

Background: Avacopan is an oral, selective C5a receptor antagonist approved by the European Medicines Agency in January 2022 for the treatment of adults with severe, active granulomatosis with polyangiitis (GPA) or microscopic polyangiitis (MPA), in combination with rituximab or cyclophosphamide. In the pivotal phase 3 ADVOCATE study, avacopan was non-inferior to prednisone-taper for achieving remission at Week 26, superior in sustaining remission at Week 52, and was associated with improved estimated glomerular filtration rate from baseline. AvacoStar, a real-world cohort study with follow up beyond 1 year, seeks to investigate long term safety of avacopan.
Objectives: AvacoStar is designed to evaluate the incidence rates of defined medical events of special interest which include liver injury, cardiac safety, serious infections, and malignancy during long-term follow-up in a real-world cohort of participants commencing avacopan for severe, active GPA or MPA. We present baseline characteristics of the first 314 patients included in the study.
Methods: AvacoStar (NCT05897684) is a non-interventional, multinational, prospective, open-label, non-randomised, post-authorisation safety study. This study will enrol 500 patients in Germany and the UK, divided into two cohorts of 250: those treated with avacopan, and a second cohort treated with a cyclophosphamide or rituximab-based induction regimen without avacopan. Eligible patients are those aged ≥18 years with severe, active GPA or MPA in the opinion of the investigator, at the time of initiation of avacopan or non-avacopan standard of care induction therapy. Patients will be followed for up to 7 years (2023–2030).
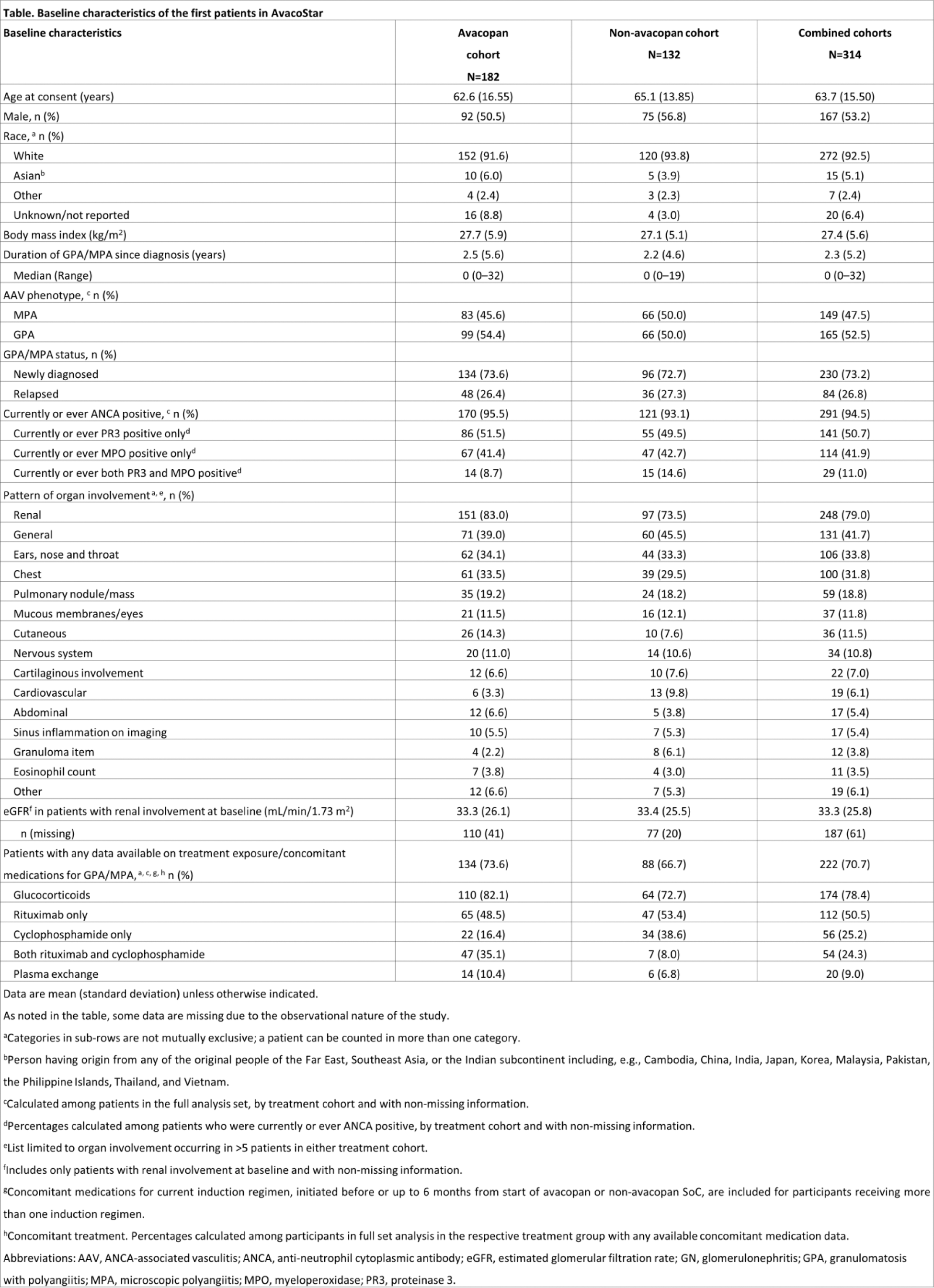
Results: From Sep 2023 until Oct 2024, 314 patients were included in this analysis (n=182 avacopan cohort; n=132 non-avacopan cohort). The main reason for choosing the avacopan regimen or non-avacopan regimen was to align with the usual standard of care at the participating centre (Figure 1). Most patients enrolled were Caucasian (92.5%), newly diagnosed (73%) and the majority had renal involvement (79%) (Table 1). More patients in the avacopan cohort have renal involvement and longer duration of GPA/MPA since diagnosis. So far, more patients in the avacopan cohort received rituximab plus cyclophosphamide induction compared to patients in the non-avacopan cohort.
Conclusion: At the time of data collection, the majority of baseline characteristics are comparable in general between the cohorts with some imbalances in duration since AAV diagnosis and organ involvement. There is a higher proportion of patients that received rituximab plus cyclophosphamide for induction in the avacopan cohort. The initial findings indicate that the outcomes of the study could provide valuable and applicable information about the safety and usage patterns of avacopan. This study is still enrolling patients. Limitations of this analysis include missing data, a higher recruitment rate in the avacopan cohort at the time of the data cut and data variability from routine clinical practice.
Reasons for selecting the avacopan regimen or non-avacopan regimen*
GC, glucocorticoids. *Patients could be counted in more than one category.
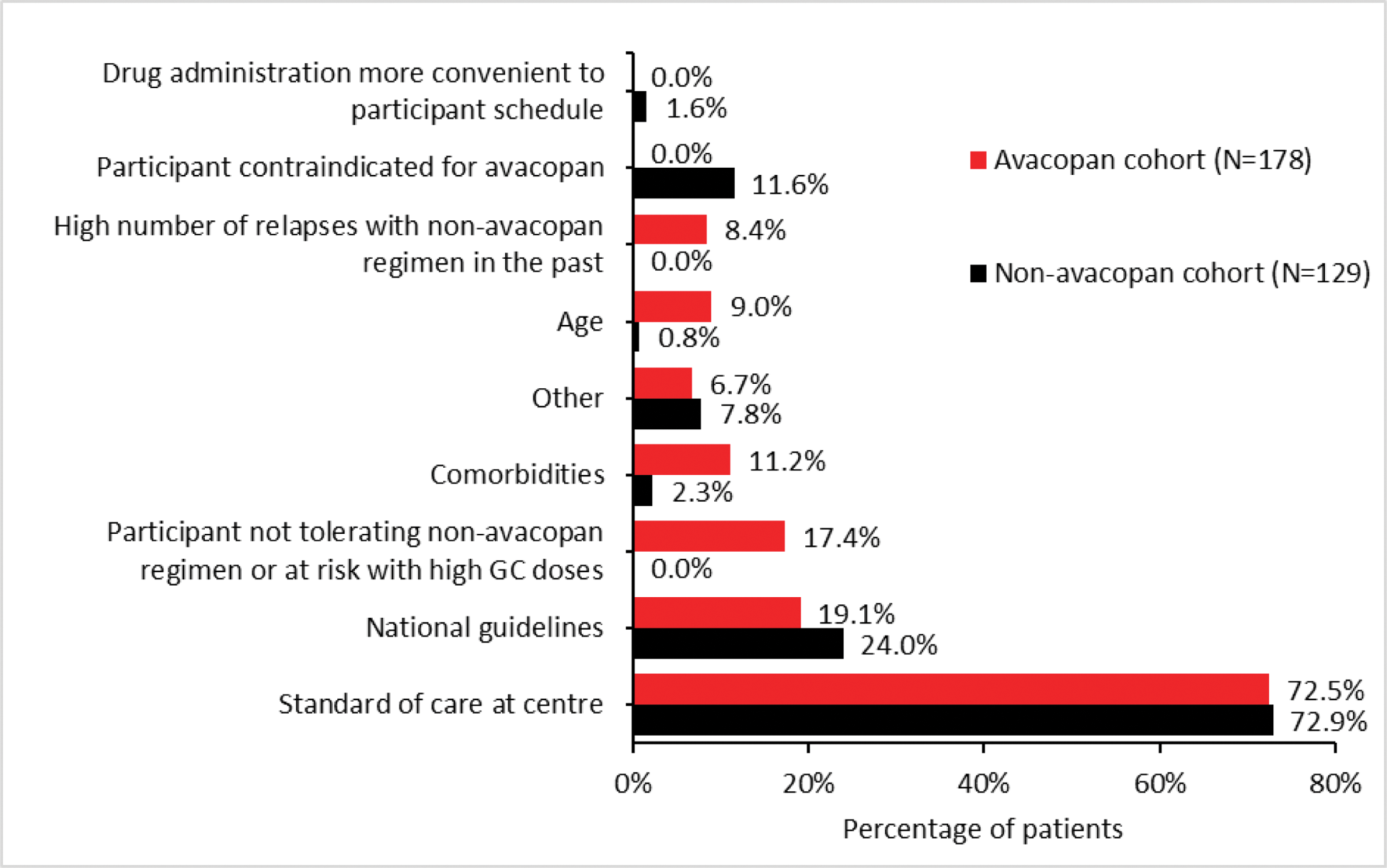
Table 1.
REFERENCES: NIL.
Acknowledgements: Medical writing support for this abstract was provided by Donna Kennedy (Obsidian Healthcare Group Ltd) and funded by Vifor Fresenius Medical Care Renal Pharma Ltd. The AvacoStar study is funded by Vifor Fresenius Medical Care Renal Pharma Ltd.
Disclosure of Interests: Bernhard Hellmich Lecture fees: Abbvie, Astra Zeneca, BMS, GSK, Janssen, InflaRx, Novartis, Pfizer, Roche, Novartis, Pfizer, Recordati, Roche, CSL Vifor, Consulting: Abbvie, Astra Zeneca, BMS, GSK, Janssen, InflaRx, Novartis, Pfizer, Roche, Novartis, Pfizer, Recordati, Roche, CSL Vifor, Raashid Luqmani Lecture fee: GSK, CSL Vifor, Advisory board: GSK, Consulting: GSK, Roche, CSL Vifor, BMS, Celgene, CSL Vifor, Benjamin Terrier Lecture fee: AstraZeneca, GSK, Boehringer Ingelheim, CSL Vifor, Novartis, Advisory board: AstraZeneca, GSK, Boehringer Ingelheim, CSL Vifor, Novartis, Monica Balcells Shares: CSL Vifor - Amgen, Employee: CSL Vifor, Marie Boff Employee: CSL Vifor, Achim Obergfell Employee: CSL Vifor, David R. W. Jayne Lecture fee: Amgen, CSL Vifor, Otsuka, Shares: Alentis, Aurinia, Consulting: Alentis, Amgen, AstraZeneca, Aurinia, BMS, Boehringer, Chinook, GSK, Novartis, Otsuka, Roche/Genentech, Takeda, CSL Vifor.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (