

Background: Pulmonary manifestations such as interstitial lung disease (ILD) are a major cause of morbidity and mortality in patients with dermatomyositis (DM) [1, 2]. The ProDERM study was a randomized Phase 3 study designed to evaluate the efficacy and safety of intravenous immunoglobulin (IVIg) in adult DM patients [3, 4].
Objectives: Data presented here were obtained through a post-hoc analysis of the ProDERM study data to assess the effect of IVIg on pulmonary symptoms of DM.
Methods: ProDERM was a placebo-controlled, double-blind, multicenter Phase 3 study involving two phases. In the first phase, from Weeks 0 to 16, patients were randomized to receive either 2.0 g/kg IVIg or placebo every 4 weeks. This was followed by an open-label extension period from Weeks 16 to 40, during which all eligible patients (n=91) received 2.0 g/kg IVIg for six additional cycles. Participating investigators assessed the pulmonary symptoms of DM at baseline and all visits during the trial by using the Myositis Disease Activity Assessment Tool (MDAAT). Visual analog scales (VAS) were scored over 10 cm to quantify overall pulmonary disease activity, with a higher score indicating worse disease. Physicians also reported presence/absence of pulmonary symptoms of dysphonia, dyspnea or cough from active reversible ILD, and dyspnea from respiratory muscle weakness on MDAAT.
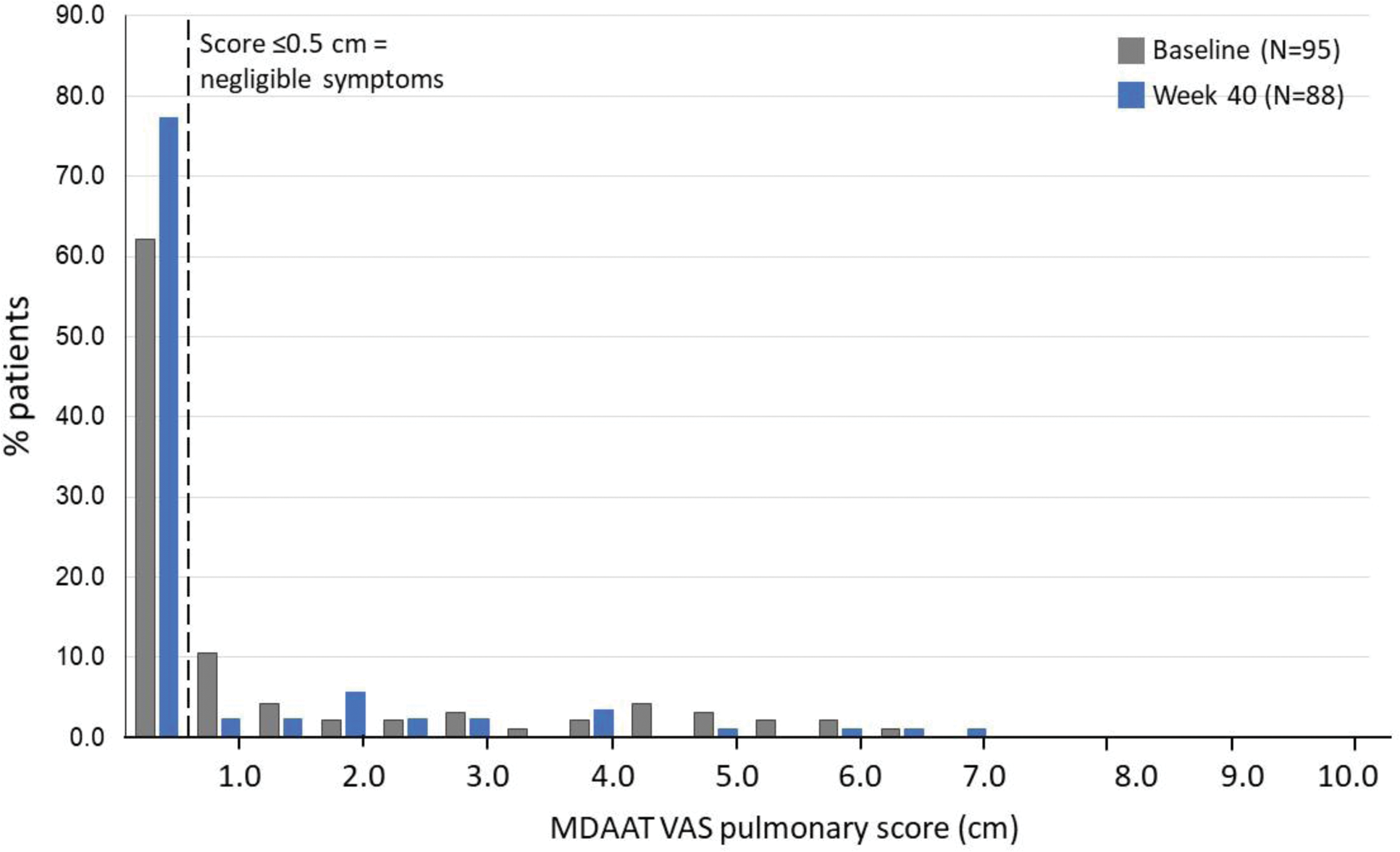
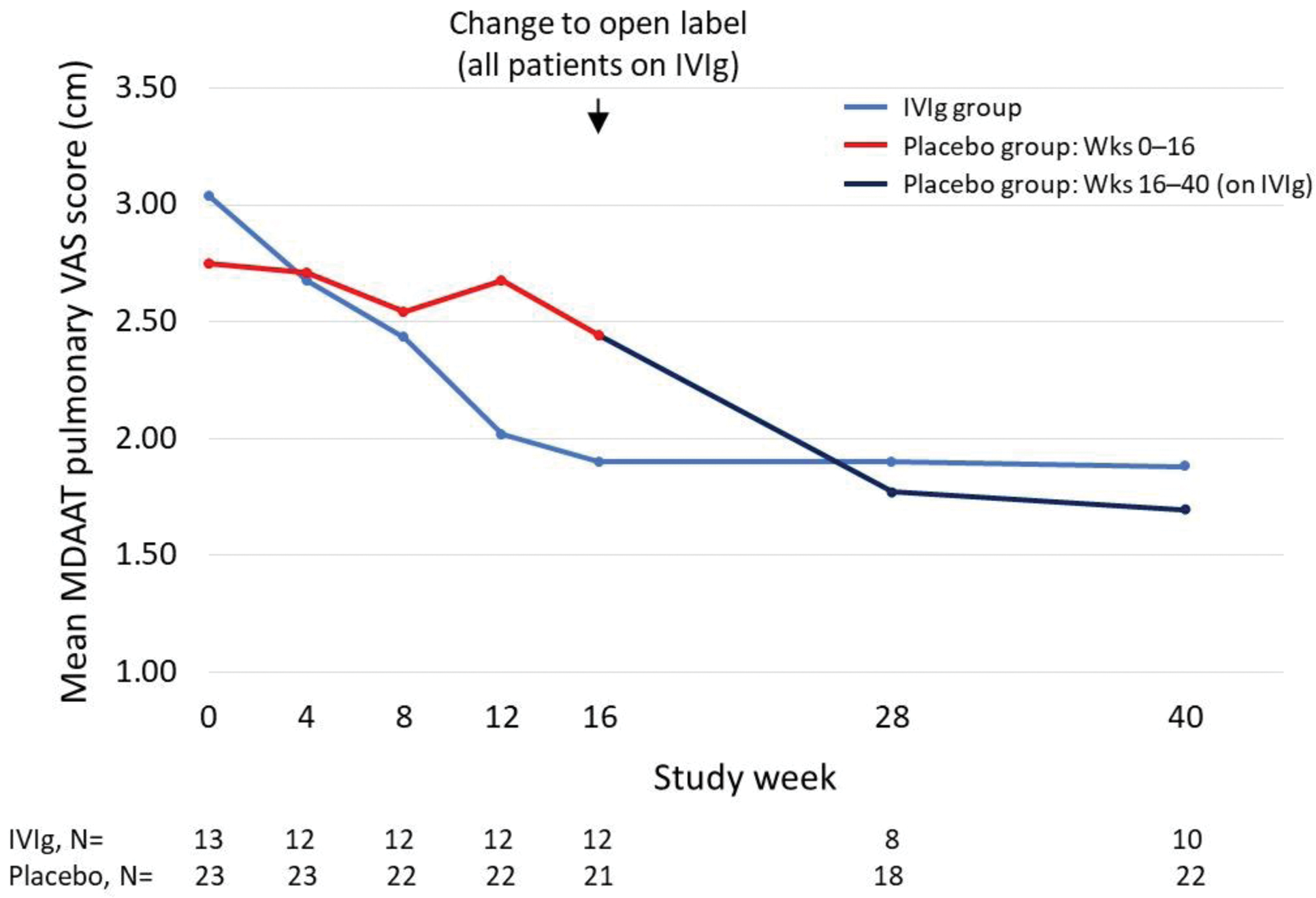
Results: Of 95 DM patients enrolled in the ProDERM study, 98% met the probable or definite criteria for inflammatory myopathy according to the 2017 European League Against Rheumatism (EULAR)/American College of Rheumatology (ACR) criteria for idiopathic inflammatory myopathies. At baseline 59/95 patients (62.1%) had a pulmonary VAS ≤0.5 cm (Figure 1), indicating no myositis lung involvement. By Week 40, the number of patients with a VAS ≤0.5 cm increased to 68 of the 88 patients with available data (77.3%; p=0.007). Patients with baseline pulmonary VAS ≤0.5 cm were unlikely to improve in terms of pulmonary manifestations during the trial, thus improvement in pulmonary score was only evaluated in patients with a baseline VAS >0.5 cm (n=36) (Figure 2). For patients with data at both baseline and Week 16, those on IVIg (n=12) experienced a mean decrease of 1.15 cm (37.7%; p=0.001) vs. 0.17 cm (6.5%) in patients on placebo (n=21; p=0.50). From baseline to Week 40, with patients on placebo having switched to IVIg at week 16, the mean decrease for the combined cohort (n=32 patients with data) was 1.08 cm (38.2%; p<0.001). The percentage of all patients with dyspnea from either respiratory muscle weakness, or from ILD, decreased numerically from baseline to Weeks 16 and 40, but the decrease in neither group reached statistical significance (Table 1). The percentage of patients with dysphonia decreased from 20% at baseline to 8% at Week 40 (p=0.04).
Conclusion: IVIg may have favorable treatment effects on active pulmonary manifestations of DM. Future studies should consider including pulmonary outcomes to assess the effect of treatment on these symptoms.
REFERENCES: [1] Marie I, Hachulla E, Chérin P, et al. Arthritis Rheum 2002;47:614–22.
[2] Hervier B, Uzunhan Y. Front Med (Lausanne) 2019;6:326.
[3] Aggarwal R, Charles-Schoeman C, Schessl J, et al. N Engl J Med 2022;387:1264–78.
[4] Aggarwal R, Schessl J, Charles-Schoeman C, et al. Arthritis Res Ther 2024;26:27.
MDAAT pulmonary VAS score at baseline and Week 40
Change in MDAAT pulmonary VAS scores from baseline*
Acknowledgements: To all patients and investigators.
Disclosure of Interests: Rohit Aggarwal Alexion, ANI Pharmaceutical, Argenx, Artasome, AstraZeneca, Boehringer Ingelheim, Bristol Myers-Squibb, CabalettaBio, Capella, Capstanx, Corbus, CSL Behring, EMD Serono, Galapagos, Horizontal Therapeutics, I-Cell, Immunovant, Janssen, Kezar, Kyverna, Lilly, Manta Medicines, Novartis, Nuvig Therapeutic, Nakarta, Octapharma, Pfizer, Proivant, Teva, Tourmaline Bio and Verismo therapeutics, Boehringer Ingelheim (BI), Bristol-Myers Squibb, EMD Serono, Janssen, Proivant, and Pfizer, Joachim Schessl Pfizer, Octapharma, Elisabeth Clodi Octapharma PPG, Christina Charles-Schoeman Pfizer, Octapharma, AbbVie, Bristol Myers Squibb, Boehringer Ingelheim, Recludix, Galapagos, Sana Biotechnology, Immunovant, Pfizer, Alexion, Priovant, CSL Behring, Janssen, Octapharma, AbbVie, Bristol Myers Squibb.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (