. JAMA, 2022. 328:1053-1062.
[2] Spiera RF et al. Sarilumab for Relapse of Polymyalgia Rheumatica during Glucocorticoid Taper.). N Engl J Med 2023; 389:1263-1272.
[3] Saraux a et al. Baricitinib in early polymyalgia rheumatica (BACHELOR): a randomized, placebo-controlled, parallel-group trial. Lancet Rheumatol. 2025. In press.
[4] Dejaco C, et al. Treat-to-target recommendations in giant cell arteritis and polymyalgia rheumatica. Ann Rheum Dis. 2024 2;83:48-57.
[5] Wendling D, et al. Recommendations of the French Society of Rheumatology for the management in current practice of patients with Polymyalgia Rheumatica. Joint Bone Spine. 2024;91:105730.
Table 1.
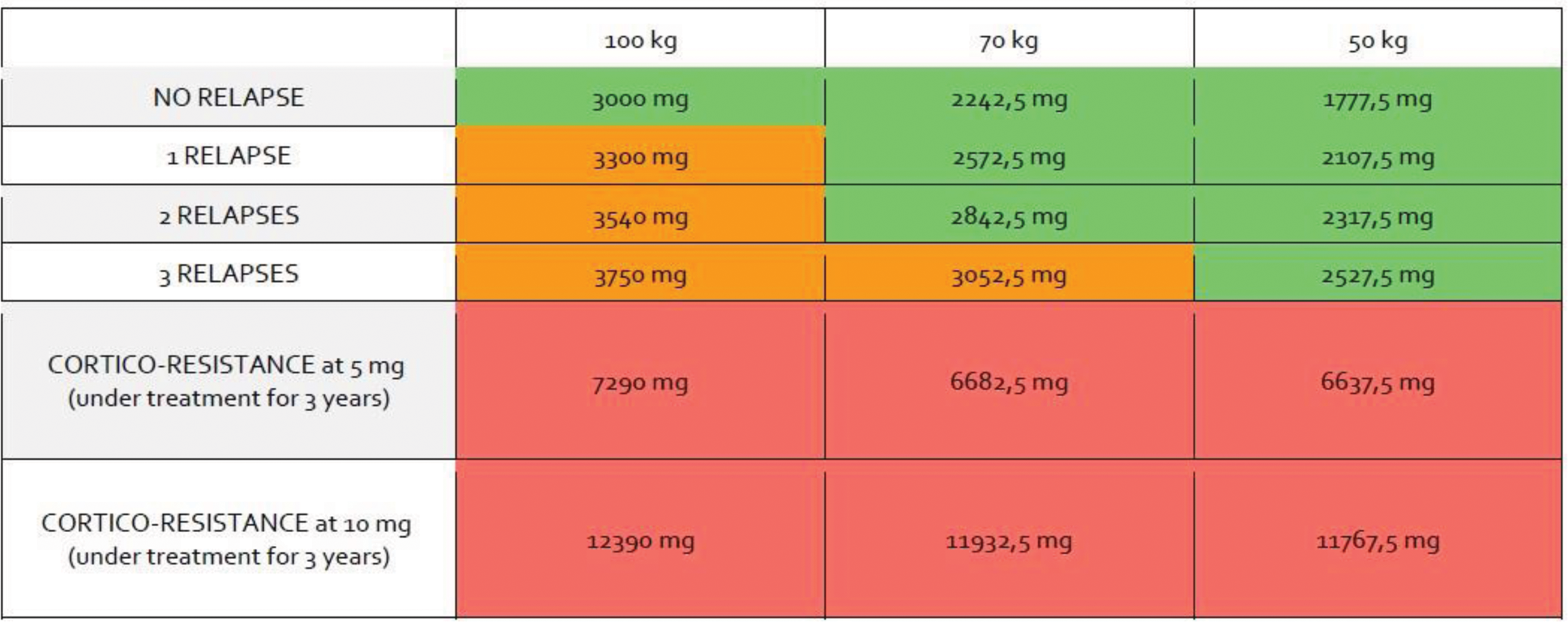
Table 2.
Acknowledgements: French Society of Rheumatology.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (