

Background: Eosinophilic granulomatosis with polyangiitis (EGPA) is an antineutrophil cytoplasmic antibodies (ANCA)-associated vasculitis (AAV) characterized by chronic asthma, ear-nose-throat (ENT) symptoms, necrotizing vasculitis, extravascular granulomas, and elevated eosinophil levels in blood and tissues. The pathogenesis involves a Th2-driven immune response, with elevated interleukin-5 (IL-5) promoting eosinophil maturation, activation and survival. A significant proportion of EGPA patients have a history of chronic rhinosinusitis with nasal polyps (CRSwNP) and adult-onset severe eosinophilic asthma.
Objectives: Benralizumab, a monoclonal antibody targeting IL-5 receptor α, has emerged as a promising treatment option for eosinophilic granulomatosis with polyangiitis (EGPA). We aimed to assess long-term effectiveness and safety of benralizumab in patients with relapsing-refractory EGPA.
Methods: This retrospective, single-center, observational study evaluated adult patients diagnosed with severe asthma and relapsing-refractory EGPA followed in a multidisciplinary setting between July 2019 and January 2023 and treated with benralizumab (30 mg/8 weeks), followed for up to 36 months. Primary objective was to assess disease remission (Birmingham Vasculitis Activity Score [BVASv3]=0 and prednisone≤4 mg/day), corticosteroid tapering, lung function, relapse, and discontinuation rates, with secondary endpoints on treatment failure and retention rates. Relapsing-refractory EGPA was defined according to EULAR 2022 recommendations, severe asthma was defined according to the European RespiratorySociety/American Thoracic Society ERS/ATS guidelines and difficult to treat asthma was defined according to ERS/ATS guidelines.
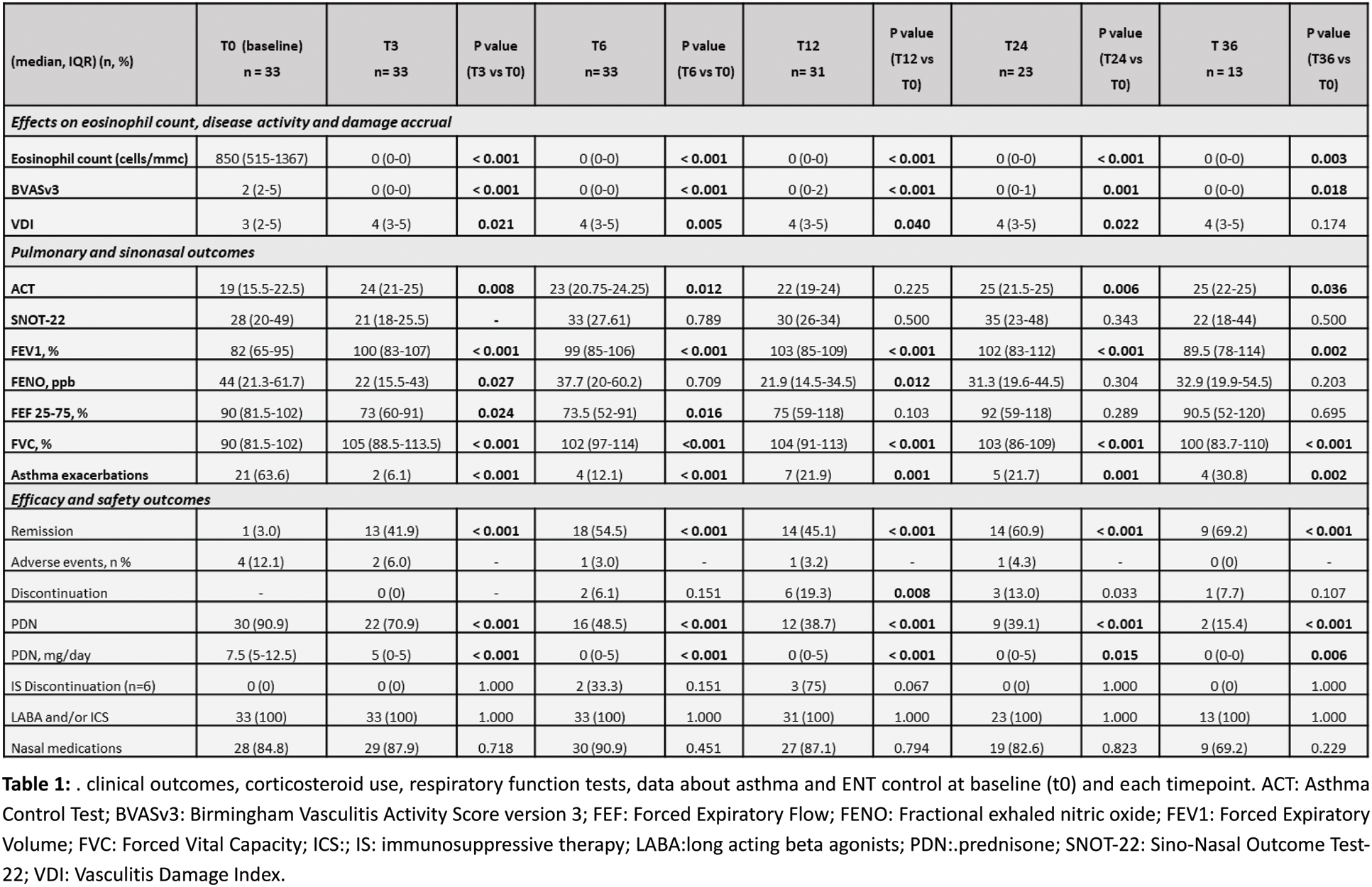
Results: The study included 33 patients with EGPA, 51.5% were male. At the time of diagnosis, a total of 11 (33.3%) patients were positive for ANCA, with anti-MPO specificity, and a median eosinophil count of 1980 cells per µL (1360-4440); At the start of benralizumab median age was 56 years [IQR 47-62], the median disease duration was 31 (16-94) months, the most common manifestations being severe asthma (87.9%) and ENT involvement (66.7%). Benralizumab was started on top of conventional immunosuppressants in 6 (7.8%) of the cases, the most commonly therapies prior to benralizumab were methotrexate (51.5%) and azathioprine (33.3%). Notably 24.2% had previously been treated with mepolizumab (either 300 mg or 100 mg). Benralizumab showed significant effectiveness, with clinical remission rates rising from 39.4% (95%CI 22.9–57.9) at 3 months to 69.2% (95%CI 38.6–90.9) at 36 months (p<0.001 for all time points). Corticosteroid use decreased from 90.9% to 15.4% (p<0.001), eosinophil count dropped from 850(515-1367) to 0(0-0) cells/µL (p<0.001), and BVAS scores declined from 2(2-5) to 0(0-0) (p<0.001). The proportion of patients experiencing asthma exacerbations reduced, alongside improved lung function. The retention rate was 81.8% at 1 year, 72.0% at 2 years, 58.0% at 3 years. The main reason for treatment discontinuation was secondary failure, primarily driven by persistent and uncontrolled ENT/CRSwNP symptoms with 36.4% of patients switch to alternative therapies, primarily mepolizumab (300 mg) or dupilumab. No serious adverse events or hospitalizations occurred. Mild adverse events were observed in 21.2% of patients.
Conclusion: These findings support the long-term effectiveness and safety of benralizumab for EGPA, highlighting its role in inducing clinical remission, reducing corticosteroid dependence and controlling disease activity. The limited response observed in patients with persistent ENT symptoms emphasizes the importance of tailoring biologic selection to individual disease phenotypes.
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: Marta Codirenzi: None declared. Federica Davanzo: None declared. Luca Iorio: None declared. Eleonora Fiorin: None declared. Gabriella Guarnieri: None declared. Alessia Achille: None declared. Fulvia Chieco Bianchi: None declared. Maria Rita Marchi: None declared. Andrea Vianello received research grants from CLS Behring, GSK, and AstraZeneca. Roberto Padoan invited as a speaker or advisory board member by GSK, AstraZeneca, Sanofi and, and CSL Vifor outside the current work, Andrea Doria: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (