

Background: The role of probiotics in rheumatoid arthritis (RA) treatment has gained attention, as gut microflora appears to be a key factor in RA pathogenesis. Immunobiotics, a type of probiotic capable of beneficially modulating the host’s immune system, may enhance anti-inflammatory responses, compete with pathogenic bacteria for nutrients, and prevent pathogen adhesion to the intestinal wall. Probiotics containing Lactobacillus and Bifidobacterium produce short-chain fatty acids (SCFAs), which exhibit anti-inflammatory effects. They interact directly with immune cells, stimulate anti-inflammatory cytokines, and improve regulatory T cell function.
Objectives: The primary objective was to evaluate the effect of immunobiotics on disease activity in RA patients. The secondary objective was to compare the tolerability of conventional DMARDs (cDMARDs) in patients with and without probiotics.
Methods: This single-center, double-blinded, placebo-controlled randomized clinical trial included 100 patients. Inclusion criteria were: RA diagnosis and initiation of peroral methotrexate (MTX) and/or corticosteroids within the last six months. MTX dosage had to remain stable for four weeks prior to screening, and corticosteroid doses were tapered to a maximum of 7.5 mg/day. Exclusion criteria included: age under 20 or over 80 years; non-RA diagnoses; biologic or JAK inhibitor therapy; history of synovectomy; gastrointestinal conditions ( Helicobacter pylori infection, bleeding, ulcers, pancreatitis, or surgery); serious infections; immune deficiencies; organ dysfunction (liver or kidney); hematologic disorders; malignancies; psychiatric disorders; or recent probiotic use exceeding seven days in the past six months. Written informed consent was obtained from all participants. Patients were randomized into two groups (n=50 each):
Group A (Experimental ): Seropositive RA patients treated with cDMARDs and/or corticosteroids, and daily immunobiotics ( Lactobacillus casei BL 2401 , Lactobacillus salivarius BL 2201 , Bifidobacterium breve BL 3406 ).
Group B (Control ): RA patients treated with cDMARDs and/or corticosteroids and placebo.
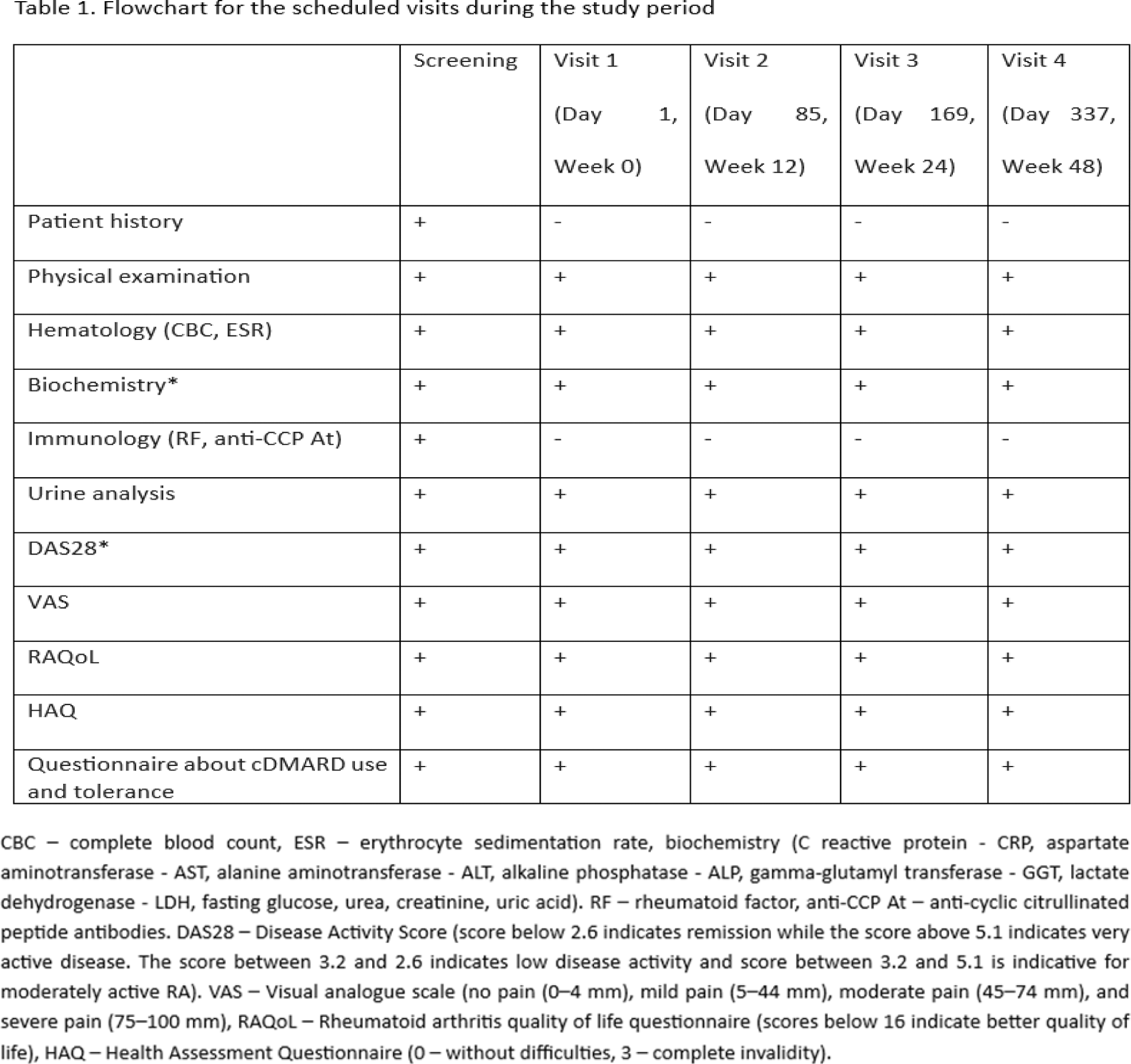
Patients were matched by age, sex, and socioeconomic status and followed for one year. Assessments were conducted at baseline and at three-month intervals, including clinical, laboratory, and patient-reported measures (Table 1). Statistical analyses were performed using SPSS version 26.
Results: Among the 100 participants (83% female), the mean age was 60 years in Group A and 54 years in Group B. At baseline, high disease activity was observed in 51% of Group A and 69% of Group B (p>0.05). After 24 weeks, remission was achieved in 61.2% of Group A and 12% of Group B (p<0.05). By week 48, remission rates were 63.3% in Group A, with no patients in remission in Group B (p<0.05). The mean erythrocyte sedimentation rate (ESR) was consistently higher in the control group compared to the experimental group at week 24, week 36, and week 48 (23 vs. 16, 28 vs. 17, 35 vs. 14; p < 0.05). Regarding C-reactive protein (CRP) levels, patients in Group A had significantly lower CRP values compared to Group B starting at week 36 (5 vs. 10 at week 36 and 4 vs. 30 at week 48; p < 0.01). Pain levels, assessed via the VAS scale, showed no significant differences at baseline. However, after 24 weeks, 61.2% of Group A reported no pain, compared to no pain-free patients in Group B (p<0.05). By week 48, 83.7% of Group A remained pain-free, while 78% of Group B experienced severe pain (p<0.01). Functional improvements (HAQ scores) in Group A were observed at 36 weeks (0.8 vs. 1.0; p=0.017) and persisted at week 48 (0.7 vs. 1.2; p=0.01). Quality of life (RAQoL) scores showed significant differences favoring Group A at week 24 (8.16 vs. 11.4; p=0.015), week 36 (7.88 vs. 12.7; p=0.001), and week 48 (7.5 vs. 14.8; p=0.001). MTX tolerability was better in Group A, with no patients discontinuing therapy or switching to parenteral formulations. In contrast, 20% of patients in Group B required switching to the parenteral route due to intolerance ( p = 0.03). Additionally, six out of 50 patients in Group B transitioned to biologics due to very high disease activity.
Conclusion: Immunobiotics combined with cDMARDs resulted in higher remission rates, reduced pain, improved functionality, and better quality of life compared to cDMARDs alone. Additionally, MTX tolerability was enhanced in the immunobiotic group. Further randomized controlled trials and meta-analyses are essential to validate these findings.
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: Branko Barac The company Save Health provided immunobiotics for the research, Sretko Lukovic: None declared, Tijana Divljakovic: None declared, Tatjana Zivanovic Radnic: None declared, Aleksandra Bibic: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (