

Background: Up to a third of lupus nephritis (LN) patients will progress to end stage kidney disease (ESKD) which carries increased morbidity and mortality. Identifying early predictors of chronic kidney disease (CKD) progression could help stratify high-risk patients and refine treatment response definitions. However, many predictors as well as the prognostic value of clinical response definitions and serological responses, remain inadequately studied due to the need for long-term follow up.
Objectives: This study aimed to identify clinical, histological, and serological predictors of kidney survival in LN.
Methods: The Accelerating Medicines Partnership (AMP) enrolled patients undergoing a clinically indicated (UPCR >0.5) kidney biopsy with resultant histology class II, III, IV, and/or V lupus nephritis (LN). Clinical, demographic and serological features were collected from the time of diagnostic biopsy. Serum samples were collected at the time of kidney biopsy and after 12-months. Autoantibodies against dsDNA, chromatin, ribosomal-P, Ro, La, Sm, SmRNP, RNP, Jo-1, Scl-70, and centromere-B were measured using the BioPlex 2200® ANA kit (Bio-Rad Technologies, Hercules, CA), while anti-C1q was measured by ELISA (QUANTA Lite®, Werfen, Bedford, MA). Due to near-universal negativity, Jo-1, Scl-70, and centromere-B were excluded from analyses. Response was defined at 1 year for patients with baseline UPCR≥1. Complete response was defined as UPCR <0.5, normal serum creatinine (≤1.3 mg/dL), or, if abnormal, ≤125% of baseline, and prednisone taper to ≤10 mg/day. Partial response was defined by >50% reduction in UPCR without meeting UPCR criterion for complete response, normal creatinine (≤1.3 mg/dL) or, if abnormal, ≤125% of baseline, and prednisone dose ≤15 mg/day. Histological features were centrally scored. Kidney function loss was defined as a sustained ≥40% decline in estimated glomerular filtration rate (eGFR) or progression to ESKD. A Cox-proportional hazards model was employed to identify predictors.
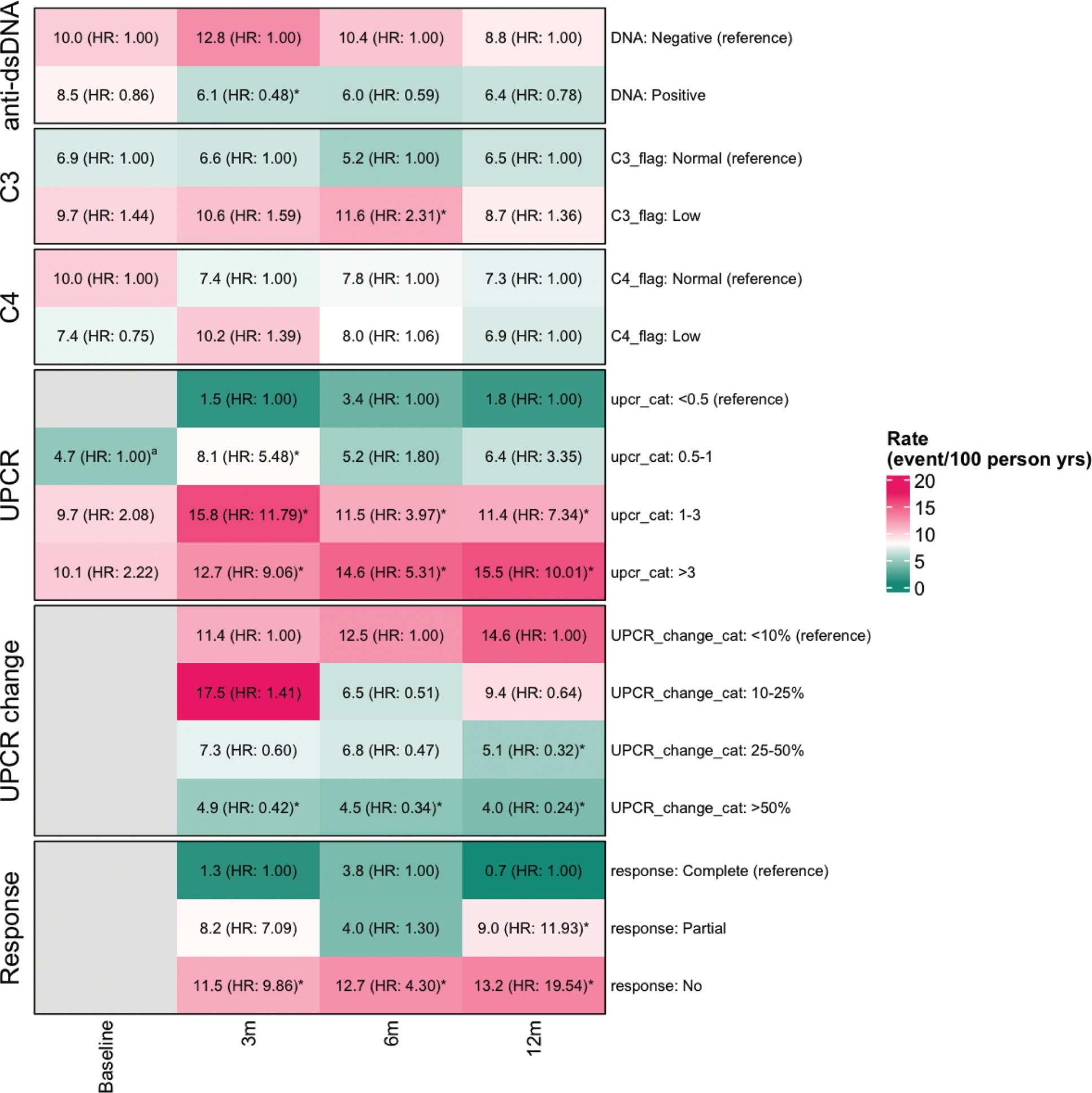
Results: We included 172 patients with a median follow-up time of 4.6 years (range 0.5-7.8), of whom 150 (87.2%) had >3 years follow up. Clinical and demographic features are summarized in Table 1. A third of patients (57/172) developed eGFR loss with a median time to event of 2.6 years (range 0.13-7.1). Predictors of eGFR loss at time of biopsy included lower eGFR (especially eGFR <30 ml/min, HR 5.4), repeat biopsy status (HR 2.5), and NIH Chronicity Index (histological damage, HR 1.3 per unit). Sex, race, age, BMI, proteinuria, ISN class, NIH Activity Index, C3, C4, and anti-dsDNA were not associated with eGFR loss. Among histological features, interstitial inflammation (HR 2.32), glomerulosclerosis (HR 1.88), fibrous crescents (HR 2.79), tubular atrophy (HR 1.96), and interstitial fibrosis (HR 1.89) were associated with eGFR loss (Table 1). Baseline and 1-year concentrations of the 9 autoantibodies analyzed were not associated with GFR loss. Autoantibody normalization at 1 year did not protect from GFR loss. Lack of clinical response at 3, 6, or 12 months was associated with future eGFR loss (Figure 1). Partial response at 12 months had higher risk of eGFR loss compared to complete response (HR 11.9), but lower than no response (HR 19.5). Reduction of UPCR >50% at 3 and 6 months was protective (HR 0.42 and 0.34, respectively). Four patients with UPCR <0.5 at 1 year developed eGFR loss (1.8/100 person years) (Figure 1).
Conclusion: Low baseline eGFR and histological damage, but not the NIH Activity Index or ISN class, predicted eGFR loss. Serological responses did not protect from eGFR loss. Improvement of UPCR was associated with lower risk of eGFR loss, though eGFR loss still occurred in patients in clinical remission (UPCR <0.5) at 1 year.
Table 1. Association of baseline clinical, demographic, and histological features with GFR loss
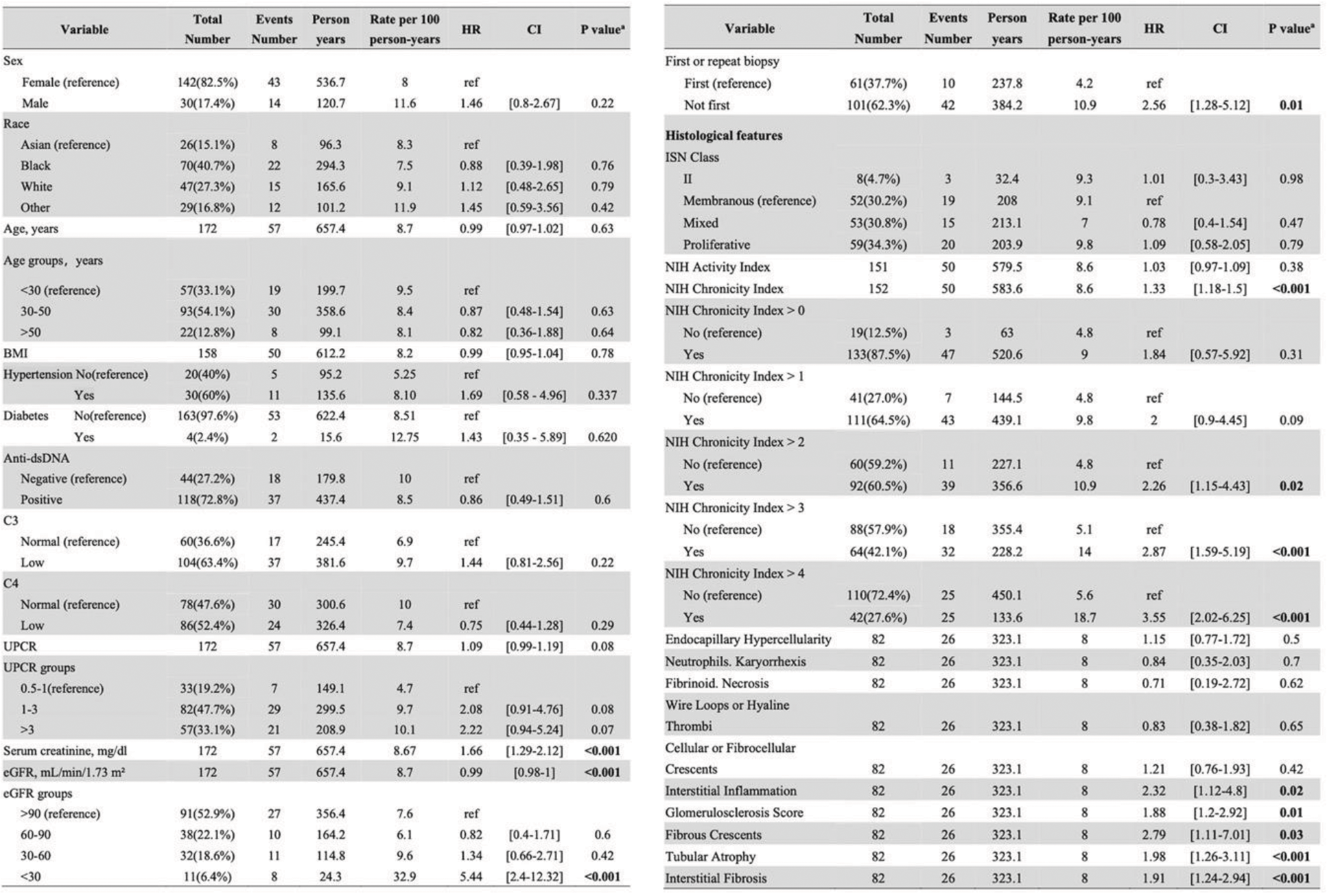
eGFR, estimated glomerular filtration rate; HR, Hazard Ratio; ISN, International Society of Nephrology; LN, lupus nephritis; UPCR, urine protein-creatinine ratio.
a Based on Cox proportional hazards model.
Heatmap summarizing the risk of future eGFR loss (Cox proportional hazards model) for clinical characteristics and treatment response at baseline and follow up timepoints (3 months, 6 months, and 12 months). Each cell indicates the rates of eGFR loss per 100 person-years and the point estimate of the Hazard ratio (HR). Statistically significant associations (P < 0.05) are indicated by *. a At baseline, the UPCR category “0.5-1” serves as the reference for analysis. UPCR change is categorized based on the percent decrease in UPCR from baseline.
REFERENCES: NIL.
Acknowledgements: This work was funded by the Plank Family Foundation and the Jerome L. Greene Foundation. The Hopkins Lupus Cohort is supported by NIH R01-DK-134625. Additionally, this research was supported by the Accelerating Medicines Partnership® Rheumatoid Arthritis and Systemic Lupus Erythematosus (AMP® RA/SLE) Network, a public-private partnership involving AbbVie Inc., Arthritis Foundation, Bristol-Myers Squibb Company, Foundation for the National Institutes of Health, GlaxoSmithKline, Janssen Research and Development, LLC, Lupus Foundation of America, Lupus Research Alliance, Merck & Co., Inc. Sharp & Dohme Corp., National Institute of Allergy and Infectious Diseases, National Institute of Arthritis and Musculoskeletal and Skin Diseases, Pfizer Inc., Rheumatology Research Foundation, Sanofi, and Takeda Pharmaceuticals International, Inc. The AMP Network aims to develop new methods for identifying and validating promising biological targets for diagnostics and drug development. Funding was provided through grants from the National Institutes of Health (UH2-AR067676, UH2-AR067677, UH2-AR067679, UH2-AR067681, UH2- AR067685, UH2-AR067688, UH2-AR067689, UH2-AR067690, UH2-AR067691, UH2-AR067694, and UM2- AR067678).
Disclosure of Interests: Shangzhu Zhang: None declared, Laurence Magder: None declared, Daniel Goldman: None declared, Judith A. James: None declared, Joel M Guthridge: None declared, Carla Guthridge: None declared, Peter Izmirly: None declared, Jill P Buyon: None declared, H.Michael Belmont: None declared, Richard A. Furie: None declared, Noa Schwartz: None declared, Chaim Putterman: None declared, Jennifer Barnas: None declared, Jennifer H. Anolik: None declared, Sarah French: None declared, Maria Dall’Era: None declared, Jeffrey Hodgin: None declared, Dawit S. Demeke: None declared, the Accelerating Medicines Partnership in RA/SLE Network: None declared, Michelle Petri Consultant: AbbVie, Alexion, Amgen, AnaptysBio, Annexon Bio, Arthros-FocusMedEd, AstraZeneca, Atara Biosciences, Aurinia, Autolus, Bain Capital, Baobab Therapeutics, Biocryst, Biogen, Boxer Capital, Cabaletta Bio, Caribou Biosciences, CTI Clinical Trial and Consulting Services, CVS Health, DualityBio, Escient Pharmaceuticals, Emergent Biosolutions, Exo Therapeutics, Gentibio, GSK, iCell Gene Therapeutics, IconPLC, IQVIA, Innovaderm Research, Kezar Life Sciences, Kira Pharmaceuticals, Eli Lilly, Merck EMD Serono, Nexstone Immunology, Nimbus Lakshmi, Novartis, Ono Pharma, Owkin, PPD Development, Proviant, Regeneron, Seismic Therapeutic, Senti Biosciences, Sinomab Biosciences, Steritas, Takeda, Tenet Medicines, TG Therapeutics, UCB, Variant Bio, Worldwide Clinical Trials, Zydus, Grant/research support: Aurinia, Exagen, GSK, Janssen, Andrea Fava consultant: AstraZeneca, BMS, Artiva, AnnexonBio, Sanofi, UCB, Bain Capital, Grant/research support: Exagen.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (