

Background: Tocilizumab (TCZ) is effective in systemic sclerosis-interstitial lung disease (SSc-ILD) patients with a potential benefit also on skin fibrosis.
Objectives: We evaluated the multi-domain safety and efficacy of TCZ in a real-life setting from an Italian-French multicenter cohort.
Methods: Patients with SSc treated with TCZ were retrospectively evaluated at 12 months prior to TCZ introduction; at baseline; at 12, and 24 months after TCZ. The following parameters were recorded: modified Rodnan skin score (mRSS), pulmonary function tests, DAS28-CRP, troponin and NTproBNP levels, ejection fraction (EF) and Health Assessment Questionnaire (HAQ). Predictors of TCZ withdrawal for inefficacy were identified.
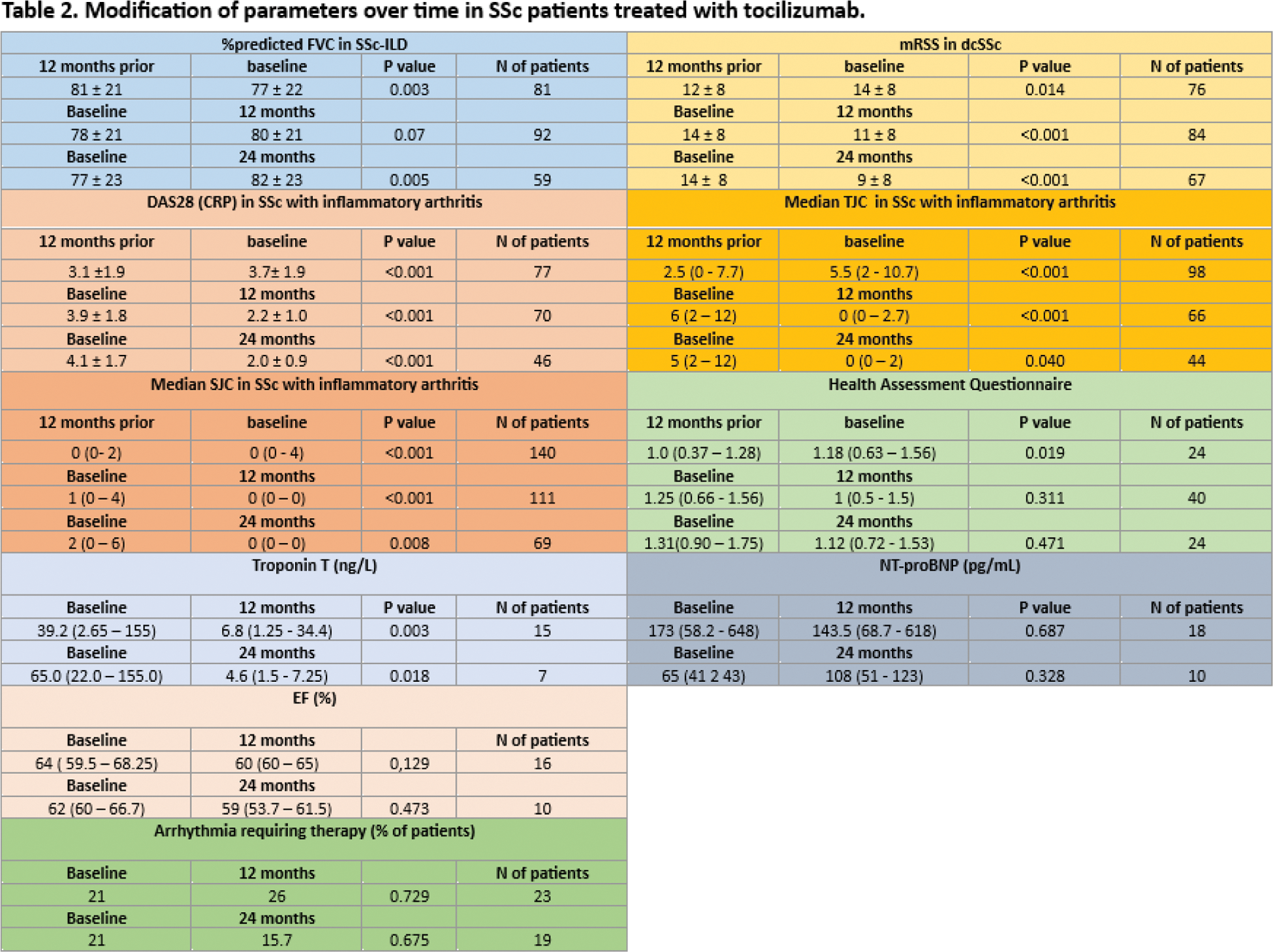
Results: 197 SSc patients treated with TCZ (88% female), median age 57(46-69) years, median disease duration 9(4-15) years, were included. Baseline features, concomitant therapies are summarized in Table 1. Reasons for TCZ introduction were persistent inflammatory arthritis in 129 (65%) patients, ILD worsening in 65 (33%) patients, skin worsening in 63 (32%) patients and myocardial involvement in 20 (10%) patients. Among SSc-ILD patients (81 patients), in the 12 months prior to TCZ introduction, %pFVC showed an overall significant decline (p=0.003) and in 36 (44%) patients a %pFVC decline ≥5 was observed. Upon TCZ, a stabilization in %pFVC was observed at 12 months (p=0.07) (Table 2), with a decrease to 22% (p=0.001) in the rate of progressors. Among those with available PFTs at the 3 timepoints, the number of progressors went from 43% to 24%, p=0.015. An improvement in %pFVC was observed (Table 2) at 24 months (p=0.005). The number of progressors from month 12 to month 24 was 19%, p=0.680. Among dcSSc patients (124), upon TCZ, a significant reduction in mRSS was observed both at month 12 and 24 (Table 2). The % of patients with digital ulcers was stable at baseline compared to 12 months prior TCZ (p=0.99), then decreased from 24% to 13% at month 12 (p=0.026) and to 8,7% from baseline to month 24 (p=0.001). In patients with arthritis, DAS28-CRP showed a significant decline (Table 2) both at month 12 and 24. This was paralleled by a reduction in TJC and SJC. Among patients with myocardial involvement, troponin levels reduced over time, together with a stabilization in NT-proBNP levels after TCZ. A stabilization was also seen in %EF and in the % of patients with arrhythmias requiring therapy (Table 2). The most common adverse events were infections (22.8%). TCZ was suspended in 63 patients (32%) after a median time of 8(4-22) months, the main reason being inefficacy (41%). 57(29%) patients were on TCZ monotherapy. The most common combination therapy was with methotrexate (35%), followed by mycophenolate (25%). At multivariate analysis the presence of PAH (5.313[1.431–19.724]) was associated with TCZ stop due to inefficacy. Conversely, raised baseline CRP (0.108[0.023–0.505]) were protective against TCZ stop due to inefficacy. No differences were found in the rate of inefficacy nor in the side effects when considering TCZ mono versus combination therapy (10% versus 14.7%, p=0.494; 20.7% versus 24.4%, p=0.709).
Conclusion: Our real-life data support the use of TCZ in SSc patients, also in combination with other immunosuppressants, both in early progressive forms and in advanced stages, in multiple organ involvements. Raised baseline CRP can predict TCZ efficacy. Infectious adverse events leading to drug suspension were limited.
REFERENCES: NIL.
Clinical and demographic characteristics of our multicenter cohort of 197 SSc patients.
| Clinical and demographic characteristics | Previous Therapies | ||
|---|---|---|---|
| Females, n (%)
| 168 (88)
| Methotrexate, n (%)
| 115 (58)
|
| Organ involvement , n (%) | Concomitant Therapies | ||
| Interstitial lung disease
| 139 (71)
| Corticosteroids, n (%)
| 137 (71)
|
N= number; Ab= antibodies; ILD= interstitial lung disease; GERD= gastro-esophageal reflux disease.
Acknowledgements: NIL.
Disclosure of Interests: Corrado Campochiaro Novartis, J&J, BI, Giulia Buonsante: None declared, Giacomo De Luca BI, J&J, Elena Marazzi: None declared, Fabio Cacciapaglia: None declared, Maria Grazia Lazzaroni: None declared, Marco de Pinto: None declared, Devis Benfaremo: None declared, Valentina Longo: None declared, Carlo Iandoli: None declared, Giovanna Cuomo: None declared, Anna Cuberli: None declared, Elisabetta Zanatta: None declared, Alain Lescoat: None declared, Silvia Bellando-Randone: None declared, Giorgia Trignani: None declared, Serena Guiducci: None declared, Lorenzo Beretta: None declared, Gianluca Moroncini: None declared, Dilia Giuggioli: None declared, Paolo Airò: None declared, Florenzo Iannone: None declared, Veronica Codullo: None declared, Silvia Laura Bosello: None declared, Nicoletta Del Papa: None declared, Marie-Elise Truchetet: None declared, Jerome Avouac: None declared, Lorenzo Dagna: None declared, Marco Matucci-Cerinic Novartis, BI, J&J, Actelion Pharmaceuticals, Biogen, Bayer, Boehringer Ingelheim, CSL Behring, Eli Lilly, Yannick Allanore Abbvie, Alpine ImmunoSciences, Argenx, Astra-Zeneca, Boehringer Ingelheim, Corvus, Merck Serono, Topadur, Abbvie, Alpine ImmunoSciences, Argenx, Astra-Zeneca, Boehringer Ingelheim, Corvus, Merck Serono, Topadur.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (