

Background: Systemic lupus erythematosus (SLE) is a chronic autoimmune disease characterized by the activation of the immune system, leading to multi-organ damage. Immune Thrombocytopenia (ITP) is a common hematological complication of SLE, affecting nearly half of the patients and often presenting as the initial symptom. Telitacicept, a dual-target inhibitor of B `lymphocyte stimulator (BLyS/BAFF) and a proliferation-inducing ligand (APRIL), has shown promising results in the treatment of SLE and other autoimmune diseases. However, its efficacy and safety in treating SLE patients with ITP have not been studied.
Objectives: This study aims to evaluate the therapeutic effect and safety of telitacicept in the treatment of patients with SLE complicated by ITP.
Methods: This study retrospectively analyzed the data of 22 SLE patients with thrombocytopenia who received telitacicept treatment at West China Hospital from February 2022 to October 2024. The treatment regimen of telitacicept was 160mg/week, with an infusion every 4 weeks, and all patients received telitacicept treatment for no less than 3 months. The clinical therapeutic effect of telitacicept was comprehensively evaluated by comparing the platelet counts at baseline and at 0.5, 1, 2, 3, and 6 months after telitacicept treatment.
Results: The median age of the patients included in this study was 37.36±12.33 years (range 13-55 years), including 18 female patients and 4 male patients. The median duration of thrombocytopenia before telitacicept treatment was 22.97 months. The previous treatments of the 22 patients included: corticosteroids (n=21), cyclosporine (n=6), tacrolimus (n=4), mycophenolate mofetil (n=5), methotrexate (n=2), cyclophosphamide (n=5), belimumab (n=3), and hydroxychloroquine (n=13). In addition, 5 patients had received intravenous immunoglobulin therapy, and 1 patient had received splenic embolization. After 6 months of telitacicept treatment, the platelet levels of patients significantly increased compared with the baseline (157.9×10^9 cells/L vs 38.8×10^9 cells/L; p<0.01). Among them, 19 patients (86.4%) achieved complete remission, and 1 patient (4.5%) achieved partial remission. In addition, telitacicept also showed a significant steroid-sparing effect. After 6 months of treatment, the median dose of glucocorticoids in patients was significantly lower than at baseline (9.7 mg/day vs 92.2 mg/day; p<0.01). The main adverse reactions observed in this study were infections, with 1 patient experiencing pulmonary infection and 1 patient developing herpes zoster.
Platelet response to telitacicept treatment. A. Platelet counts before and after telitacicept treatment. B. Overall response rates during the follow-up period at various time points. C. Glucocorticoid (GC) dosages before and after telitacicept treatment. The box plots indicate the median (central bold line), first quartile (lower box limit), and third quartile (upper box limit). Whiskers extend to the most extreme data points not considered outliers. Statistical significance is denoted as *P < 0.05 and **P < 0.01 compared to pre-treatment levels. NR: No response, PR: Partial response, CR: Complete response.
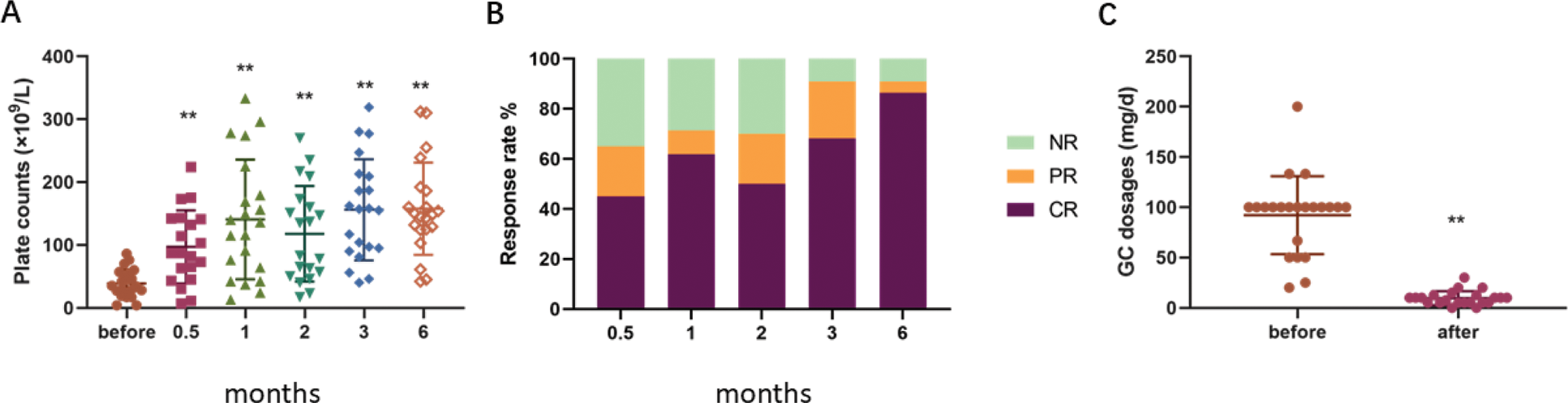
Conclusion: Telitacicept shows promising clinical results in the treatment of SLE patients with thrombocytopenia. Further prospective studies are needed to verify these findings in a larger patient population.
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (