

Background: Eosinophilic disorders (ED) are a spectrum of rare conditions associated with blood eosinophilia leading to eosinophil-associated organ damage. The mutual interaction between pregnancy and ED have been little investigated to date, still adverse pregnancy outcomes (APOs) have been reported, mainly related to high disease activity and intercurrent treatment. Monoclonal antibodies targeting interleukin (IL)-5 pathway have been approved for the treatment of most ED, including eosinophilic granulomatosis with polyangiitis (EGPA), hypereosinophilic syndrome (HES), eosinophilic asthma (EA) and chronic rhinosinusitis with nasal polyps (CRSwNP). These agents are very effective in both controlling disease activity and sparing glucocorticoids (GC). However, evidence on their use in pregnancy is still anecdotal. At present, only case reports on IL-5 pathway inhibitors (IL-5i) use in ED pregnancies are available, and case series or registries are lacking.
Objectives: To assess the effectiveness and safety profile of IL-5i during pregnancy and describe APOs in patients with eosinophilic disorders.
Methods: This retrospective, observational, multicenter study included adult women with a diagnosis of ED (i.e., EGPA, HES, EA or CRSwNP) treated with an IL-5i (mepolizumab, benralizumab, or reslizumab) during pregnancy. Data were collected from 01/01/2000 until 31/12/2024, covering demographic and clinical information of the women from ED onset until pregnancy outcomes. Descriptive statistics (percentages, medians, etc.) were used to summarize the characteristics of the cohort. Fisher’s exact test and rank sum tests were used for comparisons.
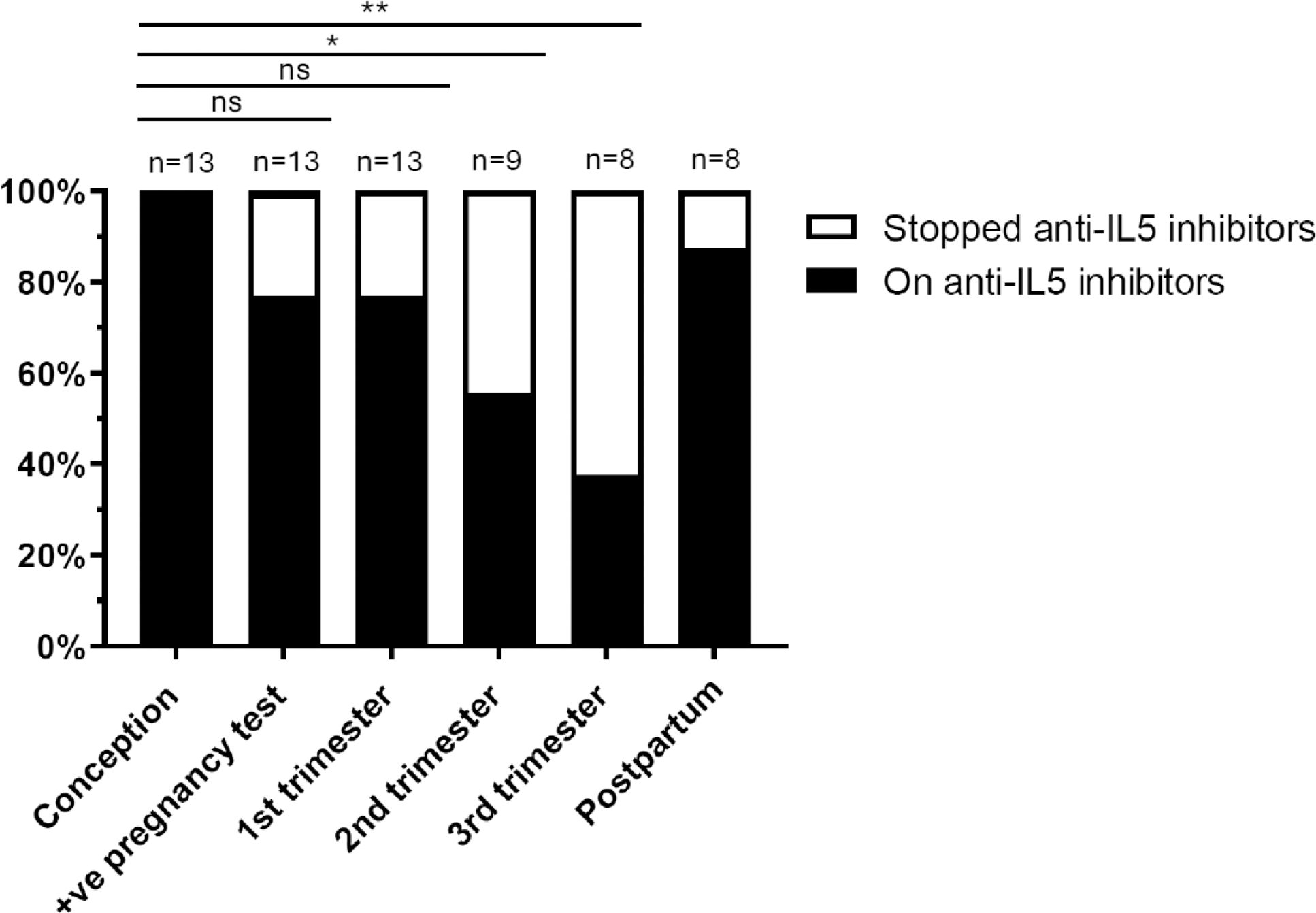
Results: The data of 13 pregnancies (one still ongoing, at 2 nd trimester) from 10 patients were enrolled, 2 affected by EA, 3 by HES and 5 by EGPA (all ANCA-negative, median BVAS at onset 14 (IQR 9, 18.5)). The median age at ED diagnosis was 28 (IQR: 26, 28.75) years old. Before the pregnancies under IL-5i exposure, all women had at least one pregnancy (median 2 (IQR: 1, 3.25)) and one delivery (median 1 (IQR: 1, 1.25)), with 40% of these with previous pregnancy complications. None of them had significant comorbidities, other systemic autoimmune disorder, or antiphospholipid syndrome, while one was an asymptomatic Lupus Anticoagulant (LAC) carrier. Of the 13 pregnancies, all women were on IL-5i at conception (11 in mepolizumab with 54% at 300mg/month; 2 in benralizumab at 30mg/month), 3 of them stopped mepolizumab at positive pregnancy test. At conception, 3 women (23%) were on GC (median dose 5 mg/day (IQR: 0, 12.5)), whereas none were on additional immunosuppressive agents. In 31% of the pregnancies, the ED was deemed as active by clinicians at first assessment. In 15% of the pregnancies, asthma was uncontrolled in the month before positive pregnancy test. No relapse of the ED during pregnancy was observed, nor GC was increased or started during the pregnancy course to control ED. Among the completed pregnancies, in 43% (3/7) of cases, ED were deemed as active in postpartum (1 HES, 2 EGPA), with 2 patients with EGPA being active from the beginning. Of note, in all these patients IL-5i were discontinued during pregnancy (two in 1 st trimester, one in 2 nd trimester). Adverse pregnancy outcomes (APOs) occurred in 38% (5/13) of pregnancies: pregnancy loss in 38% (4 pregnancy losses in 3 women before 10 th gestational week (GW), and 1 therapeutic abortion at 18 th GW for fetal malformation), maternal complications in 14% (gestational diabetes). No neonatal deaths, preterm deliveries, and small-for-gestational-age neonates were reported. In all cases, APOs were not considered as drug-related (physician’s judgment). Of note, among the 3 patients with early pregnancy loss, one was LAC carrier and 2 had a healthy pregnancy under IL-5i. In the woman with therapeutic abortion, the same fetal malformation was already reported in a previous pregnancy before HES diagnosis. When pregnancy outcomes were stratified by eosinophilic disorder type and disease activity (active vs non active), use of GC, maternal age, IL-5i type at conception, no difference was observed (p>0.05 in all the comparisons). Compared to the conception timepoint, 77% (10/13) of patients were still on IL-5i at the time of the positive pregnancy test and during the 1st trimester, 56% (5/9) during the 2 nd trimester (p=0.0172), and 38% (3/8) during the 3 rd trimester (p=0.0028). The reason for discontinuation of IL-5i was for concerns about lack of data in pregnancy. After delivery, 87.5% were on treatment (4 patients started again the IL-5i) (Figure 1).
Conclusion: ED may represent a challenging condition in pregnancy, as uncontrolled disease and high GC use could seriously affect maternal and fetal outcomes. In our retrospective study, IL-5i have proven to be an effective treatment in pregnancy of ED patients, ensuring both persistent disease control and steroid sparing. No adverse events directly attributable to IL-5i treatment have been reported.
REFERENCES: [1] Middeton Eur Respir J 2020;55:1901208.
[2] Davanzo et al. Front Lupus 2:1479884.
Acknowledgements: NIL.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (