

Background: Since its FDA approval in 2021, avacopan has been revolutionizing the induction treatment of ANCA-associated vasculitis (AAV) [1]. Starting in May 2024, it became approved and reimbursable in Italy; however, it had already been used in several Italian patients through the Early Access Program (EAP).
Objectives: The aim of this study was to evaluate the efficacy, safety, and impact on quality of life of avacopan during the EAP in a real-life cohort of Italian patients.
Methods: The prospective collection of clinical data began in May 2022 and included patients with either GPA or MPA, encompassing both newly diagnosed and relapsing cases who initiated avacopan therapy (baseline). The Italian AAV-PRO questionnaire was completed for each patient every three months [2].
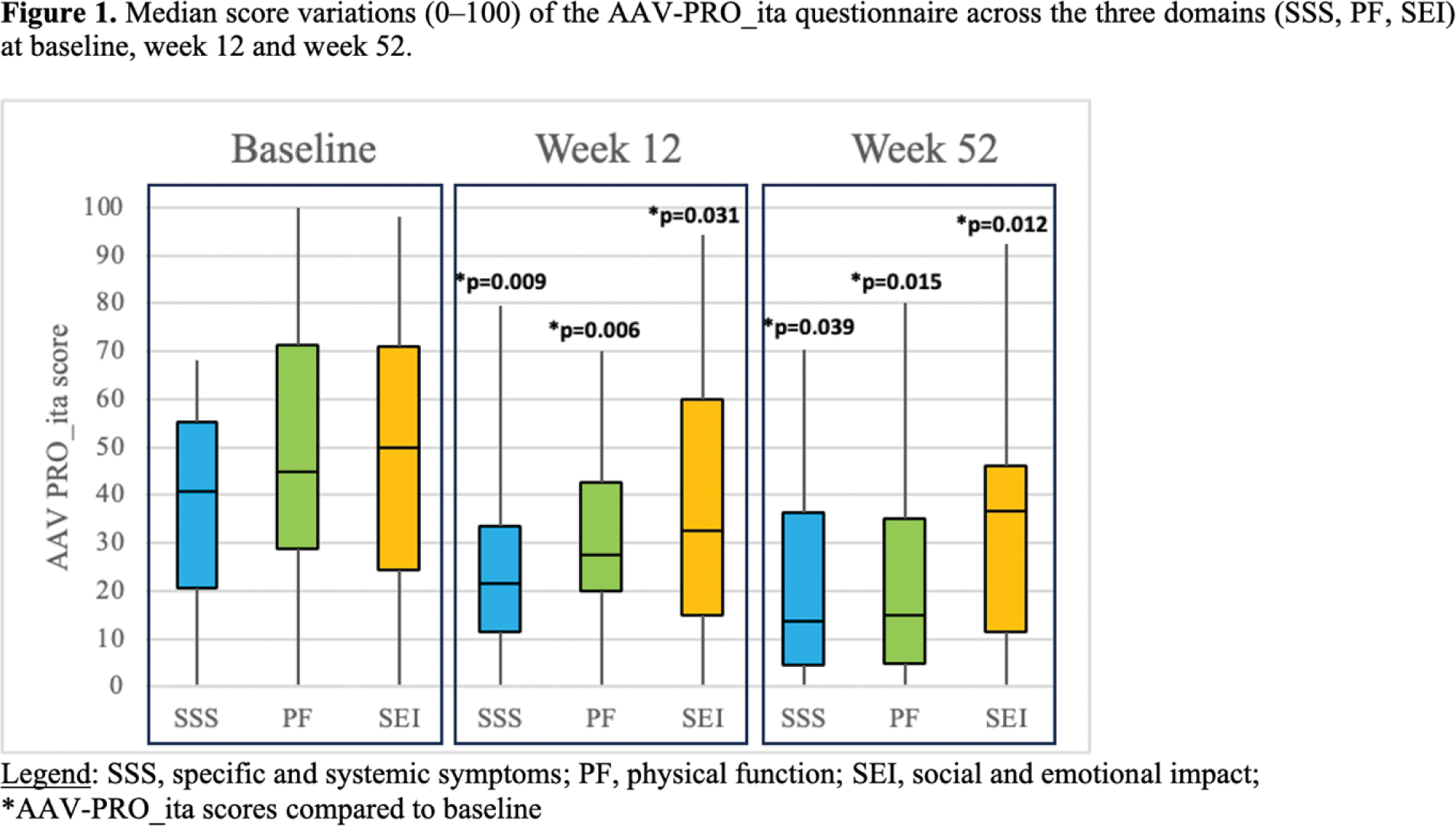
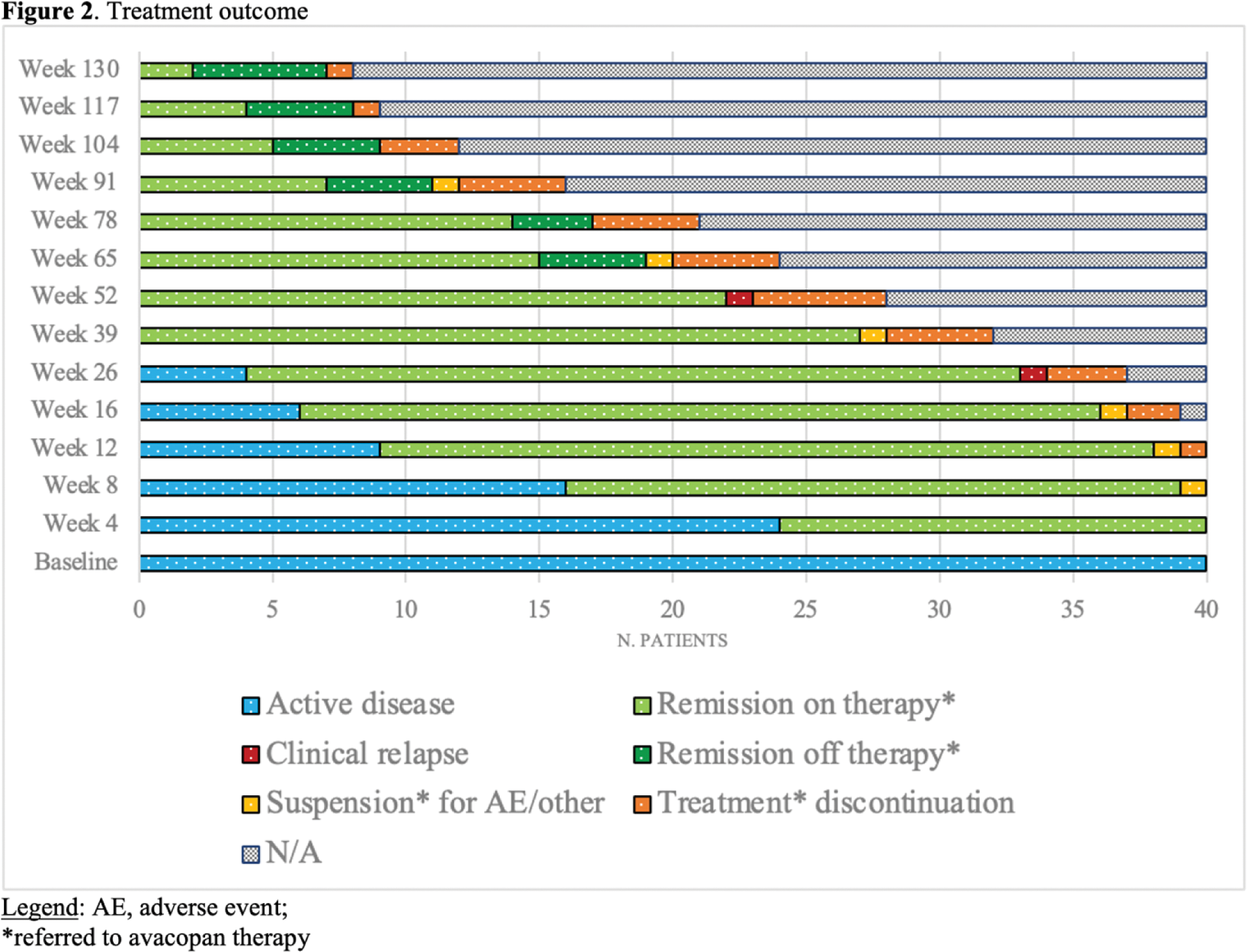
Results: Forty patients from 15 Italian Rheumatology/Nephrology departments were included: 25 with GPA (24/25 c-ANCA/PR3+; 1/25 p-ANCA/MPO+) and 15 with MPA (14/15 MPO+; 1/15 ANCA negative). At baseline, the median age of patients was 61 (IQR 52-66) years. The majority of patients were female (68%) and Caucasians (95%). Forty-five percent of the patients (18/40) initiated avacopan therapy following a new diagnosis of AAV. Patients treated after a relapse (22/40) had a median disease duration of 7 (IQR 8-13) years and prior treatments with RTX (77%), CYC (45%), MTX (27%), AZA (18%), MMF (14%). At baseline, BVASv3 and VDI were 8 (IQR 4-17) and 2 (IQR 0-4), respectively. The main disease manifestations included renal involvement (70%), with 18 out of 28 patients presenting with rapidly progressive glomerulonephritis; systemic symptoms (73%); and pulmonary involvement (48%), with 6 out of 19 patients experiencing hemorrhagic alveolitis. Avacopan was administered in combination with RTX in almost all patients (98%), with 8 patients receiving CYC IV beforehand (median cumulative dose: 1000 grams [IQR 400-1225]). Two out of 18 patients with rapidly progressive glomerulonephritis also underwent plasmapheresis. At the baseline, the median corticosteroid (CS) dose was 25 mg/day (IQR 5-41). For patients with renal involvement (28/40), the median baseline creatinine and proteinuria values were 1.95 mg/dL (IQR 1.6-2.9) and 1155 mg/24h (IQR 465-2500), respectively. Twenty-four out of 28 patients had microhematuria. At 12 weeks, 78% of patients had achieved clinical remission, with a quarter (26%) being steroid-free. The median CS dose had decreased to 5 mg/day (IQR 1-5) ( p-value <0.001 ). The median creatinine and proteinuria values had reduced to 1.7 mg/dL (IQR 1.2-1.9) ( p-value=0.002 ) and 391 mg/24h (IQR 153-1350) ( p-value=0.013 ), respectively. Only ten out of 28 patients still presented with microhematuria ( p-value>0.001 ). A statistically significant reduction in AAV-PRO questionnaire scores was observed from baseline to week 12 (Figure 1). Avacopan was continued for a median duration of 52 weeks (IQR 26–78), and slightly less than half of the patients (43%) continued avacopan beyond one year. The percentage of patients in sustained CS-free remission increased from 64% (15/22) at week 52 to 78% (7/9) at week 104 (Figure 2). Eight out of 40 patients discontinued the treatment, primarily due to relapses (2/8), infectious (4/8), frailty and comorbidities (1/8), or patient’s decision (1/8). The two clinical relapses were reported at weeks 26 and 52, necessitating the resumption of CS and RTX plus CS, respectively. One death at week 65 due to a fungal infection was registered.
Conclusion: This study highlights the early and sustained efficacy, safety, and improvements in quality-of-life scores associated with avacopan treatment in the real-world management of AAV in a cohort of Italian patients. Avacopan enables early clinical remission, CS reduction, and improvements in renal function parameters. Treatment discontinuation occurred in 20% of patients due to relapses, infections, or comorbidities; however, the continuation of treatment beyond one year in nearly half of patients suggests that avacopan is well-tolerated for long-term disease control.
REFERENCES: [1] Jayne, D.R.W.; Merkel, P.A.; Schall, T.J.; Bekker, P. Avacopan for the Treatment of ANCA-Associated Vasculitis. New England Journal of Medicine 2021 , doi:10.1056/NEJMoa2023386.
[2] Treppo, E.; Isola, M.; De Martino, M.; Padoan, R.; Giollo, A.; Urban, M.L.; Monti, S.; Sartorelli, S.; Fassio, A.; Argolini, L.M.; et al. Validation of the Italian Version of the ANCA-Associated Vasculitis Patient-Reported Outcome (AAV-PRO) Questionnaire. Rheumatol Adv Pract 2024 , 8 , rkae001, doi:10.1093/rap/rkae001.
Acknowledgements: We would like to thank the Italian Vasculitis Study Group and the following collaborators: Paolo Delvino, Alessandra Bettiol, Francesco Delle Monache, Emilio Venturelli, Marco Fornaro.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (