

Background: FINCH 1 (NCT02889796) was a Phase 3 randomized controlled 52-week trial of filgotinib, a Janus kinase 1 preferential inhibitor, in patients with rheumatoid arthritis (RA) with an inadequate response to methotrexate [1]. Patients completing FINCH 1 could continue to receive filgotinib in a long-term extension (LTE) study. In a previously reported post hoc analysis of patients treated with filgotinib 200 mg in FINCH 1, five distinct patient groups were identified based on Clinical Disease Activity Index (CDAI) outcomes, using group-based trajectory modeling [2].
Objectives: This post hoc analysis of FINCH 1 aimed to assess improvements in patient-reported outcomes (PROs), as well as radiographic and long-term safety outcomes, in these distinct CDAI trajectory groups.
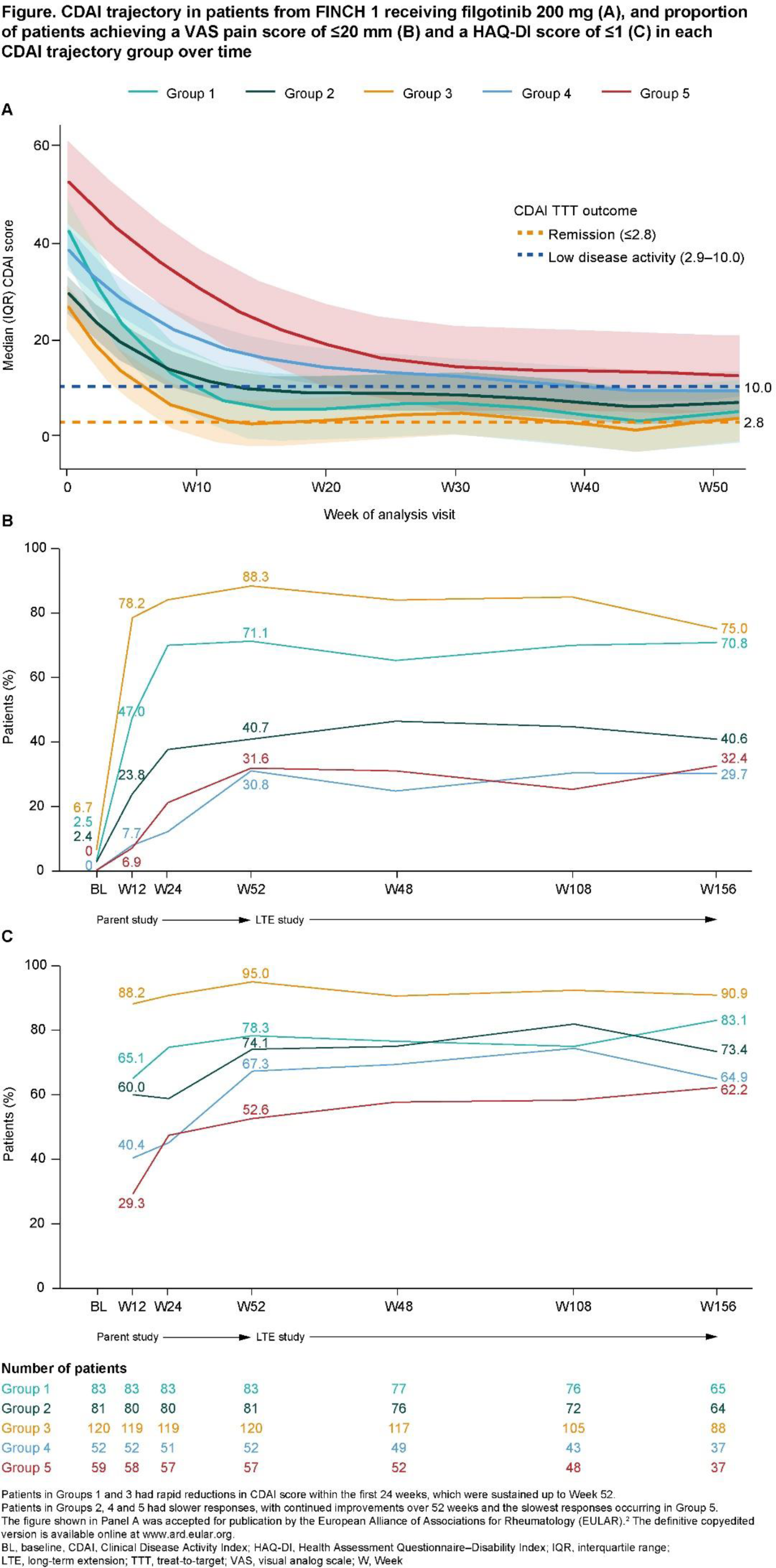
Methods: As previously reported, group-based trajectory modeling identified five distinct patterns of CDAI response (Figure 1A): patients in Groups 1 and 3 had rapid reductions in CDAI score within the first 24 weeks, which were sustained up to Week 52 [2]. Patients in Groups 2, 4 and 5 had slower responses, with continued improvements over 52 weeks; the slowest responses occurred in Group 5 [2]. The current analysis included all patients from FINCH 1 who entered the LTE study (FINCH 4), which was the majority of patients from FINCH 1. The following PROs were assessed in each of the five groups up to Week 156 of the LTE study: mean pain score (measured on a visual analog scale [VAS]), the proportion of patients achieving a VAS pain score of ≤20 mm (health status not negatively affected by pain) and the proportion of patients achieving a Health Assessment Questionnaire–Disability Index (HAQ-DI) score of ≤1 (mild disability). Radiographic progression was assessed in each group by measuring the change from baseline in modified total Sharp score (mTSS) up to Week 52 of FINCH 1. Safety was evaluated in each group by assessing the exposure-adjusted incidence rate (EAIR) per 100 patient-years (PYE) of exposure for treatment-emergent adverse events (TEAEs) up to Week 156 of the LTE study.
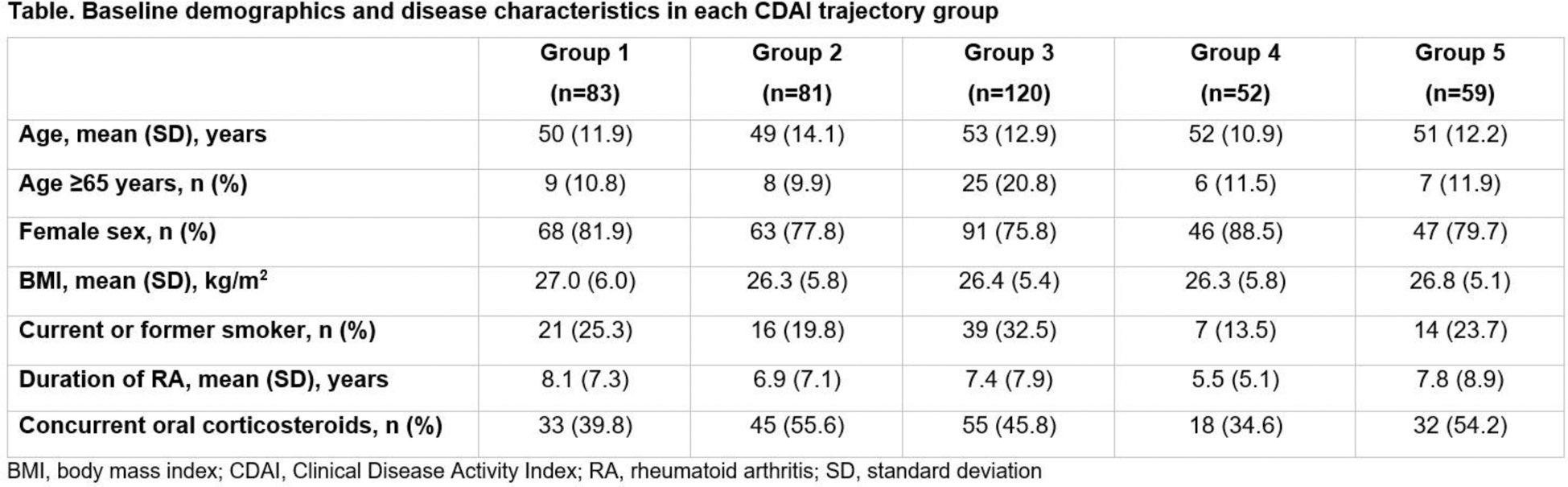
Results: Baseline demographics and disease characteristics of patients in all CDAI trajectory groups are shown in the Table 1. Age, sex and BMI were broadly similar across groups. Mean (SD) duration of RA from diagnosis was shortest in Group 4, at 5.5 (5.1) years, and longest in Group 1, at 8.1 (7.3) years. The proportion of patients receiving concurrent oral corticosteroids at baseline of FINCH 1 and FINCH 4 was 34.6–55.6%. Mean VAS pain scores rapidly decreased from baseline to Week 12 and 24 in Groups 1 and 3 (the groups with the fastest reductions in CDAI score); improvements were maintained up to Week 156. Patients in Group 5 (the group with the slowest reductions in CDAI score) showed a slower decrease in mean VAS pain score than those in Groups 1 and 3 during the initial 24 weeks. Across groups, the proportion of patients achieving a VAS pain score of ≤20 mm at Week 12 and 156, respectively, was greatest in Group 3 (78.2% and 75.0%) followed by Group 1 (47.0% and 70.8%), the two groups with the most rapid CDAI responses. The proportion of patients achieving a VAS pain score of ≤20 mm at Week 12 and 156, respectively, was smallest in Group 4 (7.7% and 29.7%) and Group 5 (6.9% and 32.4%) (Figure 1B), the groups with slower initial CDAI trajectories. Similarly, the proportion of patients who achieved a HAQ-DI score of ≤1 at Week 12 and Week 156, respectively, was greatest in Group 3 (88.2% and 90.9%), followed by Group 1 (65.1% and 83.1%), and lowest in Group 5 (29.3% and 62.2%) and Group 4 (40.4% and 64.9%) (Figure 1C).
Radiographic progression was low across all groups, with highest scores in Group 5 (slowest CDAI responders). Mean (SD) change from baseline in mTSS at Week 52 was 0.17 (0.94) in Group 1, 0.20 (0.93) in Group 2, 0.16 (0.77) in Group 3, 0.10 (0.99) in Group 4 and 0.52 (2.64) in Group 5. The incidences of TEAEs up to Week 52 of FINCH 1 and Week 156 of the LTE study were similar between the rapid and slow CDAI responders, except for serious infections, which showed a trend towards a lower occurrence in Groups 1 and 3 (rapid CDAI responders; 1.1–1.6 EAIR/100 PYE [Week 52] and 0.0–1.8 EAIR/100 PYE [Week 156]) vs the other groups (slower CDAI responders; 3.0–6.6 EAIR/100 PYE [Week 52] and 2.0–3.6 EAIR/100 PYE [Week 156]).
Conclusion: Of patients with RA with prior inadequate response to methotrexate treated with filgotinib 200 mg, those with the fastest improvements in CDAI score also generally showed the greatest improvements in VAS pain and HAQ-DI scores, with a high proportion of patients achieving a HAQ-DI score of ≤1, corresponding to mild disability. Highest radiographic progression was observed in patients with the slowest CDAI response, highlighting the potential benefits of achieving complete and sustained disease control to minimize structural damage. EAIRs of TEAEs were generally low, with small differences between groups, though trending towards lower rates of serious infection in rapid vs slow CDAI responders. Further characterization of the trajectory groups could not only support the management of patients’ expectations regarding speed of symptomatic benefit, but also facilitate identification of thresholds for early improvement as part of a more personalized approach to filgotinib treatment.
REFERENCES: [1] Combe B, et al. Ann Rheum Dis 2021;80:848–58.
[2] Taylor PC, et al. Ann Rheum Dis 2023;82:720–1.
Acknowledgements: We thank the physicians and patients who participated in these studies. The FINCH studies were co-funded by Gilead Sciences, Inc. (Foster City, CA, USA) and Galapagos NV (Mechelen, Belgium). This analysis was funded by Alfasigma S.p.A. (Bologna, Italy). Medical writing support was provided by Debbie Sherwood, BSc, CMPP (Aspire Scientific, Bollington, UK), and funded by Alfasigma S.p.A. Publication coordination was provided by Steve Winter, PhD, and funded by Alfasigma S.p.A. The authors acknowledge the contributions of Katrien Van Beneden and Hisaaki Isaji to the study.
Disclosure of Interests: Gerd R. Burmester Speaker’s bureau: AbbVie, BMS, Galapagos, Janssen, Lilly, Novartis, Pfizer, Sanofi and UCB, Consultant: AbbVie, BMS, Galapagos, Janssen, Lilly, Novartis, Pfizer, Sanofi and UCB, Bruno Fautrel Consultant: AbbVie, Amgen, Biogen, BMS, Celltrion, Chugai, Fresenius Kabi, Galapagos, Janssen, Lilly, Medac, MSD, Nordic Pharma, Novartis, Owkin, Pfizer, Roche, Sandoz, Sanofi-Genzyme, Sobi, UCB and Viatris, Grant/research support: AbbVie, Lilly, Pfizer and Sanofi-Aventis, Ly H. Le Consultant: Alfasigma S.p.A., Carole Van der Donckt Employee: Alfasigma S.p.A., Yoshiya Tanaka Speaker’s bureau: AbbVie, Asahi Kasei, Astellas, AstraZeneca, Boehringer Ingelheim, Chugai, Daiichi Sankyo, Eisai, Gilead, GSK, Lilly, Pfizer, Taisho and UCB, Grant/research support: Boehringer Ingelheim, Chugai and Taisho, Peter C. Taylor Consultant: AbbVie, Acelyrin Inc., Alfasigma S.p.A., Biogen, Fresenius Kabi, Gilead, Immunovant, Lilly, Moonlake, Nordic Pharma, Pfizer, Roche, Sanofi, Takeda and UCB, Grant/research support: Alfasigma S.p.A.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (