

Background: Vunakizumab is a novel, humanized monoclonal IgG1/k antibody targeting IL-17A. In a prior adaptive, seamless, phase 2/3 trial, vunakizumab demonstrated efficacy and tolerability in treating patients with active ankylosing spondylitis (AS) [1].
Objectives: This analysis aimed to evaluate the impact of prior tumor necrosis factor-α inhibitor (TNFi) exposure on the efficacy and safety of vunakizumab in patients with active AS.
Methods: This post-hoc analysis used data from a randomized, double-blind, adaptive, seamless, phase 2/3 trial (NCT04840485) of vunakizumab (SHR-1314) in active AS. Eligible patients had radiographic evidence of active AS meeting the modified New York criteria, had an inadequate response, intolerance or contraindication to non-steroidal anti-inflammatory drugs. Stratification factors included prior TNFi use (yes or no) and weight (≥90 kg or <90 kg). In the phase 2 part, patients were randomized (2:2:1) to receive subcutaneous injections of vunakizumab 120 mg, vunakizumab 240 mg, or placebo at weeks 0, 2, 4, 8, and 12. Based on interim analysis at week 16, the independent data monitoring committee recommended vunakizumab 120 mg as the optimal dose for the phase 3 part. In the phase 3 part, patients were randomized (2:1) to receive vunakizumab 120 mg or placebo. In both phase 2 and phase 3 parts, at week 16, patients initially receiving placebo transitioned to receive vunakizumab every 4 weeks until week 32, while those initially receiving vunakizumab continued their initial treatment. The primary endpoint was the percentage of patients with a 20% response according to the Assessment of SpondyloArthritis international Society (ASAS20) at week 16. In this analysis, baseline, efficacy and safety data were pooled for patients who received vunakizumab 120 mg or placebo in the phase 2 and phase 3 parts. Subgroup analyses were performed based on prior TNFi exposure status (yes or no). Missing data were imputed as nonresponses. Logistic regression models were used to adjust for stratification factor (weight), subgroups (prior TNFi exposure), and treatment-subgroup interactions at week 2, 4, 8, 12, 16.
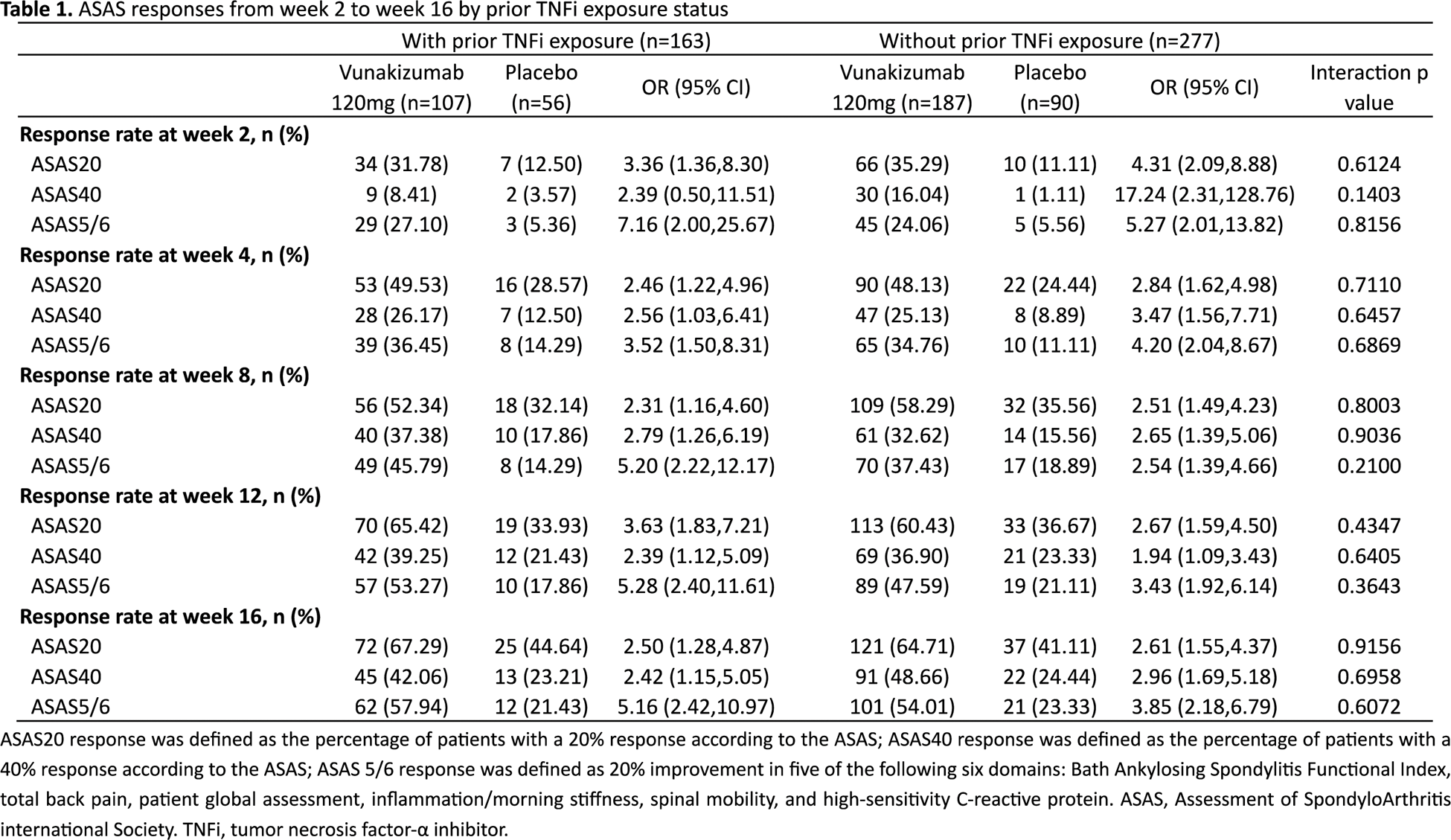
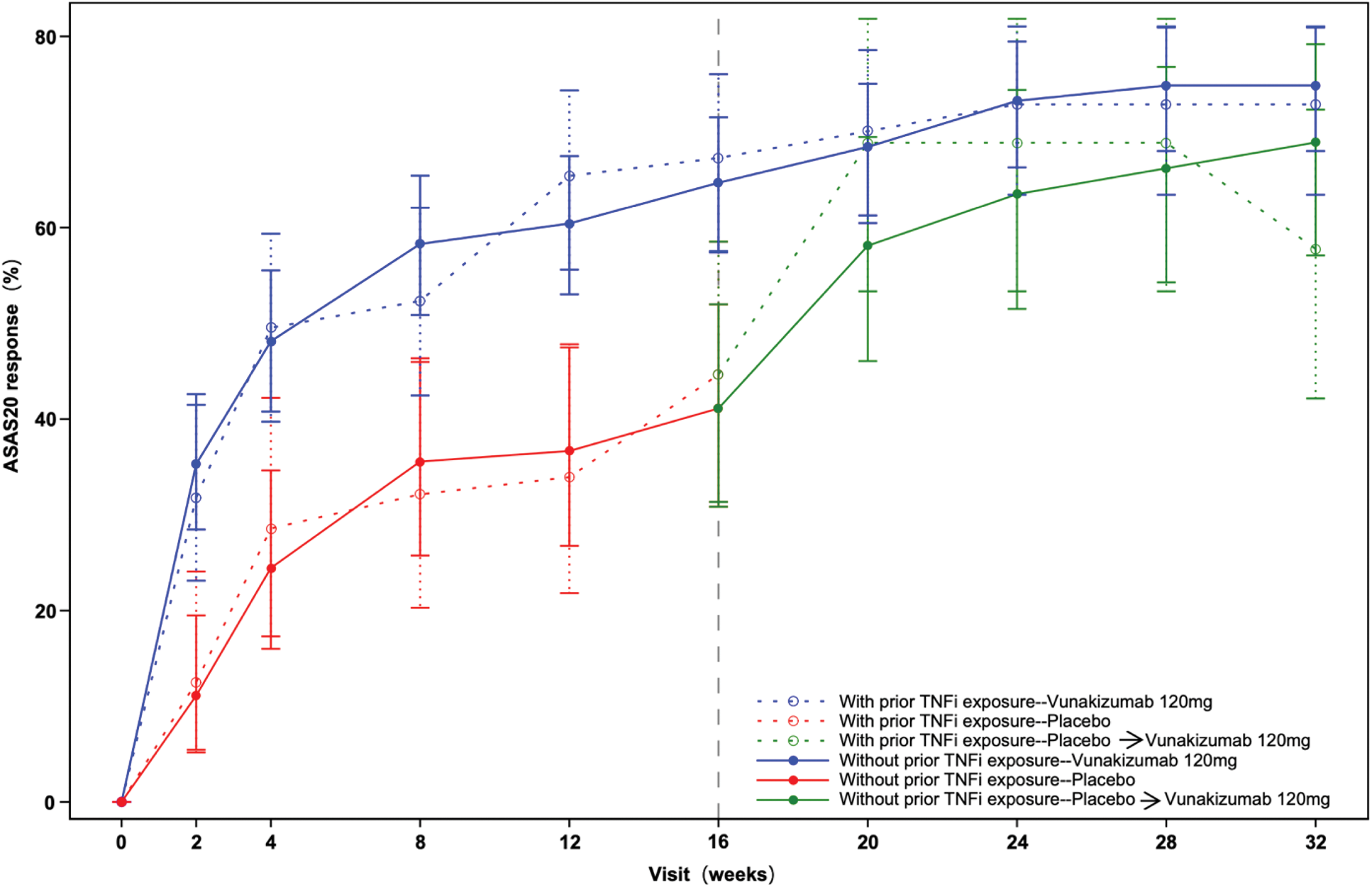
Results: A total of 440 patients were included in this analysis. Among these patients, 163 had prior TNFi exposure (vunakizumab, n=107; placebo, n=56) and 277 had no prior TNFi exposure (vunakizumab, n=187; placebo, n=90). Baseline demographic and clinical characteristics were similar between treatment arms within each TNFi exposure status subgroup. The ASAS20, ASAS40 and ASAS 5/6 responses from week 2 to week 16 are shown in Table 1. At week 16, in patients with prior TNFi exposure, the ASAS20 response rates were 67.29% for vunakizumab and 44.64% for placebo (OR: 2.50, 95% confidence interval [CI]: 1.28-4.87); in patients without prior TNFi exposure, the ASAS20 response rates were 64.71% for vunakizumab and 41.11% for placebo (OR: 2.61, 95% CI: 1.55-4.37). At week 16, in patients with prior TNFi exposure, the ASAS40 response rates were 42.06% and 23.21% for vunakizumab and placebo (OR: 2.42, 95%CI: 1.15-5.05), respectively; in patients without prior TNFi exposure, the ASAS40 response rates were 48.66% and 24.44% for vunakizumab and placebo (OR: 2.96, 95%CI: 1.69-5.18), respectively. No statistically significant interactions between prior TNFi exposure status and treatment effect were observed for ASAS20, ASAS40 and ASAS 5/6 responses from week 2 to week 16 (Table 1). In addition, the ASAS20 responses were sustained through week 32 in both TNFi exposure status subgroups (Figure 1). Safety profiles were similar across treatment arms within each TNFi exposure status subgroup. The treatment-emergent adverse events and adverse events of special interest were mainly mild to moderate during the 16-week treatment period. No opportunistic infections, serious infections, malignancies, or deaths were reported during the 32-week treatment period in either subgroup.
Conclusion: Vunakizumab showed efficacy and acceptable safety in patients with active AS, regardless of prior TNFi exposure. These findings warrant validation in future studies.
REFERENCES: [1] Huang, F. et al. POS0803 VUNAKIZUMAB IN ACTIVE ANKYLOSING SPONDYLITIS: A RANDOMIZED, DOUBLE-BLIND, ADAPTIVE, SEAMLESS, PHASE 2/3 STUDY. Annals of the Rheumatic Diseases 83 , 904-905 (2024).
Acknowledgements: NIL.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (