

Background: Romosozumab (ROMO) is a dual osteoanabolic agent that stimulates bone formation and reduces bone resorption, emerging as a promising option for treating osteoporosis (OP) in patients at very high fracture risk.
Objectives: This study aims to analyze the safety and efficacy of ROMO treatment in patients with severe OP under real-world clinical conditions.
Methods: This is a single-center, ambispective, and observational study. Patients who initiated ROMO treatment between December 2022 and August 2024 were included, with prospective follow-up until December 2024. Data were collected from electronic medical records, including demographic information, OP-related data, laboratory results (bone remodeling markers [BRM]), bone mineral density (BMD) of the lumbar spine (LS) and femoral neck (FN) measured by dual x-ray densitometry (Hologic Horizon®), and Trabecular Bone Score (TBS) at LS (Medimaps®). Data related to ROMO use, such as adverse events (AEs) and clinical fractures, were also registered. Changes in BRM and BMD from baseline to the end of follow-up were analyzed, and a bivariate analysis was conducted to assess factors associated with higher BMD gain in LS.
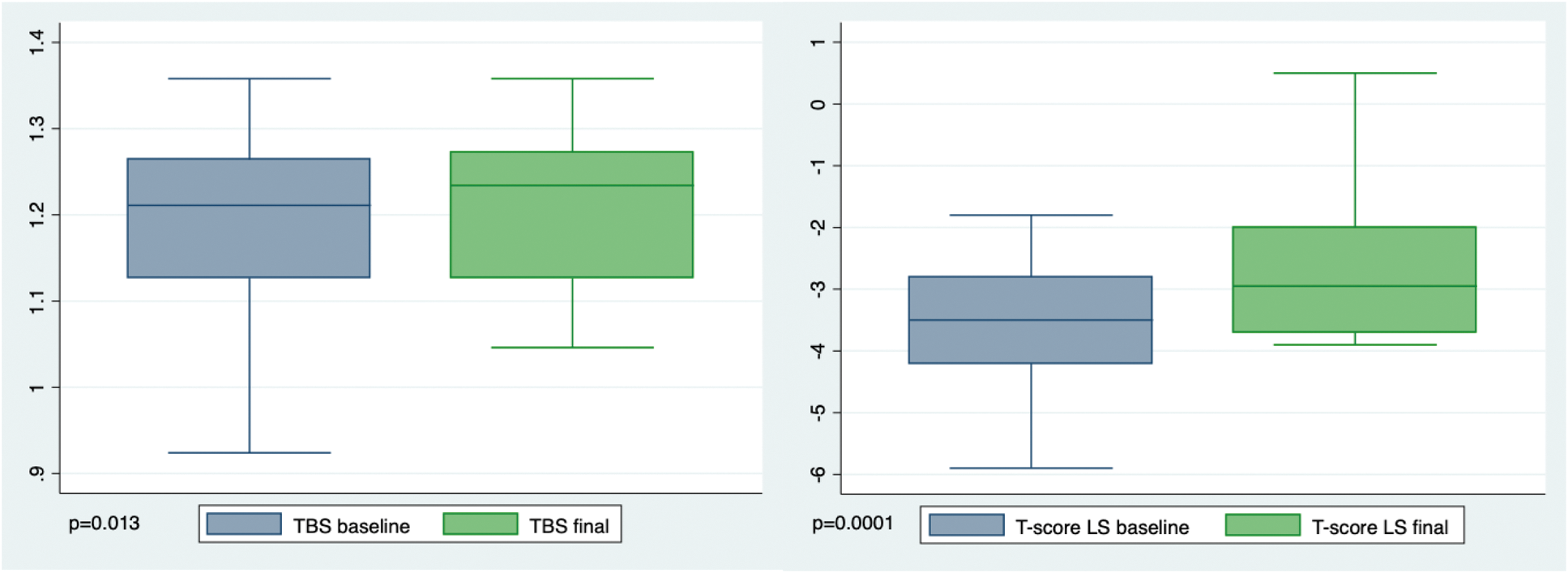
Results: Sixty-eight patients (Table 1) were included, with a marked prevalence of vertebral fractures (77.9%). Cardiovascular risk factors (CVRF) were present in 66.1% of the cohort. Prior to ROMO treatment, 75% of patients had received antiresorptive therapy, and 31% had received teriparatide. At the time of analysis, 38 patients (56%) had completed 12 doses of ROMO, while 21 patients (30%) were still receiving treatment. ROMO was discontinued in 9 patients (13.2%), 6 of them due to AEs. Two elderly patients with >1 CVRF experienced ischemic strokes, occurring one month and seven months after completing ROMO, respectively. In patients who completed ROMO and had BMD data (34 for FN and 25 for LS), BMD was stable in FN without significant T-score changes (p=0.808). However, the LS T-score showed a significant increase (-3.48 at baseline vs. -2.66 at the end; p=0.0001) (Figure 1). Additionally, LS BMD gain was >5% in 84% (21/25) of patients and >10% in 40% (10/25) of them. Factors associated with LS-BMD improvement included no prior anti-OP treatment with bisphosphonates (p=0.02) and fewer previous anti-OP therapies (p=0.20), but no correlation was found with age, OP risk factors, or the number/type of previous fractures. TBS also showed significant improvement (1.180 at baseline vs. 1.208 at 12-month follow-up; p=0.01). Only one osteoporotic fracture (Colles’ fracture) was reported in a patient eight months after completing ROMO. Regarding BRM, significantly lower Carboxy-terminal telopeptide of type 1 collagen (CTX) levels (p=0.01) and a trend toward lower alkaline phosphatase (p=0.09) were observed post-ROMO, with no differences in osteocalcin (p=0.83), calcium (p=0.19), or PTH (p=0.80).
Conclusion: In our experience, ROMO treatment for severe OP demonstrated acceptable tolerance, with significant densitometric gain in LS and stabilization in FN. Long-term real-world studies are needed to assess ROMO’s effectiveness in reducing fractures and its long-term cardiovascular safety profile.
Characteristics of the population treated with romosozumab.
| Characteristics | N = 68 patients (67 women) | |
|---|---|---|
| Age at initiation, mean ± SD | 71.3 ± 7.5 | |
| Patients with CVRF, n (%): | 45 (66.1) | |
| 1 CVRF/ ≥2 CVRF, n (%) | 25 (36.7)/ 20 (29.4) | |
| Osteoporosis risk factors: | ||
| Smoker, n (%) | 15 (22.0) | |
| Early menopause (n = 67 women), n (%) | 15 (22.1) | |
| Family history of hip fracture, n (%) | 8 (11.7) | |
| Hyperparathyroidism, n (%) | 4 (5.8) | |
| Hypothyroidism, n (%) | 16 (23.5) | |
| Chronic lung disease*, n (%) | 7 (10.3) | |
| Rheumatic or autoimmune diseases, n (%) | 10 (14.7) | |
| Treatment with OP-inducing drugs, n (%) | 30 (44.1) | |
| Previous history of fractures: | ||
| Vertebral fractures, n (%) | 53 (77.0) | |
| Hip fractures, n (%) | 6 (9.1) | |
| Other osteoporotic fractures**, n (%) | 15 (22.0) | |
|
Prior osteoporosis treatments:
| 54 (79.4)
|
|
| Bisphosphonates (oral or IV), n (%) | 48 (70.6) | |
| Denosumab, n (%) | 22 (32.3) | |
| Teriparatide, n (%) | 21 (30.9) | |
| Romosozumab treatment | ||
| Completed (12 doses), n (%) | 38 (55.9) | |
| Active (<12 doses), n (%) | 24 (35.3) | |
| Discontinuation, n (%) | 9 (8.8) | |
| Number of total AEs , n (%) | 12 (17.6) in 11 patients (16.1) | |
| Type of AEs | 2 CV events, 5 skin reactions, 3 myalgia, 1 headache | |
| Discontinuation due to AEs , n (%) | 6 (8.8) | |
| Dual x-ray densitometry and bone markers | Baseline | 12 months |
| Femoral Neck T-score, mean ± SD (n=34) | -2.70 ± 0.9 | -2.72 ± 0.8 |
| Lumbar Spine T-score, mean ± SD (n=25) | -3.48 ± 1.17 | -2.66 ± 1.06 |
| TBS, mean ± SD (n=19) | 1.180 ± 0.106 | 1.208 ± 0.099 |
| Calcium (mg/dL), mean ± SD (n=35) | 9.32±0.54 | 9.21±0.36 |
| Alkaline Phosphatase, mean ± SD (U/L) (n=33) | 89±27 | 83±30 |
| PTH (pg/mL), mean ± SD (n=18) | 81.5±28 | 82±29 |
| Osteocalcin (ng/mL), mean ± SD (n=8) | 19.9±7 | 29.2±11 |
| CTX (ng/mL), mean ± SD (n=8) | 0.397±0.26 | 0.148±0.03 |
*Chronic lung disease refers to patients with COPD, chronic bronchiectasis, and asthma.
**Including radius and humerus.
Abbreviations: AEs: adverse events. BMD: bone mineral density; CTX: C-terminal telopeptide of type 1 collagen; CV: cardiovascular; COPD: chronic obstructive pulmonary disease; CVRF: cardiovascular risk factors; OP: osteoporosis; PTH: parathyroid hormone; SD: standard deviation; TBS: trabecular bone score.
Evolution of LS bone dual x-ray densitometry under romosozumab therapy.
Abbreviations: LS: lumbar spine; TBS: trabecular bone score.
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (