

Background: Antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis (AAV) is a systemic small-vessel vasculitis [1]. AAV patients with life-threatening organ lesions, including rapidly progressive glomerulonephritis and alveolar hemorrhage, have a poor prognosis. Systemic glucocorticoids in combination with intravenous cyclophosphamide (CY) or rituximab (RTX) are recommended as the remission induction therapy for these severe patients [2]. Randomized clinical trials showed that there was no difference between RTX and CY in remission rate and incidence of side effects at 6 and 12 month after induction therapy [3, 4]. However, these studies included AAV patients with non-life-threatening complications, and follow-up period was limited. It remains unknown whether RTX is more effective than intravenous CY (IVCY) for severe AAV patients in a long-term, real-world setting.
Objectives: This study aimed to investigate the effectiveness and safety of rituximab (RTX) as a remission induction therapy in severe AAV compared to IVCY.
Methods: Microscopic polyangiitis (MPA) and granulomatosis with polyangiitis (GPA) patients treated with systemic glucocorticoids and IVCY or RTX as initial remission induction therapy were extracted in multicenter REVEAL study between 2005 and 2024 [5, 6]. They were diagnosed using the 2012 Chapel hill classification. We compared the effectiveness and safety outcomes between two groups. Effectiveness was evaluated by mortality, glucocorticoid (GC)-remission (i.e. Birmingham Vasculitis Activity score-remission plus a daily prednisolone dosage of <10mg) rate, and relapse rate. Safety was also evaluated by the mortality due to severe infections. Selection bias was reduced to a minimum using propensity score-based inverse probability of treatment weighting (IPTW).
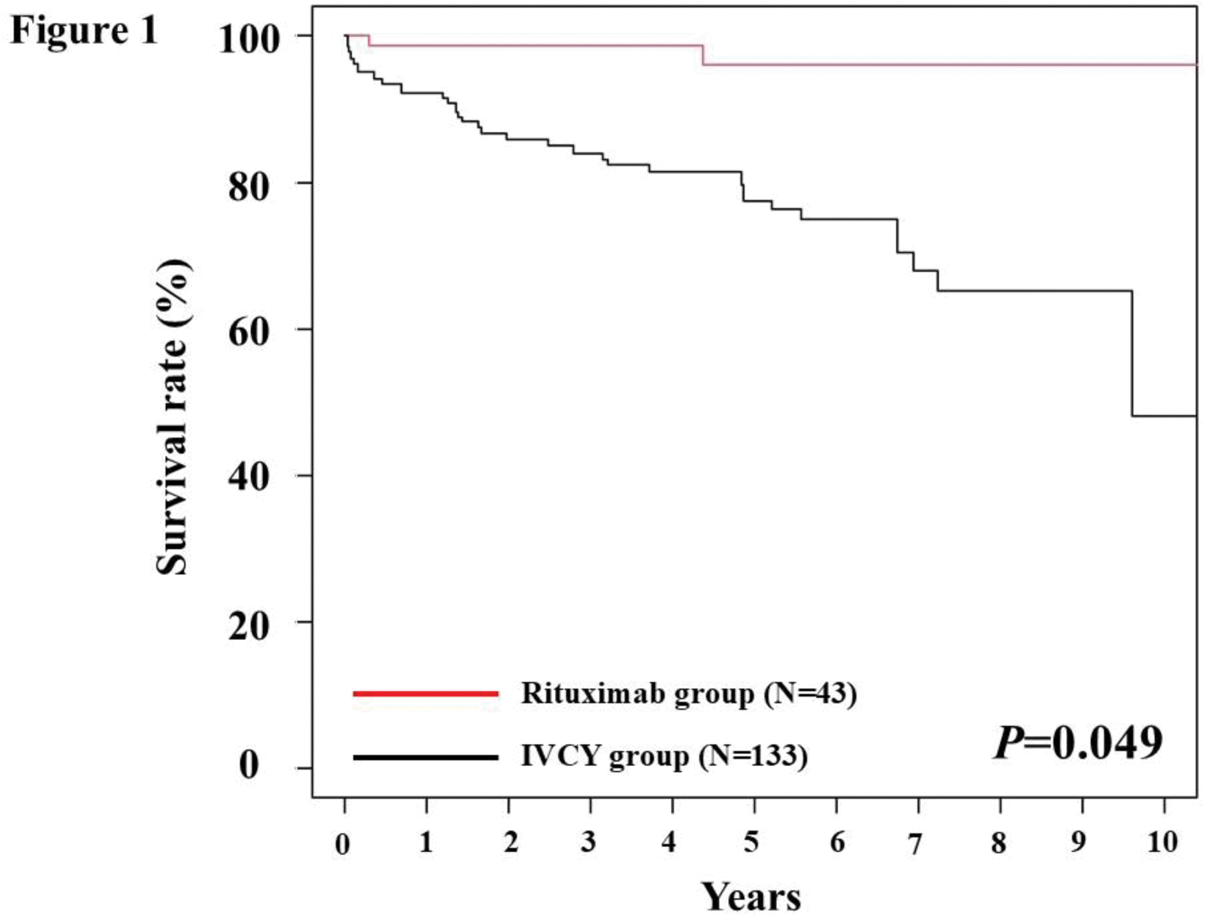
Results: Of the 397 MPA and GPA patients, 177 severe AAV patients were extracted. (IVCY group: N=132, RTX group: N=45) The median age, CRP, and BVAS of eligible patients were 73 years, 8.5 mg/mL, and 17, respectively. After adjustment by IPTW, there were no significant difference in baseline clinical characteristics between IVCY and RTX group (After IPTW: IVCY group: N=133, RTX group: N=43). The 10-year survival rate was significantly higher in the RTX group compared to IVCY group. (P=0.049, log rank test) (Figure 1) Also, GC-remission rate at 6 months was superior in RTX group (53%) compared to IVCY group (30%) (P=0.008). Relapse rates were comparable between two groups. Regarding safety, there were 21 deaths due to severe infection in the IVCY group, but none in the RTX group. (P=0.006)
Conclusion: In severe AAV, RTX remission induction therapy has superior effectiveness on mortality and GC-remission, and was associated with fewer infection-related death compared to IVCY treatment. These findings reveal the efficacy and safety of RTX remission induction therapy in a Japanese real-world practice.
REFERENCES: [1] Jennette JC, Falk RJ, Bacon PA, et al. 2012 revised International Chapel Hill Consensus Conference Nomenclature of Vasculitides. Arthritis Rheum. 2013;65(1):1-11.
[2] Hellmich B, Sanchez-Alamo B, Schirmer JH, et al. EULAR recommendations for the management of ANCA-associated vasculitis: 2022 update. Ann Rheum Dis. 2024;83(1):30-47.
[3] Jones RB, Tervaert JW, Hauser T, et al. Rituximab versus cyclophosphamide in ANCA-associated renal vasculitis. N Engl J Med. 2010;363(3):211-20.
[4] Stone JH, Merkel PA, Spiera R, et al. Rituximab versus cyclophosphamide for ANCA-associated vasculitis. N Engl J Med. 2010 Jul 15;363(3):221-32.
[5] Matsuda S, Kotani T, Okazaki A, et al. Prediction model for respiratory-related mortality in microscopic polyangiitis with interstitial lung disease: multicentre REVEAL cohort study. Rheumatology (Oxford). 2024;63(6):1607-1615.
[6] Matsuda S, Kotani T, Okazaki A, et al. Poor prognostic factors for relapse of interstitial lung disease in microscopic polyangiitis: the Japanese multicentre REVEAL cohort study. Arthritis Res Ther. 2024;26(1):221.
Acknowledgements: None.
Disclosure of Interests: Shogo Matsuda Grant/research support: Japan Intractable Diseases Research Foundation and Promotion and Mutual Aid Corporation for Private Schools of Japan, Takuya Kotani Speakers bureau:Abbvie, Bristol-Myers Squibb, Chugai, Eisai, Eli Lilly, Pfizer, Ayana Okazaki: None declared, Yuichi Masuda: None declared, Mayu Shiomi: None declared, Ryu Watanabe Speakers bureau: AbbVie, Asahi Kasei, Chugai, Eli Lilly, GSK, and UCB Japan, Grant/research support: AbbVie, Muneyuki Hatta: None declared, Tomoki Taniguchi: None declared, Mikihito Shoji: None declared, Tsuneyasu Yoshida: None declared, Ryosuke Hiwa Speakers bureau: Abbvie, Asahi Kasei, Brystol-Meyers Squibb, Eisai, Eli Lilly, GSK, Kissei, and Pfizer, Grant/research support:GSK, Youhei Fujiki: None declared, Naoko Ito: None declared, Hirofumi Miyake Speakers bureau: Abbvie, Asahi Kasei, AstraZeneca, Boehringer Ingelheim, Bristol-Myers Squibb, Chugai, Eisai, Eli Lilly, GlaxoSmithKline K.K, Sanofi, and Tanabe Mitsubishi, Wataru Yamamoto: None declared, Motomu Hashimoto Speakers bureau: Eli Lilly, Chugai, Tanabe-Mitsubishi, Bristol Myers Squibb, Esai, Grant/research support:AbbVie, Asahi Kasei Pharma, Astellas, Bristol Myers Squibb, Esai, Daiichi Sankyo, Eli Lilly, Novartis, Tohru Takeuchi Speakers bureau: Asahi Kasei, Chugai, Bristol-Myers Squibb, Tanabe-Mitsubishi, AbbVie, Esai, Grant/research support: Chugai.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (