

Background: Diagnosing EGPA is challenging due to its rarity, multisystem involvement, and common onset with unspecific symptoms like asthma [1], which often delays diagnosis and treatment, increasing the risk of organ damage [2]. Early identification is crucial to improve patient outcomes [3].
Objectives: EGPAware is a multidisciplinary Delphi study designed to reach a consensus among a large sample of European experts. Its goal was to develop a checklist of clinical features that raise suspicion of EGPA, assisting clinicians in early recognition of this disease.
Methods: EGPAware is a Delphi study led by a steering committee of global EGPA experts, formed by 4 rheumatologists, 2 internal medicine specialists, 2 pulmonologists, a nephrologist and an immunologist from 7 European countries. The Delphi questionnaire had three sections: 1) panellists’ profile; 2) current clinical management; and 3) red flags for EGPA suspicion (in adult patients with asthma and blood eosinophilia (>1000 cells/µL or >500 cells/µL, if they have previously received any medication that may have altered the blood eosinophil count). The committee defined the profiling questions and listed the proposed red flags. Consensus was determined when ≥70% of participants agreed (or disagreed) on a statement. Statements with <70% consensus in the first round were asked again in a second round. In round two, an additional section was included for panellists to separately rank the 10 most pertinent signs based on two criteria: clinical relevance (considering their practical importance and potential to influence clinical decision-making), and frequency of observation (based on their clinical experience). Data was analysed using descriptive statistics.
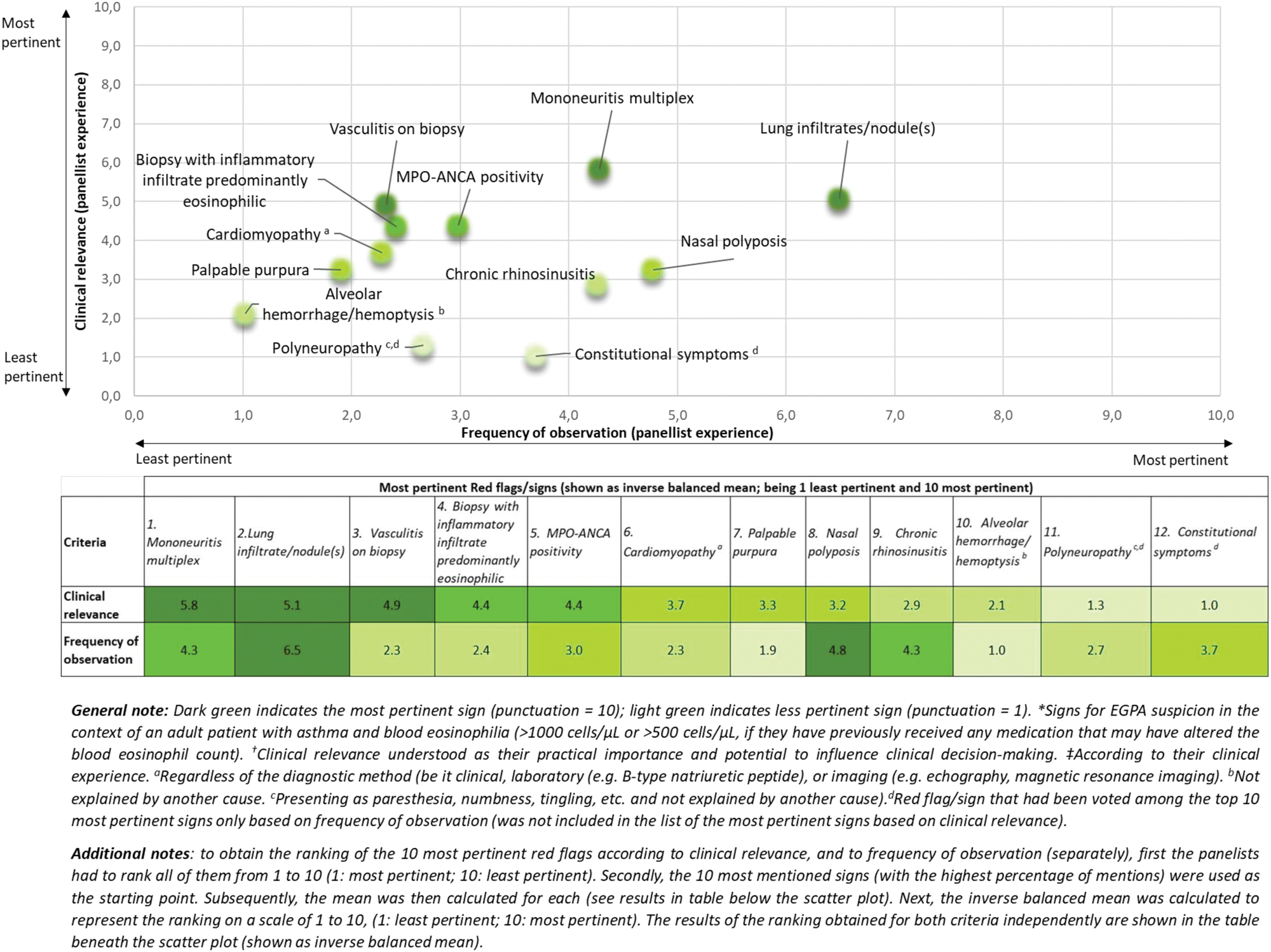
Results: A broad group of 53 EGPA-treating physicians from 15 different European countries completed the two rounds of the Delphi survey. The panel consisted mainly of rheumatologists (35.8%) and pulmonologists (30.2%), followed by internal medicine physicians (11.3%), immunologists (9.4%), nephrologists (5.7%), allergologists (5.7%) and a dermatologist (1.9%). Half of them reported having more than 15 years of experience managing EGPA. In addition, all participants indicated employing in a multidisciplinary approach to patient management and being actively involved in every phase of the EGPA patient journey. Consensus on agreement was reached on 25 out of 47 proposed red flags for EGPA suspicion, including mononeuritis multiplex (98.1% ); lung infiltrate/nodule(s ) (92.5%); alveolar haemorrhage (79.2%); retinal vasculitis (73.6%), and episcleritis/scleritis (71.7%). General signs such as constitutional symptoms (weight loss, fever, fatigue, etc.] (79.2%), and a specific vascular feature, digital ischaemia (73.6%), also achieved consensus. Of note, typical features of EGPA such as alveolar hemorrhage and digital ischemia easily reached consensus despite their rarity whilst frequent, non-specific findings such as proteinuria did not. Contrary, consensus on agreement was not achieved for signs such as proteinuria (>550mcg/24 hours), (67.9%), nor elevated creatinine with or without active urine sediment (haematuria, proteinuria, cellular casts), (66.0%), despite these being part of the diagnosis of glomerulonephritis, which itself achieved high consensus as a sign for suspicion (89.4%). Other signs like cardiomegaly (60.4%), chronic diarrhoea (18.9%) (both not explained by another cause), and venous thrombosis (54.7%) also failed to reach consensus, likely due to their nonspecific presentation. However, in the context of a patient with asthma and blood eosinophilia, these signs could serve as potential warning indicators to rule out other conditions. The most important red flags ranked by clinical relevance included mononeuritis multiplex; lung infiltrates/nodules; vasculitis on biopsy; and MPO-ANCA positivity (Table 1). According to the panellists, the most frequently observed red flags in their clinical experience were lung infiltrates/nodules, nasal polyposis, and mononeuritis multiplex. The results of the prioritization, considering both variables, are shown in Figure 1 .
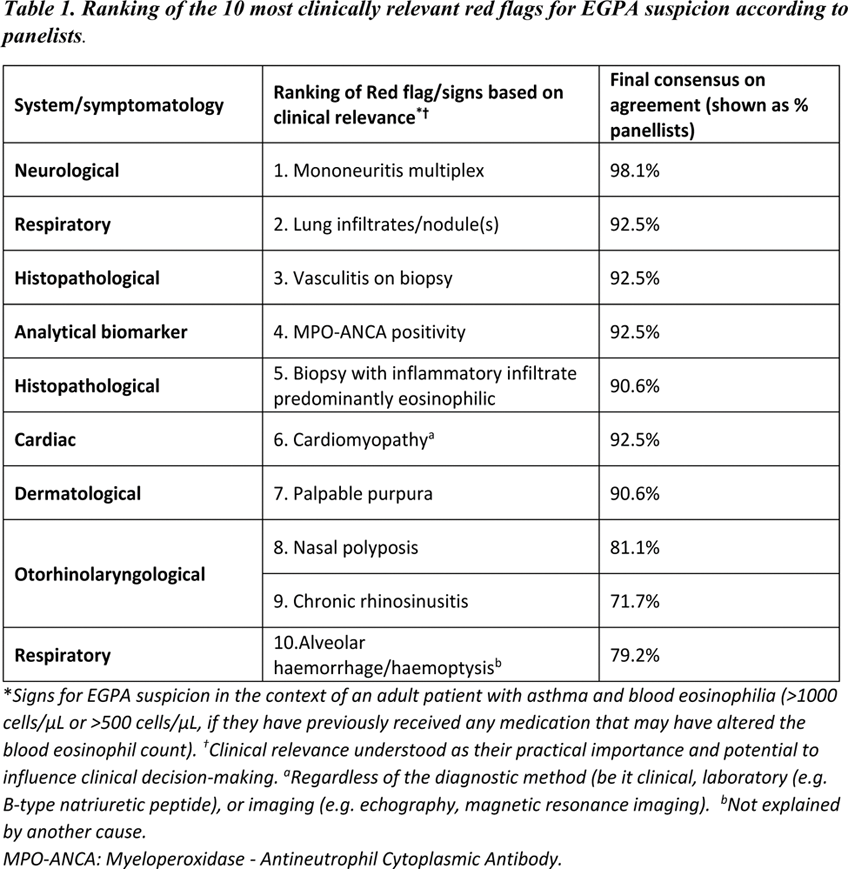
Scatter plot of the most pertinent Red Flags according both to clinical relevance *† and frequency of observation ‡ for EGPA suspicion.
Conclusion: EGPAware is the first European study to provide a checklist of red flags, developed with input from a multidisciplinary panel of EGPA-treating physicians, to aid clinicians in identifying potential EGPA cases and, therefore, improve referral and treatment timelines. The EGPAware study reveals variability in how physicians prioritize red flags, emphasizing the need for unified training and a multidisciplinary approach to EGPA management.
REFERENCES: [1] Emmi, G. et al . (2023). Nat Rev Rheumatol, 19.
[2] Jakes, RW, et a l. (2024). ERJ Open Res, 10(4).
[3] Berden, A, et al . (2012) BMJ, 344:e26.
Acknowledgements: Authors would like to acknowledge GSK who provided funding and reviewed a draft of the abstract for data accuracy only, but the final content is the authors’ sole work. Authors would like to thank all the panellists who participated in the Delphi process without which this project could not have been carried out. Also, the authors would like to thank Nerea Toro, Ana Fernández and Marta Barragán at Adelphi Targis, for their support implementing the project.
Disclosure of Interests: Bernhard Hellmich AbbVie, Amgen, AstraZeneca, Boehringer Ingelheim, Bristol Myers Squibb, Chugai, GSK, InflaRx, Janssen, MSD, Novartis, Pfizer, Phadia, Roche, and CSL Vifor, AbbVie, Amgen, AstraZeneca, Boehringer Ingelheim, Bristol Myers Squibb, Chugai, CSL Vifor, GlaxoSmithKline, InflaRx, Janssen, MSD, Novartis, Pfizer, Phadia, Roche, AbbVie, Amgen, AstraZeneca, Boehringer Ingelheim, Bristol Myers Squibb, Chugai, GSK, InflaRx, Janssen, MSD, Novartis, Pfizer, Phadia, Roche, and CSL Vifor, AbbVie, Amgen, AstraZeneca, Boehringer Ingelheim, Bristol Myers Squibb, Chugai, CSL Vifor, GlaxoSmithKline, InflaRx, Janssen, MSD, Novartis, Pfizer, Phadia, Roche, Apostolos Bossios AstraZeneca, Chiesi and GlaxoSmithKline, AstraZeneca, Chistian Domingo-Rivas ALK-Abelló, AstraZeneca, Chiesi, Esteve, GlaxoSmithKline, Menarini, Novartis and Sanofi, Giacomo Emmi AstraZeneca, CSL Vifor, GlaxoSmithKline, Novartis, SOBI, AstraZeneca, CSL Vifor, GlaxoSmithKline, Novartis, SOBI, Cristina Ponte AbbVie, AstraZeneca, CSL Vifor, GlaxoSmithKline, Novartis and Roche, AbbVie, AstraZeneca, CSL Vifor, GlaxoSmithKline, Novartis and Roche, Maria Roser Solans-Laque GlaxoSmithKline, Íñigo Jesus Rúa-Figueroa GlaxoSmithKline, Augusto Vaglio: None declared, Dimitrios Vassilopoulos Abbvie, Aenorasis, Genesis – Pharma, GlaxoSmithKline, Janssen, Lilly, Novartis, Pfizer, Sobi, UCB, Abbvie, Aenorasis, Genesis – Pharma, GlaxoSmithKline, Janssen, Lilly, Novartis, Pfizer, Sobi, UCB, Gabriele Brembilla Employee and holds financial equities in GlaxoSmithKline, Nadia Kallinikou Employee and holds financial equities in GlaxoSmithKline, Benjamin Terrier Amgen, AstraZeneca, Boehringer Ingelheim, CSL Vifor, GlaxoSmithKline, Janssen, and Novartis, Amgen, AstraZeneca, Boehringer Ingelheim, CSL Vifor, GlaxoSmithKline, Janssen, and Novartis.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (