

Background: The mechanisms of muscle damage and wasting in systemic sclerosis (SSc) are multifactorial, including reduced physical activity, systemic inflammation, muscle inflammation, and nutritional deficits. Sarcopenia—characterized by muscle loss, weakness, and dysfunction—emerges as a common yet underrecognized concern, affecting up to 22% of SSc patients [1]. It is linked to reduced functional capacity, lower quality of life, and increased mortality. However, many patients with autoimmune rheumatic diseases (ARDs), like SSc, are at risk of progression without having developed definitive sarcopenia. Indeed, previous studies in other ARDs, such as systemic lupus erythematosus (SLE), show that even early, subclinical changes in muscle structure, assessed by ultrasound (US), are linked to key sarcopenia measures like reduced grip strength and decreased physical function. US is a promising, non-invasive tool for assessing muscle mass, quality, and stiffness in rheumatic diseases. It detects both quantitative (e.g., atrophy) and qualitative changes (e.g., increased echogenicity, indicating fibrosis or fat infiltration). Furthermore, shear wave elastography is being explored as a tool to assess muscle status in these patients. Despite its clinical significance, muscle involvement in SSc remains insufficiently studied. While US has shown potential in evaluating muscle changes in conditions like SLE and rheumatoid arthritis, its role in early muscle involvement in SSc requires further investigation.
Objectives: To explore muscle mass, quality, and stiffness using ultrasound in patients with SSc in comparison with healthy controls (HCs), and to examine the correlation between the ultrasound features and muscle strength, assessed using the handgrip (HG) strength test.
Methods: Consecutive patients with SSc and HCs were enrolled in this cross-sectional, multi-center study. Demographic and clinical data were collected for the SSc patients. Bilateral ultrasound scans of the quadriceps muscle (QM) were performed on SSc patients (without a history or current diagnosis of inflammatory myositis) and HCs, following a pre-determined protocol [2]. The scans were carried out to evaluate muscle mass (calculated as the sum of the thicknesses of the rectus femoris and vastus intermedius muscles), muscle quality (assessed via muscle echogenicity using a semi-quantitative modified Heckmatt scale [mHS] and grey-scale histogram analysis [GSA], processed with ImageJ software), and muscle stiffness (measured using shear-wave elastography [SWE]).
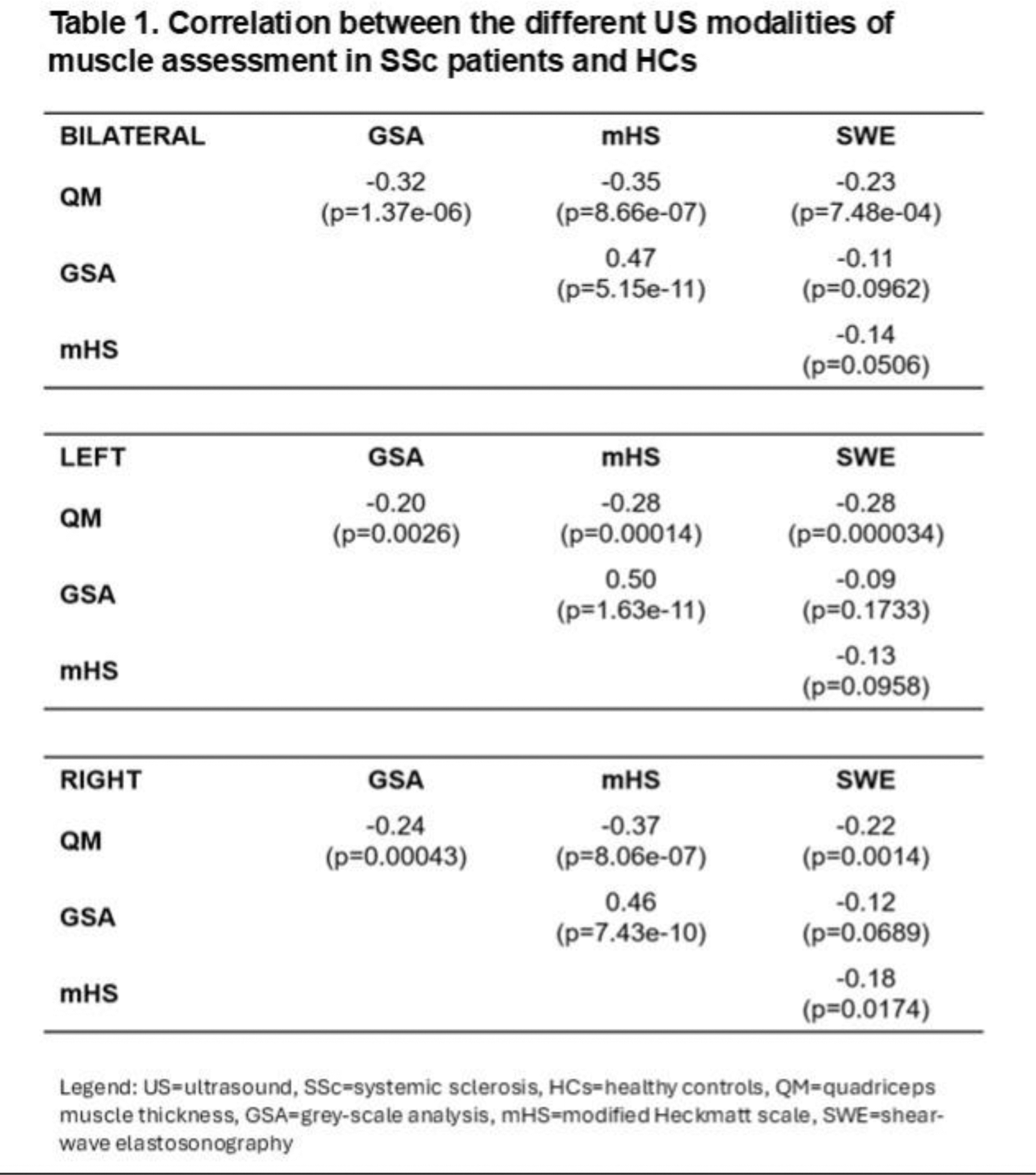
Results: In this study, we included 81 SSc patients and 24 HCs. SSc patients were significantly older than HCs, with a median age of 58 years (IQR 52, 67) versus 45 years (IQR 39, 54) (p<0.001). Their median BMI was also slightly higher (24.6 [IQR 21.8, 29.3] vs. 22.5 [IQR 20.6, 24.7]; p=0.050), although this difference was not significant after correcting for multiple comparisons (q=0.10). The median disease duration for SSc patients was 7 years (IQR 3, 11), with most having limited cutaneous SSc (74.1%) and a median modified Rodnan skin score (mRSS) of 3.0 (IQR 0, 8.0). A majority of patients (77.8%) had at least one major organ involvement. Fifteen SSc patients (18.5%) met the criteria for “probable sarcopenia” according to the European Working Group on Sarcopenia in Older People definition [3]. Additionally, SSc patients had significantly lower handgrip strength compared to HCs (27.4 ± 9.6 kg vs. 34.2 ± 9.6 kg, p=0.003). With respect to the ultrasound findings, SSc patients exhibited reduced muscle mass (QM thickness: 26.68 ± 7.37 mm vs. 31.24 ± 7.55 mm, p=0.009), lower muscle quality, indicated by increased muscle echogenicity (mHS: 1.5 [1, 2.5] vs. 0 [0, 0.5]; q<0.001; GSA: 93 [83, 108] vs. 71 [54, 82]; q<0.001), and reduced muscle stiffness (SWE: 1.60 [1.34, 1.86] vs. 1.71 [1.59, 1.85]; p=0.032). After adjusting for age, differences in mHS, GSA (p<0.001), and SWE (p=0.004) remained significant, while QM thickness did not (p=0.433). The relationships between the various ultrasound measurements (i.e., muscle mass, muscle echogenicity, and SWE) are reported in Table 1. In SSc patients, the HG strength test was positively correlated with QM thickness (ρ=0.412; p<0.001) and inversely correlated with mHS (ρ=-0.467; p<0.001) and GSA (ρ=-0.448; p<0.001). There was a statistical trend toward an inverse correlation between the HG strength test and SWE values (ρ=-0.186; p=0.07).
Conclusion: Ultrasound has proven to be an effective tool for evaluating muscle involvement in SSc, providing valuable information on muscle mass, quality, and stiffness, while also correlating with functional outcomes related to sarcopenia, such as handgrip strength. Early detection of muscle abnormalities in SSc patients (i.e., preferably before the onset of overt sarcopenia) could have significant implications for patient management (i.e., implementation of regular exercise, use of medications and supplements).
REFERENCES: [1] Tu X, et al. Sarcopenia in systemic sclerosis: prevalence and impact—a systematic review and meta-analysis. BMJ Open 2024.
[2] Di Matteo A, et al. Muscle involvement in systemic lupus erythematosus: multimodal ultrasound assessment and relationship with physical performance. Rheumatology (Oxford). 2022.
[3] Cruz-Jentoft AJ, et al. Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing. 2019.
Acknowledgements: NIL.
Disclosure of Interests: Riccardo Bixio GSK, Accord Healthcare, Stefano Di Donato: None declared, Maria Giovanna Lommano: None declared, Gianluca Smerilli: None declared, Sonia Farah: None declared, Seda Çolak: None declared, Marco Minerba: None declared, Francesca Pistillo: None declared, Richard J Wakefield: None declared, Fausto Salaffi: None declared, Ombretta Viapiana Gilead, Fresenius Kabi, Biogen, Ely-Lilly, UCB, AbbVie, MSD, and BMS, Rossella De Angelis: None declared, Maurizio Rossini AbbVie, Eli-Lilly, Italfarmaco, Neopharmed-Gentili, Theramex, UCB, Edoardo Cipolletta: None declared, Emilio Filippucci: None declared, Luca Idolazzi Eli Lilly, UCB Pharma, AbbVie, Amgen, and Johnson&Johnson outside the submitted work, Eli Lilly, UCB Pharma, and Boehringer Ingelheim, Francesco Del Galdo: None declared, Andrea Di Matteo Janssen.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (