

Background: Non-infectious uveitis may be due to several immune-mediated inflammatory diseases (IMIDs) that can lead to severe ocular damage. Uveitis may be refractory to conventional immunosuppressants and even biological therapy. Janus Kinase inhibitors (JAKINIB) had shown efficacy in refractory cases.
Objectives: To assess efficacy and safety of JAKNIBs, in patients with uveitis due to IMIDs refractory to different immunosuppressive or biologic therapy, and to review the literature.
Methods: Multicenter study of 16 patients with refractory IMIDs-related uveitis treated with JAKNIBs. Our evaluated variables were best corrected visual acuity (BCVA), anterior chamber cells and presence of cystoid macular edema. These outcomes were recorded from baseline up to 1 year treatment. We searched on PubMed, Embase and the Cochrane library from their inception to 1 st December 2024 for the literature review, including patients with non-infectious uveitis treated with JAKNIBs.
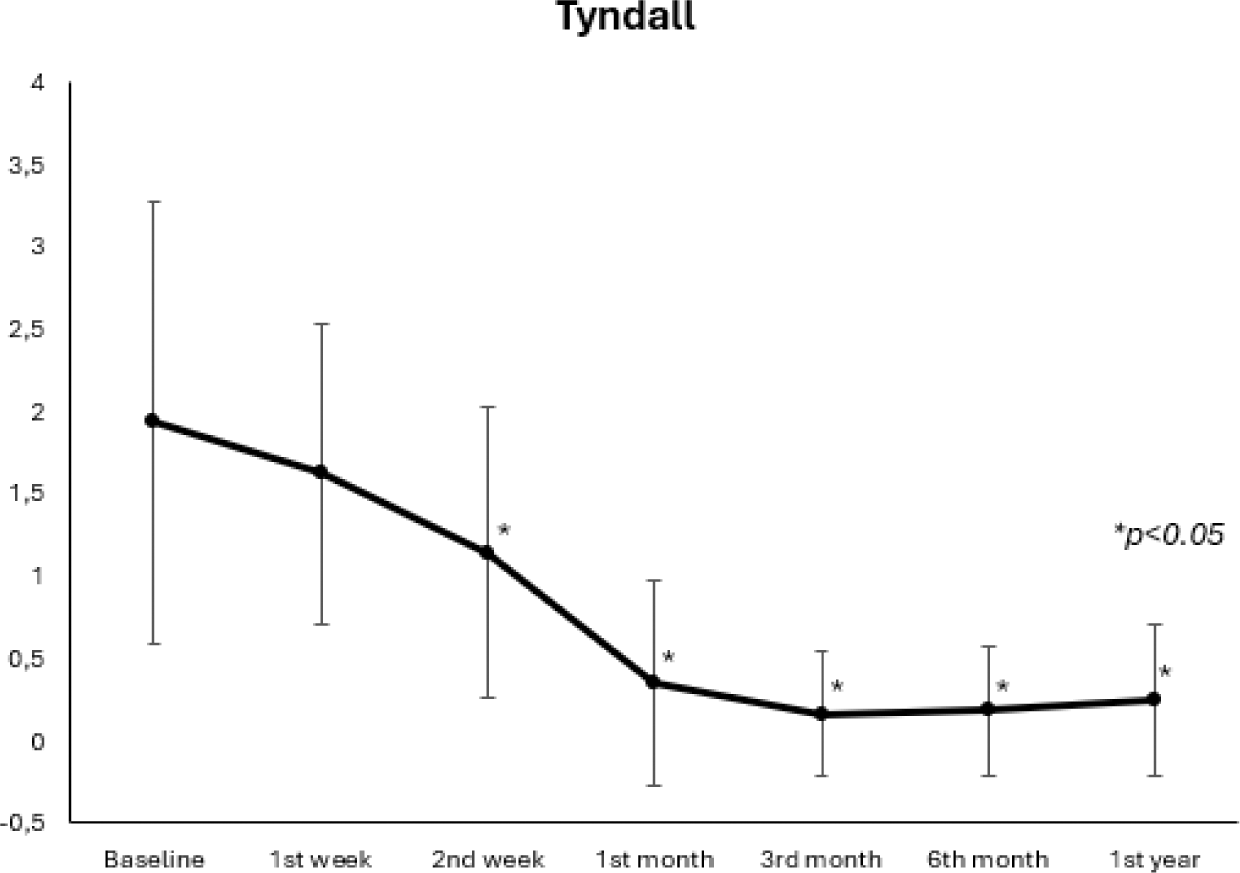
Results: We studied 16 patients from Spain and 13 cases from the literature review. There were 22 women and 7 men. There were panuveitis (n=12), anterior (n=15;), intermediate (n=1) and posterior uveitis (n=1). There were 24 patients with IMIDs associated uveitis while 5 cases were idiopathic. The main underlying IMIDs were spondylarthritis (n=13) and juvenile idiopathic arthritis (n=6) (Table 1 ). Treatment with conventional (n=24) or biological immunosuppressive drugs (n=25) was required before the JAKNIBs. The JAKINIB most widely used was Upadacitinib (n= 15). Complete clinical improvement was observed after starting JAKNIBs treatment, 26 patients presented a complete improvement, and the remaining patients showed partial improvement. In the multicenter study, BCVA showed a rapid and maintained improvement (Figure 1) after 13 months of follow-up. Twelve patients had Tyndall + at baseline with a partial decrease from the first month (n=4) and with resolution by the sixth month (Figure 2). One patient had cystic macular edema at baseline, resolved at 3 months of follow-up. No serious adverse effects were observed.
Conclusion: It seems that treatment with JAKNIBs in uveitis related to different IMIDs may be effective and safe, even in refractory to previous biological drugs cases.
Table 1. Cases reports and Literature review of patients with uveitis treated with Janus Kinase Inhibitors.
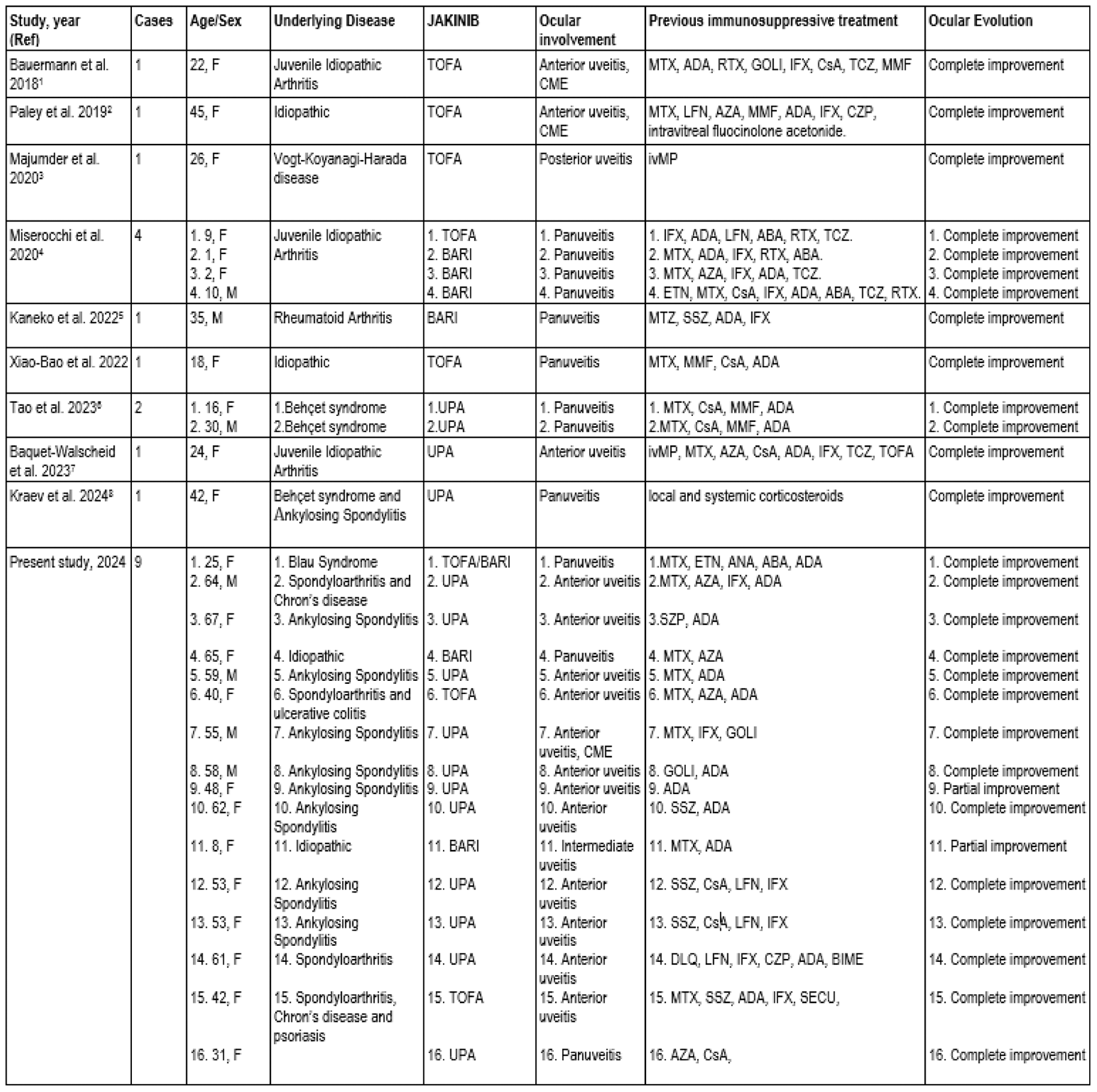
Rapid and maintained improvement of best corrected visual acuity (BCVA) following the initiation of JAKINIB.
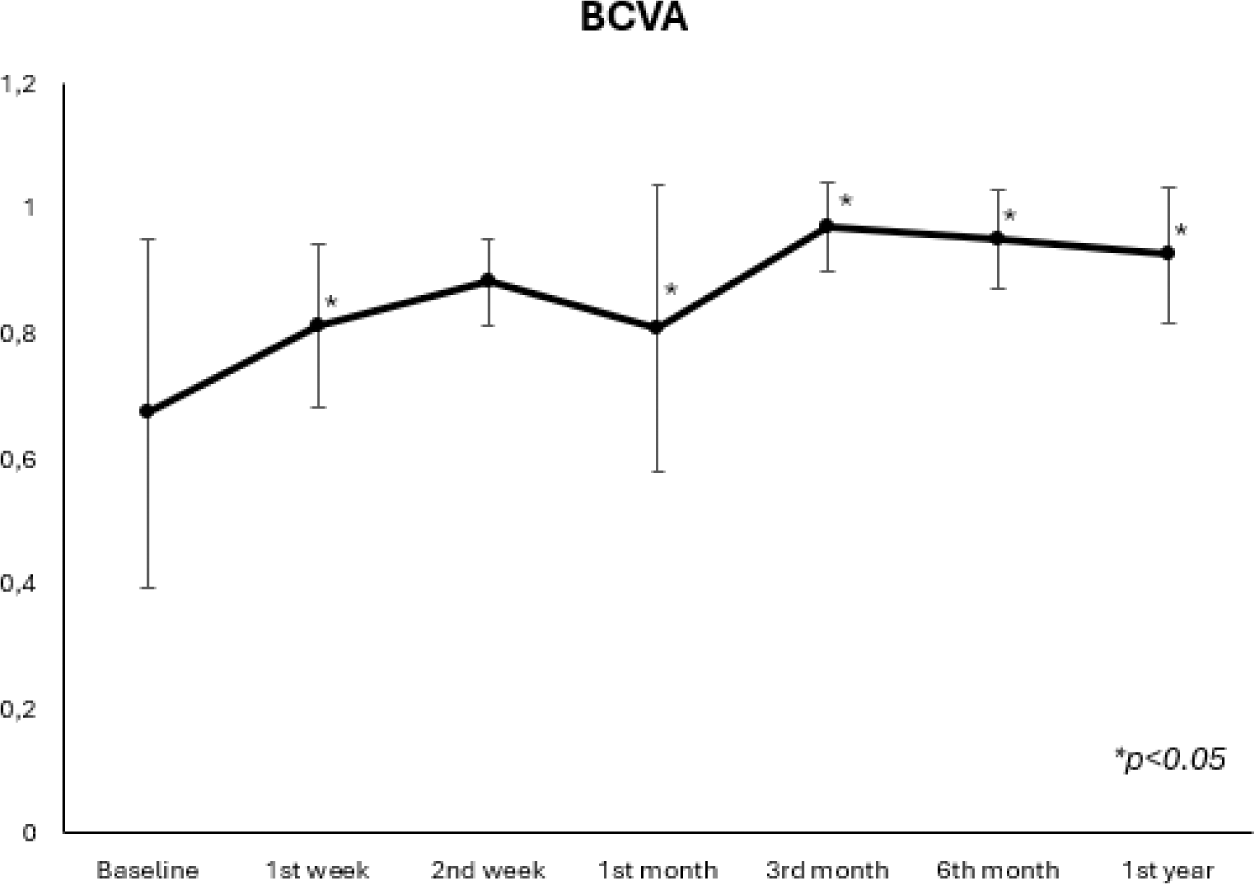
Decrease of Tyndall following the initiation of JAKNIB.
REFERENCES: [1] Bauermann P, Heiligenhaus A, Heinz C. Effect of Janus Kinase Inhibitor Treatment on Anterior Uveitis and Associated Macular Edema in an Adult Patient with Juvenile Idiopathic Arthritis. Ocul Immunol Inflamm. 2019;27(8):1232-1234
[2] Paley MA, Karacal H, Rao PK, Margolis TP, Miner JJ. Tofacitinib for refractory uveitis and scleritis. Am J Ophthalmol Case Rep. 2018 Dec 4;13:53-55.
[3] Dutta Majumder P, Shah A, Kaushik V. Tofacitinib in Vogt-Koyanagi-Harada disease. Indian J Ophthalmol. 2020 Sep;68(9):1938-1939.
[4] Miserocchi E, Giuffrè C, Cornalba M, Pontikaki I, Cimaz R. JAK inhibitors in refractory juvenile idiopathic arthritis-associated uveitis. Clin Rheumatol. 2020 Mar;39(3):847-851.
[5] Kaneko Y, Murakami T, Nishitsuka K, Takakubo Y, Takagi M, Yamashita H. Effectiveness of Baricitinib in Refractory Seronegative Rheumatoid Arthritis and Uveitis: A Case Report. Front Med (Lausanne). 2022 Jan 14;8:764067.
[6] Tao T, He D, Peng X, Huang Z, Su W. Successful Remission with Upadacitinib in Two Patients with Anti-TNF-Refractory Macular Edema Associated with Behçet’s Uveitis. Ocul Immunol Inflamm. 2023 Oct 6:1-4.
[7] Baquet-Walscheid K, Heinz C, Rath T, Scheel M, Heiligenhaus A. Beneficial Effect of Upadacitinib in an Adult Patient with Juvenile Idiopathic Arthritis-associated Uveitis after Unsatisfactory Response to Tofacitinib: A Case Report. Ocul Immunol Inflamm. 2023 Jul;31(5):1079-1080.
[8] Kraev K, Uchikov P, Hristov B, Kraeva M, Basheva-Kraeva Y, Popova-Belova S, et al. Coexistence of ankylosing spondylitis and Behçet’s disease: Successful treatment with upadacitinib. Immun Inflamm Dis. 2024 Apr;12(4):e1242.
Acknowledgements: NIL.
Disclosure of Interests: Nuria Barroso García: None declared, Lara Sanchez-Bilbao Roche, Lilly, and Pfizer, José Luis Martín-Varillas AbbVie, Pfizer, Lilly, Celgene, Janssen, and UCB Pharma, Vanesa Calvo-Río AbbVie, Lilly, Celgene, Grünenthal, and UCB Pharma, MSD and Roche, María del Mar Esteban-Ortega: None declared, Santiago Muñoz-Fernández: None declared, José Luis Álvarez Vega: None declared, Emma Beltrán Abbvie, Amgen, Janssen, MSD, Pfizer, Lilly, Novartis and UCB, Vega Jovani: None declared, Olga Maíz-Alonso: None declared, Raúl Veroz González: None declared, Ángel García-Aparicio: None declared, Marta Garijo Bufort: None declared, Ricardo Blanco AbbVie, Pfizer, Roche, Bristol-Myers, Janssen, and MSD, Abbvie, MSD, Roche.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (