

Background: Systemic Sclerosis (SSc) is a rare immune-mediated rheumatic disease, often manifesting in multiple organs [1]. Current SSc treatment can consist of several medications targeting individual organ-based manifestations, potentially leading to polypharmacy [2]. Polypharmacy is a term commonly applied to situations where a patient is prescribed five or more medications [3]. The World Health Organisation has described polypharmacy as a major and growing global healthcare issue, which has been associated with increased healthcare costs, increased risk of harm from adverse events and non-adherence to medication [4, 5]. However, there is limited research investigating the effects of polypharmacy in SSc [6].
Objectives: To describe polypharmacy in SSc and its influence on treatment adherence and satisfaction, from the patient and physician perspective across real-world clinical settings.
Methods: Data were drawn from the Adelphi Real World SSc Disease Specific Programme™, a cross-sectional survey of physicians actively managing patients with SSc in France, Germany, Italy, Japan, Spain, the United Kingdom and the United States of America from April – August 2022. Physicians reported data on patients’ demographics and clinical characteristics, SSc treatments and adherence to medication, as well as satisfaction with their patients’ current treatment regimen for consecutively consulting patients with SSc. Patients self-reported treatment satisfaction, and adherence to their current treatment regimen through the Adelphi Adherence Questionnaire (ADAQ), a validated patient-reported outcome measure of medication adherence scored on a scale of 0 (completely adherent) to 4 (completely non-adherent). Data for body areas affected by SSc, treatment adherence and satisfaction were stratified by the number of SSc medications patients were prescribed [1, 2, 3, 4], with patients prescribed five or more forming a single group (5+). Differences across these groups were measured using Chi-squared, Fisher’s exact and ANOVA statistical tests.
Results: Rheumatologists (n=325) and internists (n=17) reported data for 1820 patients with SSc, of whom 680 voluntarily self-reported data. The average age of patients with SSc was 53.4 years and 81% were female (Table 1). Disease characteristics can be seen in Table 1. The mean (standard deviation; SD) number of body areas affected by SSc differed significantly across our groups (p<0.001), ranging from 2.5 (2.0) in patients prescribed a single medication to 6.1 (3.3) in patients prescribed five or more medications. The mean (SD) number of medications prescribed for the treatment of SSc was 3.4 (2.2), with 16% of patients prescribed a single medication versus 23% prescribed five or more (Table 1). The three most common medications prescribed were calcium channel blockers (49%), proton pump inhibitors (38%) and methotrexate (24%) (Table 1). Physician-reported treatment adherence differed significantly across groups (p<0.001), ranging from 74% of patients prescribed a single medication to 45% of those prescribed five or more medications described as being ‘completely adherent’ (Figure 1a). Physician-reported treatment satisfaction rates differed significantly across groups (p<0.001), ranging from 85% satisfaction for patients prescribed one medication to 47% satisfaction for patients prescribed five or more (Figure 1c). Mean (SD) ADAQ score also differed significantly across groups (p<0.001), ranging from 0.29 (0.37) for those prescribed one medication to 0.54 (0.55) for those prescribed four (Figure 1b). Patient-reported satisfaction rates also differed significantly across groups (p<0.001), ranging from 85% satisfaction for patients prescribed one medication to 48% satisfaction for patients prescribed five or more (Figure 1d).
Conclusion: Treatment adherence and satisfaction levels were lower for patients prescribed five or more medications than those prescribed a single medication to treat their SSc. Patients with SSc would benefit from the approval of systemic treatments that target multiple manifestations, reducing the burden of polypharmacy and improving treatment adherence.
REFERENCES: [1] Denton CP, Khanna D. Systemic sclerosis. The Lancet. 2017 Oct 7;390(10103):1685-99.
[2] Pope JE, Denton CP, Johnson SR, Fernandez-Codina A, Hudson M, Nevskaya T. State-of-the-art evidence in the treatment of systemic sclerosis. Nature Reviews Rheumatology. 2023 Apr;19(4):212-26.
[3] Masnoon N, Shakib S, Kalisch-Ellett L, Caughey GE. What is polypharmacy? A systematic review of definitions. BMC geriatrics. 2017 Dec;17:1-0.
[4] World Health Organization, 2019. Medication safety in polypharmacy: technical report (No. WHO/UHC/SDS/2019.11). World Health Organization.
[5] Hajjar ER, Cafiero AC, Hanlon JT. Polypharmacy in elderly patients. The American journal of geriatric pharmacotherapy. 2007 Dec 1;5(4):345-51.
[6] Boukhlal S, Chouchana L, Saadi M, Casadevall M, Cohen P, Dunogue B, Murarasu A, Regent A, Mouthon L, Chaigne B. Polypharmacy, drug-drug interactions, and adverse drug reactions among systemic sclerosis patients: A cross-sectional risk factor study. In Seminars in Arthritis and Rheumatism 2024 (Vol. 67, p. 152469).
Table 1. Patient demographics, disease characteristics and current treatments.
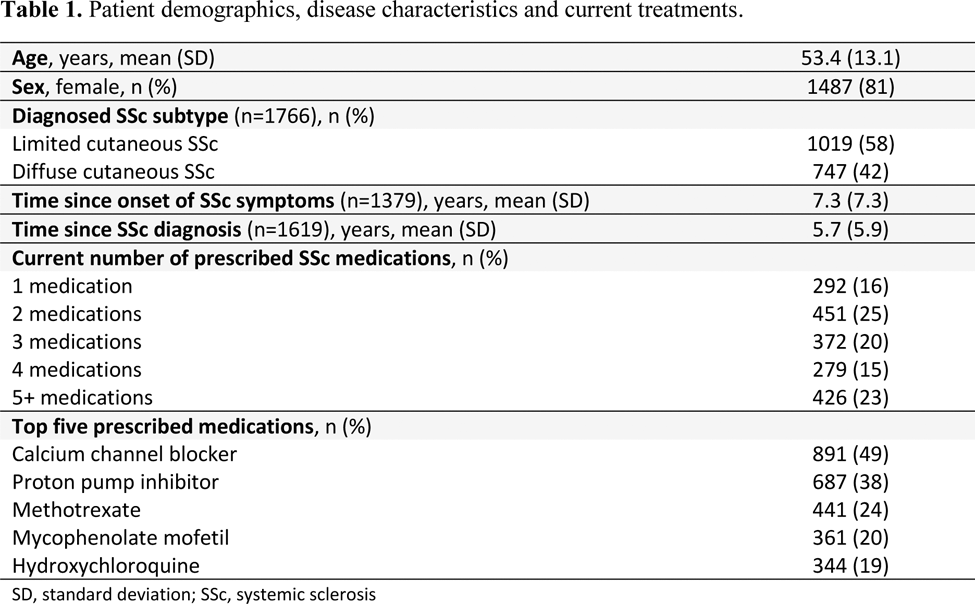
Physician and patient-reported treatment adherence and satisfaction stratified by number of prescribed SSc medications.
Acknowledgements: NIL.
Disclosure of Interests: Dan Twigg is an employee of Adelphi Real World, Isabel Truman is an employee of Adelphi Real World, Naomi Brown is an employee of Adelphi Real World, Sophie Barlow is an employee of Adelphi Real World.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (