

Background: An increased prevalence of polyautoimmunity (defined as having two or more autoimmune disease (AD)) in patients with rheumatoid arthritis (RA) compared to the general population has been widely demonstrated [1, 2] and a shared dysregulation of the inflammatory response has been proposed as a possible explanation [3]. Biologic disease-modifying antirheumatic drugs (bDMARDs) targeting immunological inflammatory modulators [4], have in recent years gained grounds as an effective treatment for RA, and research has begun to investigate the impact of bDMARDs on the development of polyautoimmunity in patients with RA.
Objectives: The objective of this study was to compare the incidence of polyautoimmunity in patients with RA treated with bDMARDs compared to conventional disease-modifying antirheumatic drugs (csDMARDs).
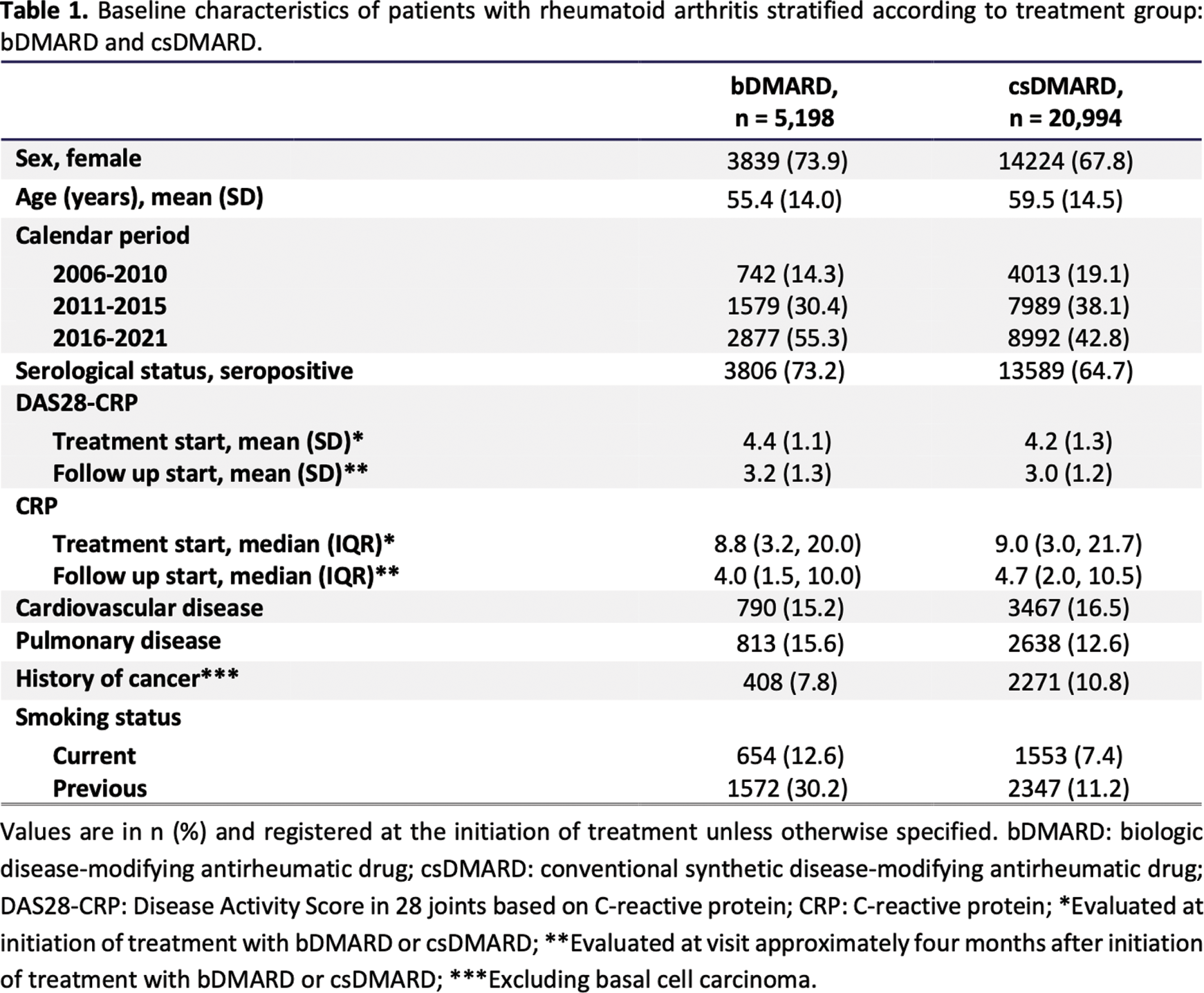
Methods: This Danish nationwide register-based cohort study included 26,192 adult (18+) patients diagnosed with RA (International Classification of Diseases, 10th revision (ICD-10) ¨M05 or M06) and in treatment with a disease-modifying antirheumatic drug (DMARD). Eligibility required registration in the Danish Rheumatology Quality Register (DANBIO) between 2006 through 2021. Status on polyautoimmunity was determined regarding 21 specific non-rheumatic ADs selected through a Danish epidemiology study: Autoimmune thyroid disease (AITD); Type 1 diabetes mellitus (T1DM); primary adrenocortical insufficiency; autoimmune polyglandular failure; Crohn’s disease (CD); ulcerative colitis (UC); primary biliary cirrhosis; autoimmune hepatitis; primary sclerosing cholangitis; celiac disease; autoimmune pancreatitis; multiple sclerosis; neuromyelitis optica; Guillain-Barré syndrome; myasthenia gravis; pemphigus vulgaris; bullous pemphigoid; dermatitis herpetiformis; alopecia areata; vitiligo; or lichen sclerosus et atrophicus [5]. The Danish National Patient Registry (DNPR), The National Prescription Registry (NPR), and an algorithm utilizing these registries in combination with inclusion and exclusion of specific ICD-10 diagnostic codes were applied to identify the outcome ADs listed above with the algorithm specifically applied to avoid misclassification of AITD and T1DM. Three cause-specific cox analyses were performed to calculate hazard ratios (HRs) comparing bDMARDs treated patients to patients treated with csDMARDs: one crude analysis was performed, and two with adjustments for the following covariates age, sex, seropositive or seronegative RA, disease activity (measured through 28 Joint Disease Activity Score (DAS28)), AD prior to index date, and comorbidities (cardiovascular disease, pulmonary disease, and cancer excluded basal cell carcinoma (BCC)).
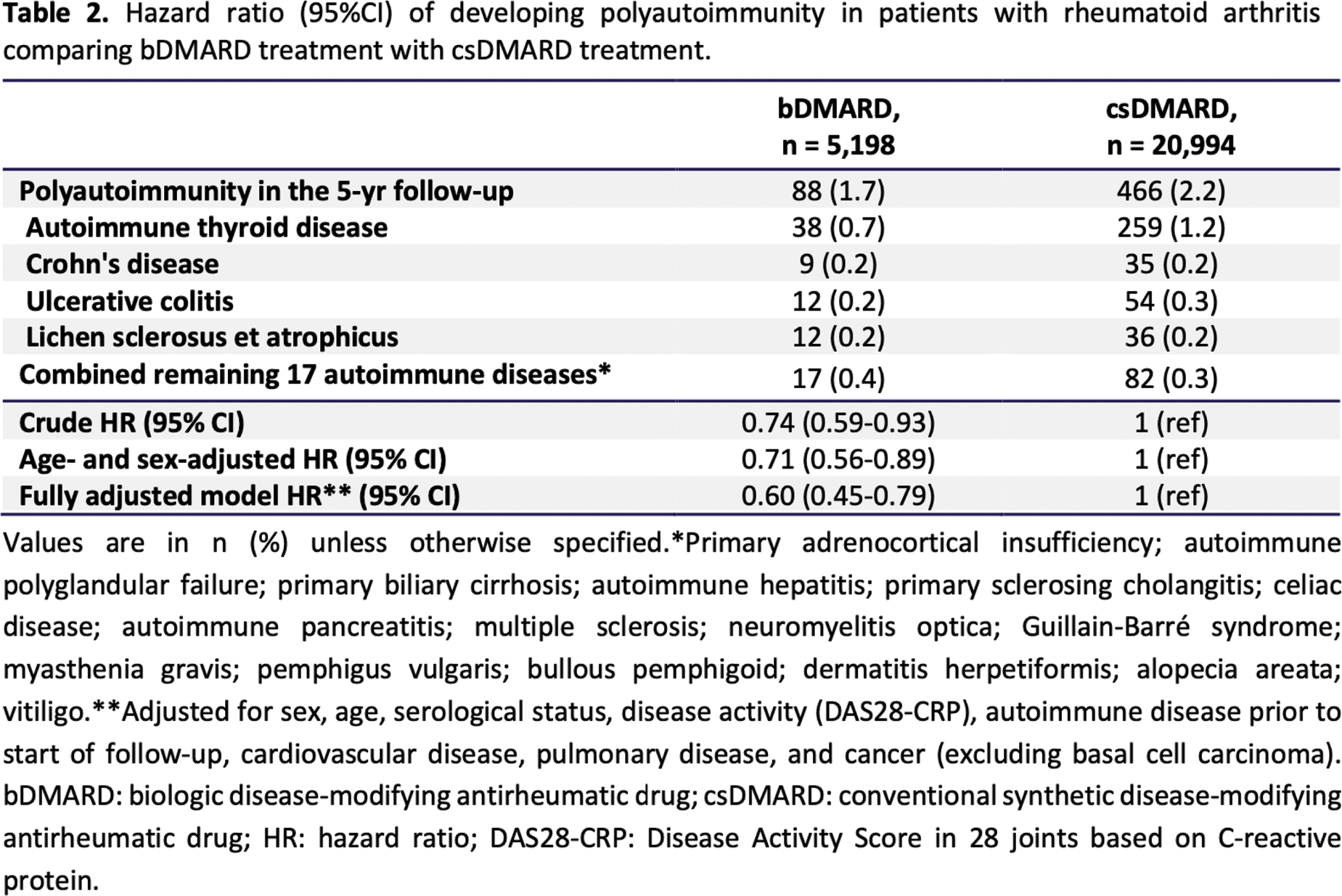
Results: The study cohorts consisted of 5,198 patients treated with bDMARDs and 20,994 patients treated with csDMARDs. After a 5-year follow-up period (median follow-up time 4.74 years) 1.7% of patients in the bDMARD treated group developed an additional AD compared to 2.2% in the csDMARD group. Adjusted cox analysis showed a statistically significant lower risk for developing polyautoimmunity among patients treated with a bDMARD compared to a csDMARD with a HR of 0.60 (95% confidence intervals (CI) 0.45-0.79). The most frequently developed AD was autoimmune thyroid disease (AITD) accounting for 0.7% and 1.2% of the cohorts, respectively.
Conclusion: This study demonstrated a reduced risk of developing polyautoimmunity in patients with RA treated with a bDMARD as opposed to a csDMARD both before and after adjusting for relevant covariates. These results warrant further research delving into the different subtypes of bDMARDS or the inclusion of other ADs.
REFERENCES: [1] Somers EC, Thomas SL, Smeeth L, Hall AJ. Are Individuals With an Autoimmune Disease at Higher Risk of a Second Autoimmune Disorder? Am J Epidemiol . 2009;169(6):749-755. doi:10.1093/AJE/KWN408.
[2] Rojas-Villarraga A, Amaya-Amaya J, Rodriguez-Rodriguez A, Mantilla RD, Anaya JM. Introducing polyautoimmunity: Secondary autoimmune diseases no longer exist. Autoimmune Dis . 2012;1(1). doi:10.1155/2012/254319.
[3] Anaya JM. The autoimmune tautology. Arthritis Res Ther . 2010;12(6). doi:10.1186/ar3175.
[4] Aletaha D, Smolen JS. Diagnosis and Management of Rheumatoid Arthritis: A Review. JAMA . 2018;320(13):1360-1372. doi:10.1001/JAMA.2018.13103.
[5] Eaton WW, Rose NR, Kalaydjian A, Pedersen MG, Mortensen PB. Epidemiology of autoimmune diseases in Denmark. J Autoimmun . 2007;29(1):1-9. doi:10.1016/J.JAUT.2007.05.002
Acknowledgements: The authors extend their gratitude to Danish rheumatologists for their contributions to the DANBIO register. We are grateful to Reimar Bo Thomsen who provided the initial advice on the algorithm to define type 1 diabetes mellitus.
Disclosure of Interests: Sissel Stocklund: None declared, Frederikke Olivia Frimer: None declared, Bergur Magnussen: None declared, Kirsten S. Duch: None declared, Lene Dreyer has received a research grant (paid to her institution) from BMS and AbbVie outside the current manuscript. She is member of the steering committee of the Danish Rheumatology Quality Registry (DANBIO, DRQ), which receives public funding from the hospital owners and funding from pharmaceutical companies, Mette Holland-Fischer is a member of the speakers’ bureau at AbbVie, Salome Kristensen: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (