

Background: Recent guidelines and recommendations for lupus nephritis (LN) recommend rapid glucocorticoid (GC) reduction; however, robust supporting evidence remains limited.
Objectives: This study aimed to evaluate the impact of rapid GC reduction on renal outcomes in patients with proliferative LN.
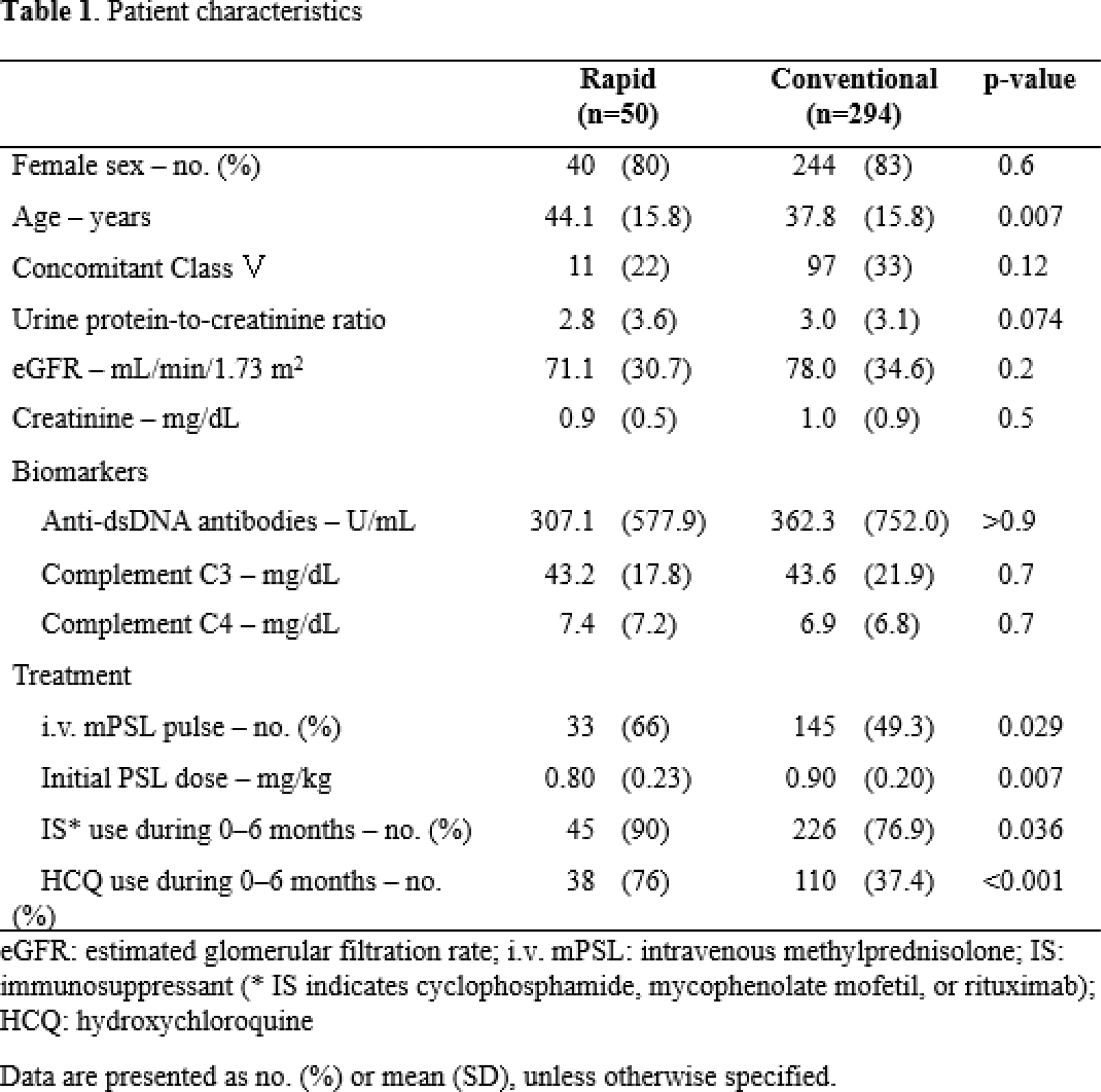
Methods: We conducted a multicentre retrospective chart review of patients with GC-naïve, biopsy-proven proliferative LN with available urinary protein-to-creatinine ratio (UPCR) data before and 52 weeks after GC treatment. Patients who reduced their prednisolone-equivalent dose to ≤ 7.5 mg/day within 6 months (rapid GC reducers) were compared with those who did not (conventional GC reducers) regarding partial renal response (PRR) and complete renal response (CRR) at 12 months. PRR and CRR was defined based on the BLISS-LN clinical trial (N Engl J Med 2020; 383: 1117). Modified Poisson regression analysis was used to adjust for confounding factors.
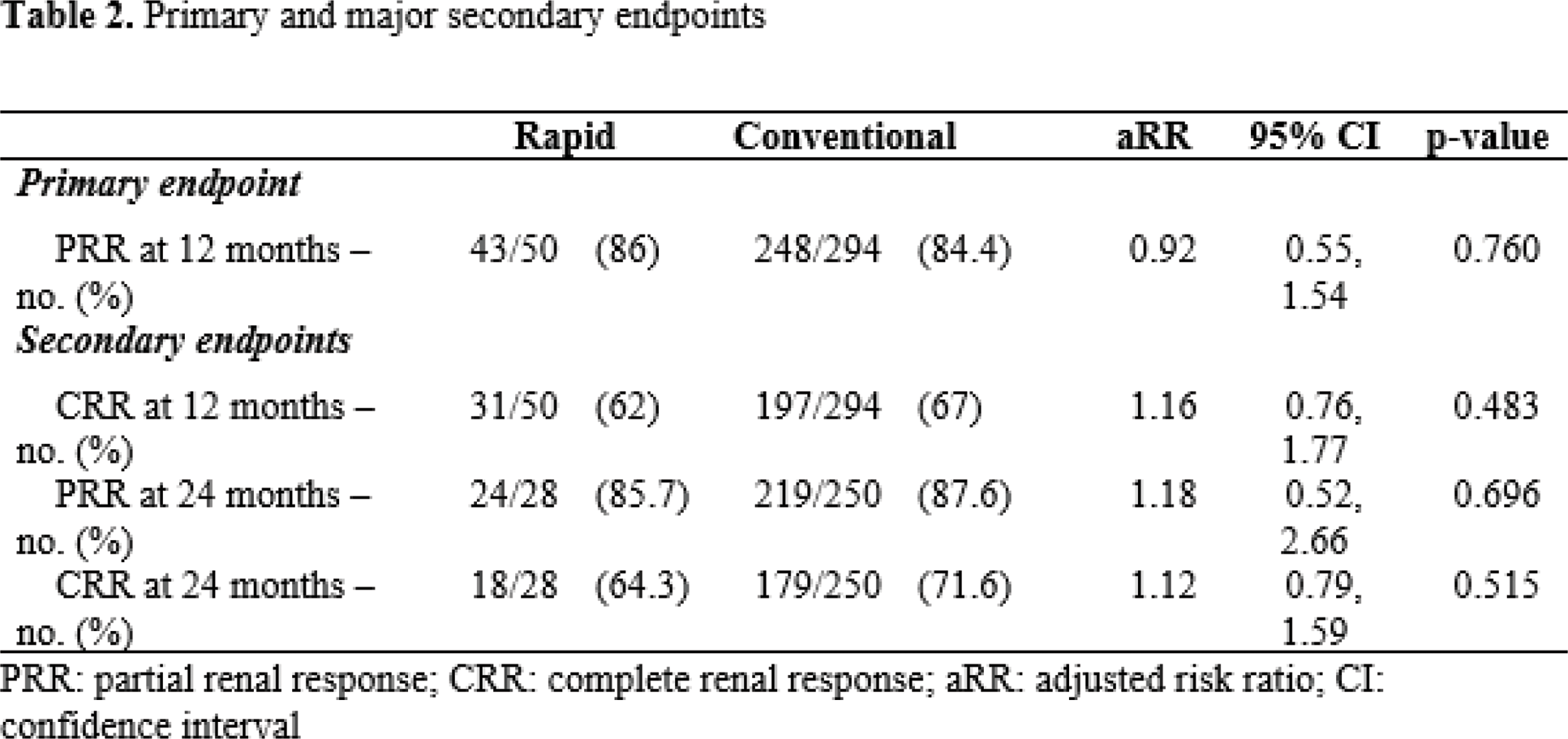
Results: A total of 344 patients from 17 centres were included: 50 rapid GC reducers and 294 conventional GC reducers. PRR at 12 months was achieved by 43/50 (86%) in the rapid GC group and 248/294 (84.4%) in the conventional group. After adjusting for age, initial UPCR, initial estimated glomerular filtration rate, initial GC dose, use of methylprednisolone pulse therapy, strong immunosuppressants (mycophenolate mofetil, cyclophosphamide, or rituximab), and hydroxychloroquine, no significant difference was observed in PRR and CRR at 12 months (p = 0.760, 0.483, respectively). Recurrence rates and serious adverse events over 2 years of follow-up were also comparable between the groups.
Conclusion: Rapid GC reduction to ≤ 7.5 mg/day within 6 months did not compromise renal outcomes or increase recurrence in proliferative LN.
REFERENCES: NIL.
Acknowledgements: We appreciate all the members of the Research Committee of Systemic Lupus Erythematosus and Primary Anti-Phospholipid Syndrome in the Ministry of Science and Welfare, and of the Committee of Guideline for the management of Systemic Lupus Erythematosus in Japan College of Rheumatology for their valuable advice.
Disclosure of Interests: Koichiro Ohmura AbbVie Japan GK, Asahi KASEI Pharma Co., Ltd., Astellas Pharma Inc., AstraZeneca plc., Ayumi, Boehringer Ingelheim Japan, Inc., Bristol Myers Squibb Co., Ltd., Chugai Pharmaceutical Co., Ltd., Eisai Co., Ltd., Eli Lilly and Company, Gilead Sciences, Inc., GlaxoSmithKline K.K., Mitsubishi-Tanabe Pharma Co., Ltd., Novartis Pharma K.K., Otsuka Pharmaceutical Co., Ltd., Taisho Pharmaceutical Co., Ltd., Janssen Pharmaceutical K.K., UCB Japan Co. Ltd., AbbVie Japan GK, Asahi KASEI Pharma Co., Ltd., Chugai Pharmaceutical Co., Ltd., Mitsubishi-Tanabe Pharma Co., Ltd., Hayato Shimizu: None declared, Yoshiya Tanaka: None declared, Keiju Hiromura Astellas Pharma, AstraZeneca, Asahi Kasei Pharma, GlaxoSmithKline, Kissei Pharmaceutical, Otsuka Pharmaceutical, Otsuka Pharmaceutical, GlaxoSmithKline, Chugai Pharmaceutical, Hiroki Hayashi Asahi Kasei Pharma Co., AstraZeneca plc., Boehringer Ingelheim Japan, Inc., GlaxoSmithKline, Otsuka Pharmaceutical Co., Kazuro Kamada: None declared, Kentaro Minowa: None declared, Yutaka Kawahito Asahi Kasei Pharma Co., Ltd., AbbVie Japan GK, Astellas Pharma Inc., AstraZeneca plc., Ayumi Pharmaceutical Co., Boehringer Ingelheim Japan, Inc., Bristol Myers Squibb Co., Ltd., Chugai Pharmaceutical Co., Ltd., Eisai Co., Ltd., Daiichi Sankyo., GlaxoSmithKline K.K., Janssen Pharmaceutical K.K., Kissei Pharmaceutical Co., Ltd., Mitsubishi-Tanabe Pharma Co., Nippon Kayaku Co., Ltd., Novartis Pharma K.K., Pfizer Japan Inc., Taisho Toyama Pharmaceutical Co., Ltd., UCB Japan Co. Ltd. and Viatris Pharmaceuticals Japan Inc., Asahi KASEI Pharma Co., Ltd., AbbVie Japan GK, Boehringer Ingelheim Japan, Inc., Chugai Pharmaceutical Co., and Eisai Co., Ltd., Akiho Iwashita: None declared, Shinya Kaname: None declared, Shinsuke Yasuda: None declared, Kazuoto Hiramoto: None declared, Shoichi Maruyama: None declared, Nobuyuki Yajima: None declared, Yasunori Iwata: None declared, Isao Matsumoto: None declared, Takahisa Gono: None declared, Hiroko Sato: None declared, Satoshi Kubo: None declared, Shingo Nakayamada: None declared, Hidekazu Ikeuchi: None declared, Yukio Yuzawa: None declared, Michihito Kono AbbVie, Actelion, Asahi-Kasei, Astellas, AstraZeneca, AYUMI, Bristol-Myers Squibb, Chugai, Daiichi Sankyo, Eisai, Eli Lilly, Gilead Sciences, GlaxoSmithKline, Janssen, Kowa, KYOCERA, LOTTE, Nippon Boehringer Ingelheim, NIPPON SHINYAKU, Mitsubishi Tanabe, MOCHIDA, Otsuka, Pfizer, Taiju Life Social Welfare Foundation, Taisho, Takeda, Terumo, UCB Japan and YAMAZAKI BAKING, Naoto Tamura Asahi Kasei Pharma, AstraZeneca, AbbVie, Eli Lilly Japan, GlaxoSmithKline, Chugai, Novartis, Bristol Myers Squibb, Janssen, Asahi Kasei Pharma, Asahi Kasei Medical, Ayumi, AbbVie, Eisai, Nippon Boehringer Ingelheim, Taisho, Tanabe Mitsubishi, Chugai, Takahiro Seno Asahi Kasei Pharma Co., Ltd., AbbVie Japan GK, Astellas Pharma Inc., AstraZeneca plc., Boehringer Ingelheim Japan, Inc., Bristol Myers Squibb Co., Ltd., Chugai Pharmaceutical Co., Ltd., Eisai Co., Ltd., Eli Lilly and Company, GlaxoSmithKline K.K., Janssen Pharmaceutical K.K., Kissei Pharmaceutical Co., Ltd., Kyorin Pharmaceutical Co., Ltd., Mitsubishi-Tanabe Pharma Co., Novartis Pharma K.K., Pfizer Japan Inc., Taisho Toyama Pharmaceutical Co., Ltd., and UCB Japan Co. Ltd., Takashi Kida: None declared, Aki Sakashita: None declared, Akira Onishi: None declared, Akio Morinobu: None declared, Takahisa Kawakami: None declared, Tadashi Hosoya: None declared, Taiki Yamaguchi: None declared, Yuko Kaneko AbbVie Japan GK, Asahi KASEI Pharma Co., Ltd., Astellas Pharma Inc., AstraZeneca plc., Ayumi, Boehringer Ingelheim Japan, Inc., Bristol Myers Squibb Co., Ltd., Chugai Pharmaceutical Co., Ltd., Eisai Co., Ltd., Eli Lilly and Company, Gilead Sciences, Inc., GlaxoSmithKline K.K., Mitsubishi-Tanabe Pharma Co., Ltd., Novartis Pharma K.K., Otsuka Pharmaceutical Co., Ltd., Pfizer Japan Inc., Taisho Pharmaceutical Co., Ltd., Janssen Pharmaceutical K.K., and UCB Japan Co. Ltd., Asahi KASEI Pharma Co., Ltd., Boehringer Ingelheim Japan, Inc., Chugai Pharmaceutical Co., Ltd., Eisai Co., Ltd., Mitsubishi-Tanabe Pharma Co.,Ltd, and Pfizer Japan Inc, Hironari Hanaoka: None declared, Sawako Kato: None declared, Kayaho Maeda: None declared, Shiori Nakagawa: None declared, Yuya Kondo: None declared, Masataka Kuwana: None declared, Tomonori Ishii: None declared, Shuji Sumitomo: None declared, Chisato Miyakoshi: None declared, Tatsuya Atsumi Chugai Pharmaceutical Co., Ltd. and Nippon Shinyaku Co. Ltd. Report Janssen Pharmaceutical K.K., Mitsubishi Tanabe Pharma Co., Daiichi Sankyo Co. Ltd., Pfizer Inc., Alexion Inc., Novartis Pharma K.K., Eli Lilly Japan K.K., Kyowa Kirin Co., Ltd., AbbVie Inc., Nippon Boehringer Ingelheim Co., Ltd, Amgen Inc., UCB Japan Co. Ltd., AstraZeneca plc., Asahi Kasei Pharma Co. Ltd., Eisai Co., Ltd., Takeda Pharmaceutical Co., Ltd., Astellas Pharma Inc., Sanofi K.K., AstraZeneca plc, Idorsia Pharmaceuticals Ltd, Otsuka Pharmaceutical Co., Gilead Sciences, Inc., GlaxoSmithKline plc., Sanofi K.K., Eli Lilly Japan K.K., Nippon Boehringer Ingelheim Co., Ltd., Janssen Pharmaceutical K.K., UCB Japan Co. Ltd., KISSEI PHARMACEUTICAL CO., LTD., Takeda Pharmaceutical Co., Ltd., Astellas Pharma Inc., Mitsubishi Asahi Kasei Pharma Co. Ltd., Otsuka Pharmaceutical Co., Nippon Boehringer Ingelheim Co., Ltd., TEIJIN PHARMA LIMITED., Eisai Co., Ltd., Eli Lilly Japan K.K., Alexion Inc., Mitsubishi Tanabe Pharma Co., Chugai Pharmaceutical Co., Ltd.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (