

Background: Tirzepatide is a dual gastric inhibitory peptide (GIP)/glucagon-like peptide 1 (GLP-1) receptor co-agonist that is currently approved in the United States (U.S.) and European Union for weight loss among patients with obesity or those with body mass index (BMI) ≥ 27 kg/m 2 with weight-related comorbidities. It has been hypothesized that GLP-1-based therapies may improve osteoarthritis symptoms indirectly through weight loss resulting in reduced mechanical stress to joints as well as directly through anti-inflammatory mechanisms [1, 2, 3]. The STEP 9 clinical trial recently demonstrated that patients with obesity and knee osteoarthritis who received semaglutide treatment experienced improvement in knee pain and function, as well as reduced use of non-steroidal anti-inflammatory drugs (NSAIDs) [4]. Tirzepatide notably differs from semaglutide in its dual agonism and comparatively increased weight loss [5]. There is currently no data on musculoskeletal pain outcomes or risky analgesic use for patients receiving tirzepatide.
Objectives: The aim of the present study was to examine whether patients who initiated treatment with tirzepatide had improved joint pain and required less analgesic use than those receiving phentermine (the most commonly prescribed anti-obesity medication in the U.S.) thereafter.
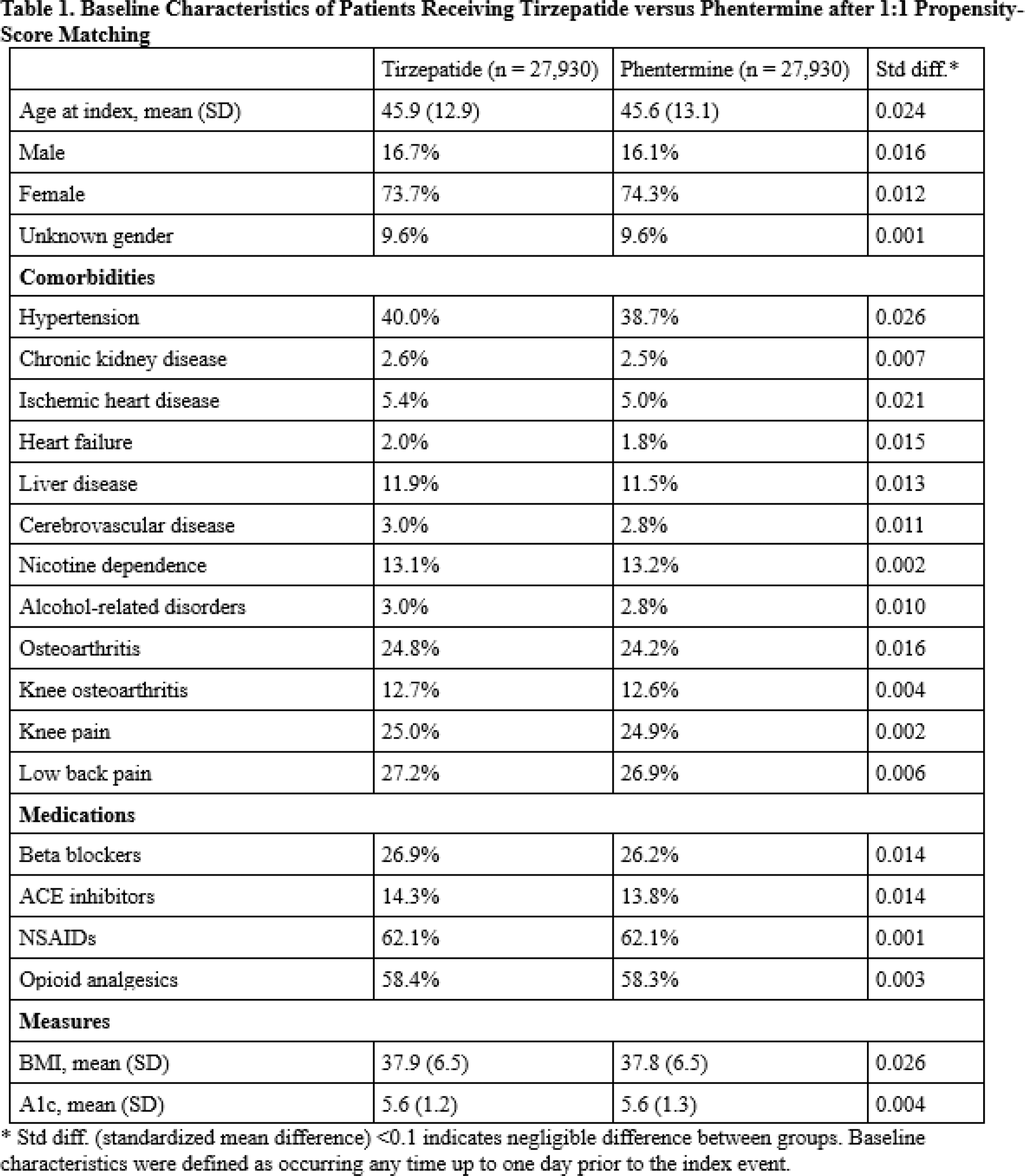
Methods: We conducted a population-based cohort study using the TriNetX Research network, a large, multicenter network of U.S. electronic health records and claims data. International Classification of Diseases, Tenth Revision (ICD-10) codes identified non-diabetic patients with BMI ≥ 27 kg/m 2 . Using the intention-to-treat method, we analyzed the overall rate of prespecified musculoskeletal pain encounters and risky analgesic use (i.e., NSAIDs and opioids), as well as cardiovascular endpoints following initiation of therapy with tirzepatide or phentermine, excluding patients with prior history of the outcomes of interest. Outcomes were defined as ≥ 1 encounter with applicable ICD-10 code occurring one day to any time after the index event. Nausea served as a positive control outcome. Propensity score matching included over 400 prognostic factors to adjust for demographics, comorbidities, medications, family history, surgical history, and baseline BMI; representative covariates are included in Table 1. Kaplan-Meier analysis and Cox proportional hazards models estimated the risk of the outcomes of interest.
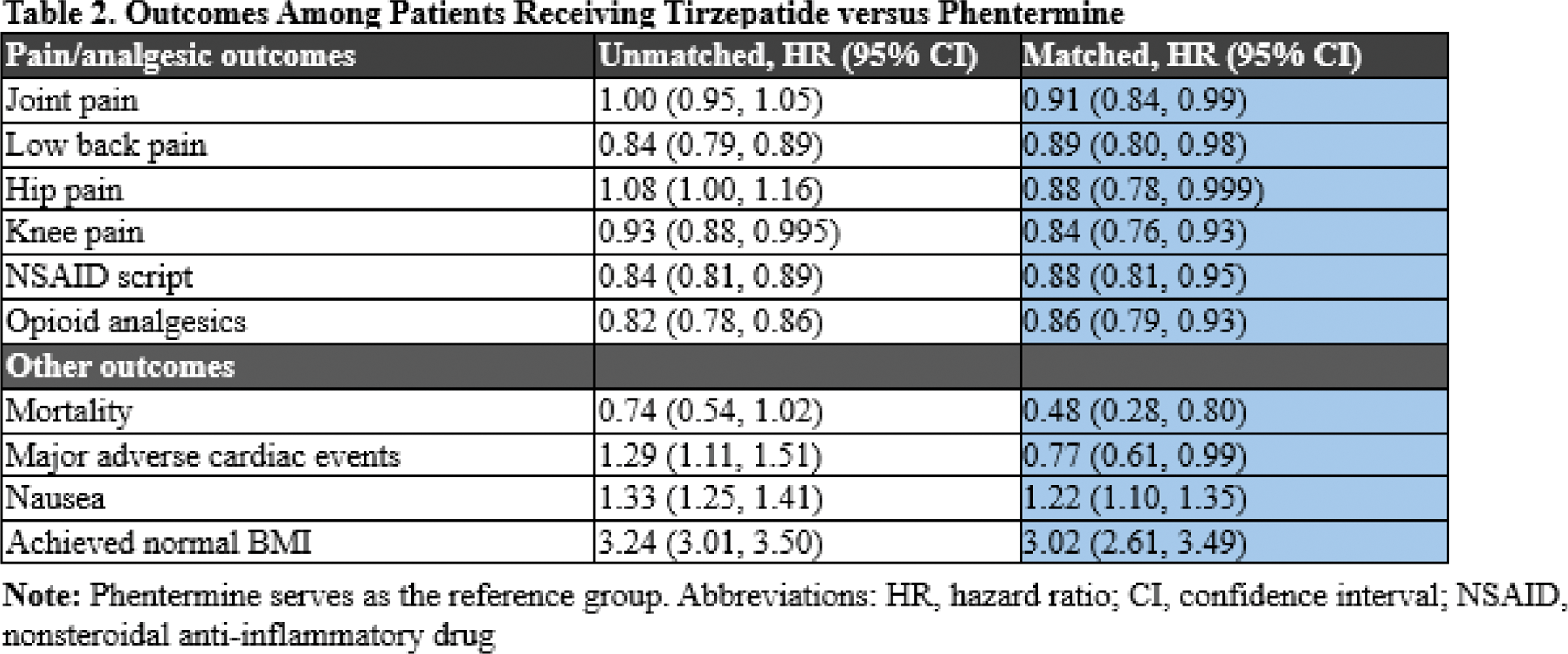
Results: A total of 62,722 patients started on tirzepatide and 119,020 patients started on phentermine were included. After 1:1 propensity score matching, 27,930 patients in each group were comparable with well-balanced baseline characteristics (Table 1; all standardized mean differences < 0.1). Mean follow-up time was 276 days (with standard deviation of 278 days) among patients on tirzepatide compared with mean follow-up time of 318 days (with standard deviation of 296 days) among patients on phentermine. Patients receiving tirzepatide were significantly more likely to achieve a normal BMI level than those receiving phentermine, with hazard ratio (HR) of 3.02 (95% CI: 2.61, 3.49). Initiation of tirzepatide was associated with overall lower risk of joint pain with HR 0.91 (95% CI: 0.84, 0.99), including low back pain (HR 0.89 with 95% CI: 0.80, 0.98), hip pain (HR 0.88 with 95% CI: 0.78, 0.999), and knee pain (HR 0.84 with 95% CI: 0.76, 0.93). Tirzepatide was also associated with a lower risk of NSAID (HR 0.88 with 95% CI: 0.81, 0.95) and opioid prescriptions (HR 0.86 with 95% CI: 0.79, 0.93). As expected, the risk of nausea was increased among those receiving tirzepatide (HR 1.22 with 95% CI: 1.10, 1.35), while risks of mortality and overall major adverse cardiac events (acute myocardial infarction, cerebral infarction, and/or mortality) were reduced (respectively, HR 0.48 with 95% CI of 0.28, 0.80; and HR 0.77 with 95% CI of 0.61, 0.99).
Conclusion: This large population-based study of non-diabetic patients with overweight or obesity demonstrates improvement in musculoskeletal pain requiring clinical care, and risky analgesic use as well as cardiovascular outcomes, among those receiving tirzepatide in comparison to patients receiving phentermine. This provides further support for the use of tirzepatide in patients with overweight or obesity to reduce musculoskeletal pain and the need for NSAID and opioid medications.
REFERENCES: [1] PMID: 35091584.
[2] PMID: 39476345.
[3] PMID: 35280931.
[4] PMID: 39476339.
[5] PMID: 38976257.
Acknowledgements: NIL.
Disclosure of Interests: Gregory Challener: None declared , Kevin Sheng-Kai Ma: None declared , Natalie McCormick: None declared , Minna Kohler: None declared , Janeth Yinh: None declared , Chio Yokose: None declared , Sharan Rai: None declared , Florence Porterfield: None declared , April Jorge: None declared , Hyon Choi Ani, Protalix, Horizon, LG Chem, Shanton, LG Chem.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (