

Background: Ivarmacitinib (SHR0302), a selective Janus kinase 1 inhibitor, has demonstrated substantial improvements in patients with active ankylosing spondylitis (AS).
Objectives: This post-hoc analysis evaluated the effects of Ivarmacitinib on various dimensions of patient-reported outcomes (PROs) in active AS patients, utilizing data from a phase II/III clinical trial (NCT04481139).
Methods: Patients received either Ivarmacitinib 4 mg (n=187) or placebo (n=186) daily for 12 weeks. Patients with placebo switched to Ivarmacitinib thereafter until week 24. PROs included total back pain and night pain by visual analog scale (VAS), morning stiffness, Patient Global Assessment of Disease Activity (PtGA), and AS Quality of Life (ASQoL).
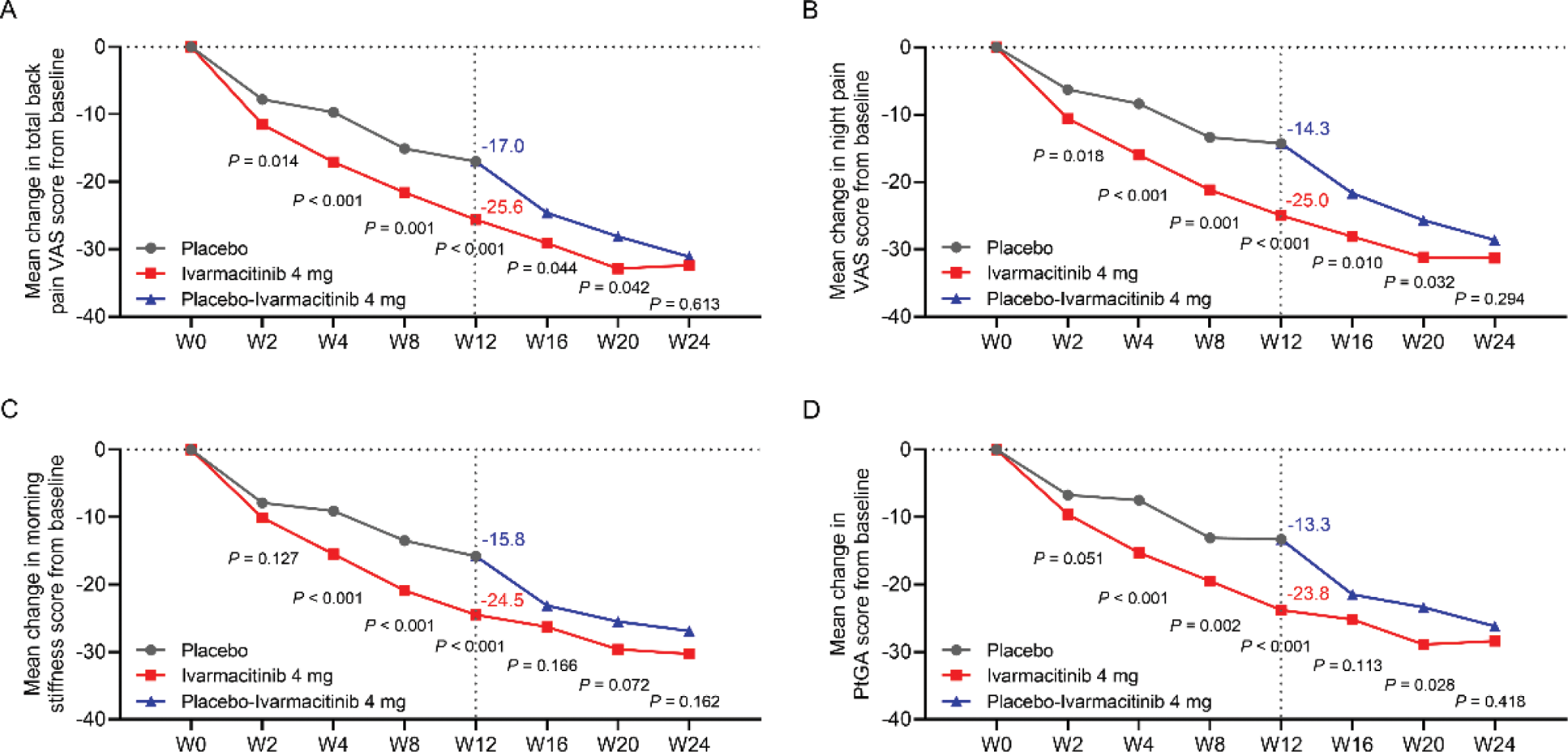
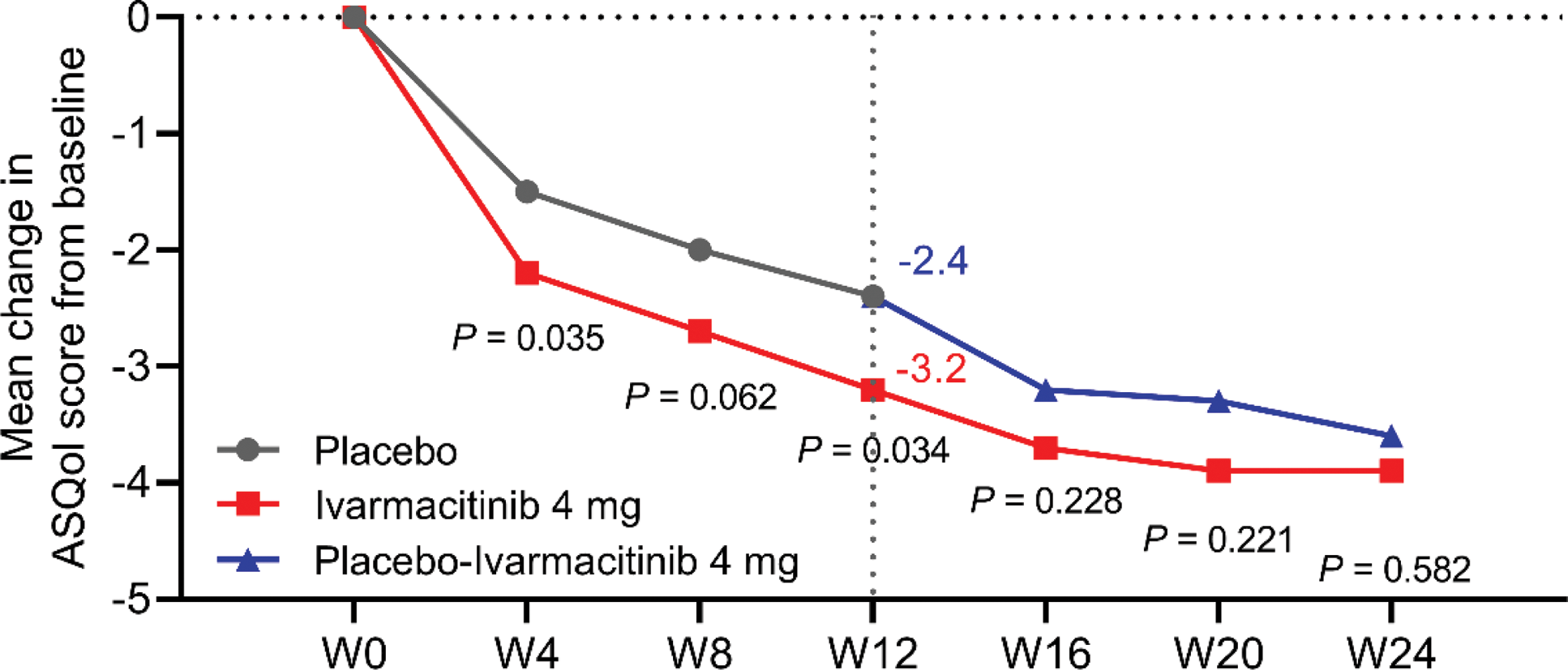
Results: Ivarmacitinib 4 mg group showed significantly improvement in total back pain VAS (-25.6 vs. -17.0, P<0.001), night pain VAS (-25.0 vs. -14.3, P<0.001), morning stiffness (-24.5 vs. -15.8, P<0.001), PtGA (-23.8 vs. -13.3, P<0.001), and ASQoL (-3.2 vs. -2.4, P=0.034) scores compared to placebo after 12 weeks of treatment. By week 24, patients who switched from placebo to Ivarmacitinib 4 mg achieved comparable PROs improvements to those who initially received Ivarmacitinib from baseline (Figures 1 and 2).
Conclusion: Ivarmacitinib significantly enhances multiple dimensions of PROs in active AS patients, supporting its utility in managing PROs in AS. Switching to Ivarmacitinib also provides substantial benefits, ensuring that patients who delay initiation of Ivarmacitinib also experience meaningful improvements in PROs.
REFERENCES: NIL.
Comparison of the changes in total back pain VAS score (A), night pain VAS score (B), morning stiffness score (C), and PtGA score (D) from baseline between groups.
Comparison of the changes in ASQoL score from baseline between groups.
Acknowledgements: NIL.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (