

Background: Patients with systemic lupus erythematosus (SLE) and lupus nephritis (LN) are at increased risk of infection, particularly respiratory infections including COVID-19 [1–3]. Demographic factors, comorbidities and medication use are related to COVID-19 severity in patients with SLE; however, there is limited evidence beyond the initial pandemic years [4–6].
Objectives: To describe patient characteristics, comorbidities and medication use amongst patients with SLE (overall and according to LN status) with COVID-19, and to estimate the frequency and rate of severe COVID-19 amongst patients with SLE in the US.
Methods: This observational, retrospective, nationwide cohort study used electronic health records (EHRs) of patients with SLE (overall and according to LN status) with healthcare encounters between 1 January 2000 and 23 April 2024. Deidentified patient EHRs arose from a network of 68 US healthcare organisations that included 118 million individuals. Index date was considered as the first SARS-CoV-2 viral positive test occurring between 1 February 2020 and 31 January 2024. Adult patients had an SLE or LN diagnosis at least 90 days pre-index and EHR activity in the 365 days pre-index. The following cohorts were identified: SLE regardless of LN status, SLE with LN (hereafter referred to as LN patients) and SLE without LN. Patients with SLE had ≥2 outpatient visits ≥7 days apart or ≥1 inpatient visit with an SLE diagnosis (ICD-9-CM 710* or ICD-10-CM M32*). LN patients had ≥2 outpatient visits at least 7 days apart or ≥1 inpatient visit with an LN diagnosis (ICD-10-CM M32.14 or ICD-9-CM 710*, or ICD-10-CM M32* and ICD-9-CM 580-583 excluding 581.2, 582.2, 583.2, 583.6 and 583.7 on or after the SLE diagnosis). SLE without LN patients were those in the SLE cohort who did not have LN as of the index date. Patient sociodemographic characteristics, comorbidities and COVID-19 vaccination history were assessed. End-stage kidney disease (ESKD) and extra-renal organ involvement due to SLE were identified between 1 Jan 2000 to 30 days pre-index. Hydroxychloroquine, systemic glucocorticoid steroids (oral or intravenous) and immunosuppressants were assessed between 1 January 2020 to 30 days pre-index. COVID-19 antivirals were assessed 30 days post-index. Primary analyses identified patients who had a hospitalisation that occurred within 14 days post-index and had a COVID-19 diagnosis in the first 3 days of admission. Frequency and incidence of ≥1 COVID-19 hospitalisation amongst those with SLE (overall and according to LN status) were calculated overall and by calendar time. COVID-19 hospitalisation rate per 1000 person days (pd) was estimated by following each cohort from 1 February 2020 until the last encounter, 14 days post-index, end of the study period or death, whichever occurred first. Additional analyses estimated the frequency and rate per 1000 pd of a) intensive care unit (ICU) stay, ventilation or respiratory failure and b) all-cause mortality in the 30 days post-index. COVID-19 hospitalisation rates were stratified according to immunosuppressant use, extra-renal organ involvement and ESKD. Additionally, COVID-19 hospitalisations per 100,000 patients were calculated over time to assess trends amongst the study cohorts relative to available COVID-NET surveillance data for the adult general population (Figure 1).
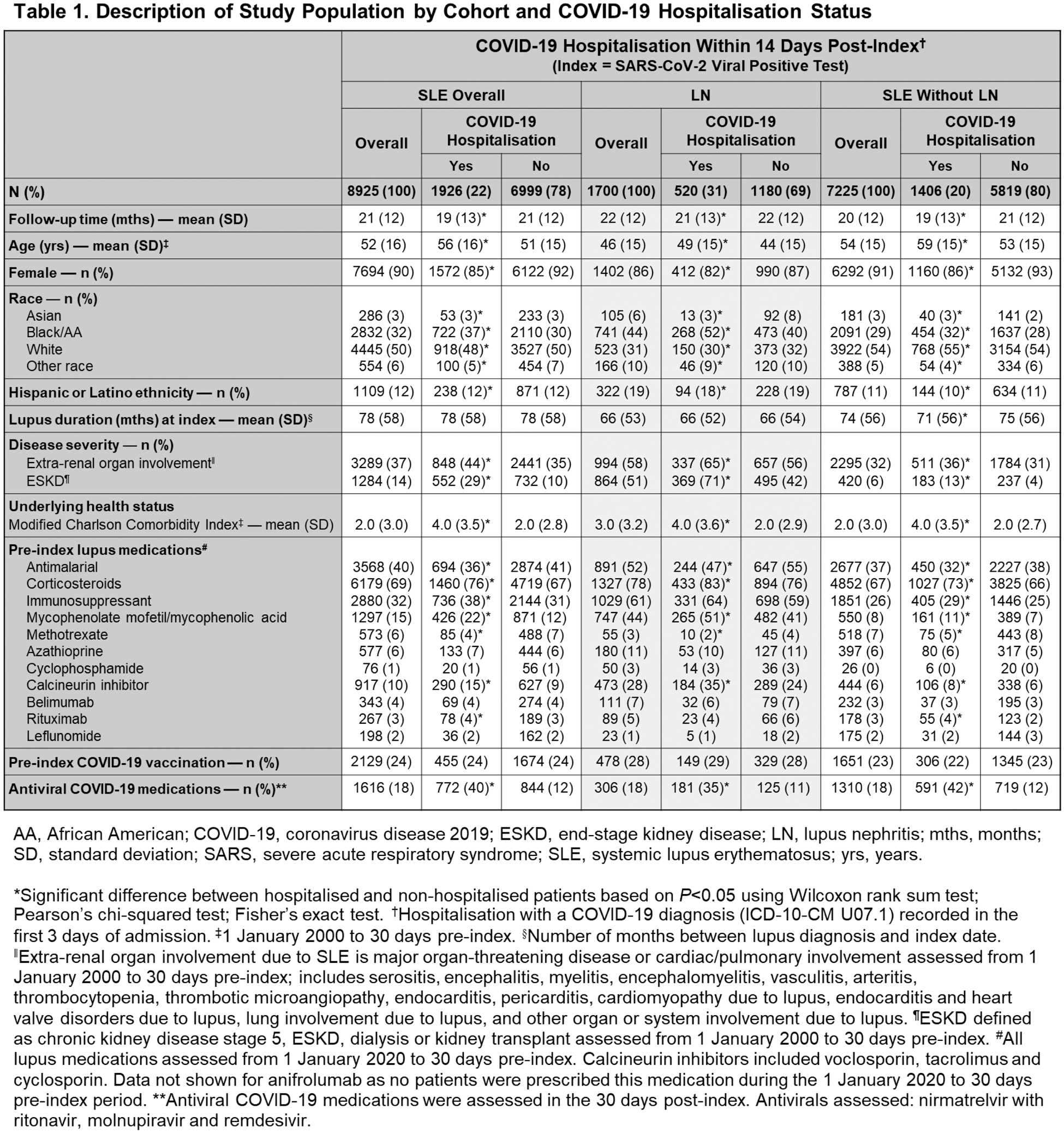
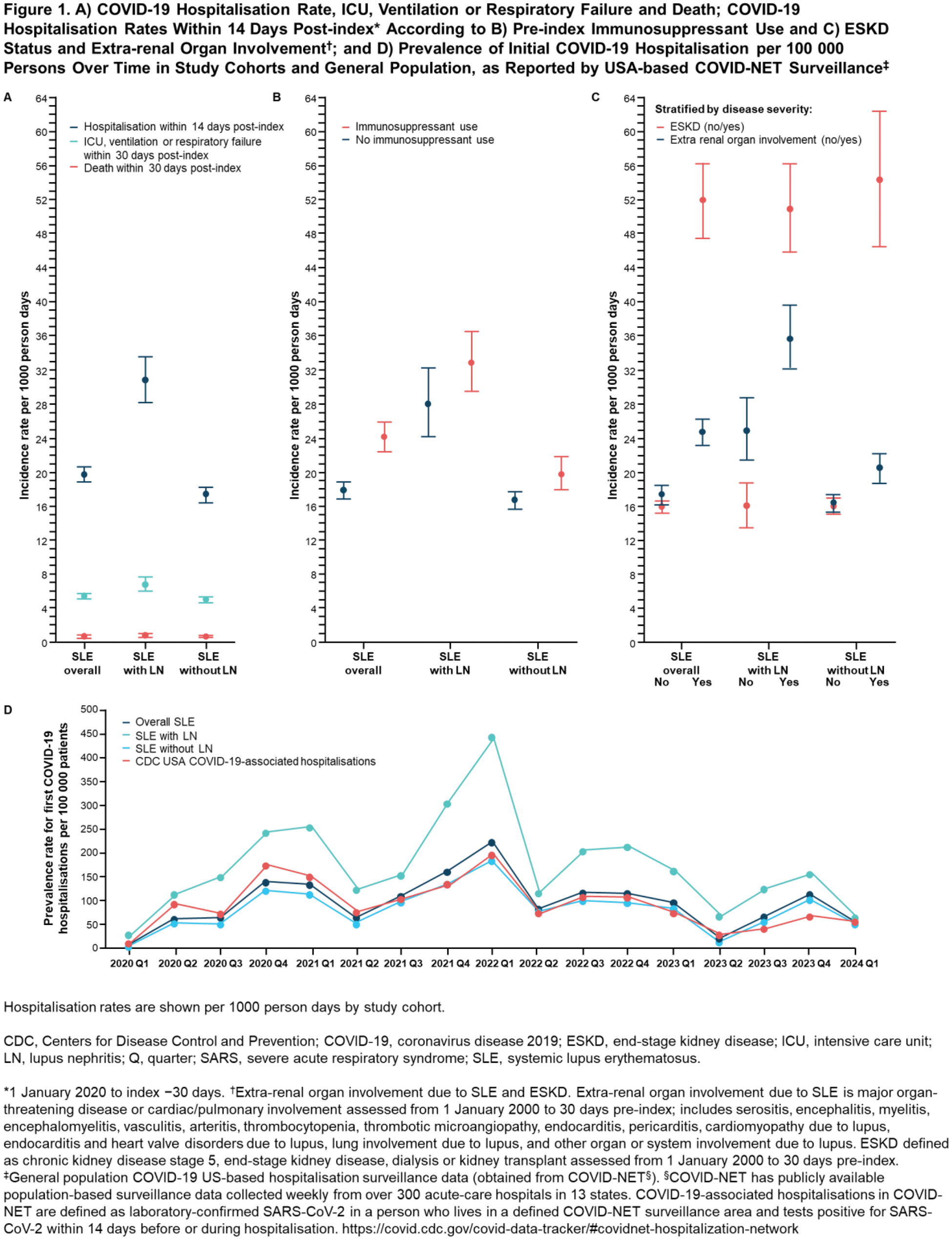
Results: Study cohorts are described overall and according to COVID-19 hospitalisation in Table 1. LN patients were younger and more racially and ethnically diverse than SLE patients. A minority of patients were prescribed antivirals within 30 days post-index. COVID-19 hospitalisations were significantly higher in the following groups of patients: LN, severe (particularly ESKD) or comorbid disease, receiving immunosuppressants (Table 1), males, >65 years, Black and non-Hispanic. COVID-19 hospitalisations were significantly lower in patients prescribed antimalarials and methotrexate. Vaccinations outside of the healthcare system were not observable and the low coverage likely underestimated true COVID-19 vaccination status. Figure 1A shows COVID-19 hospitalisation, ICU/ventilation/respiratory failure and mortality rates for each cohort. Figures 1B and 1C show cohort-specific COVID-19 hospitalisation rates according to immunosuppressant use and disease severity (major organ involvement other than LN and ESKD). Temporal trends in COVID-19 hospitalisations were similar to the general population based on US surveillance data (Figure 1D). ICU stay, ventilation or respiratory failure occurred in 13.7%, 17.1% and 12.9% for SLE, LN and SLE without LN patients respectively. All-cause mortality occurred in 2.0%, 2.4% and 1.9% for SLE, LN and SLE without LN patients respectively.
Conclusion: SLE patients with LN, ESKD and severe lupus due to extra-renal organ involvement had elevated rates of COVID-19 hospitalisation. Immunosuppressant use was associated with significantly elevated rates of COVID-19 hospitalisation; few patients received antivirals. COVID-19 trends identified in the SLE study cohorts were similar to national general population surveillance data. Planned work will assess rates of severe COVID-19 via multivariable modelling.
REFERENCES: [1] Bruera S, et al. Lupus Sci Med . 2023;10:e000750.
[2] Huang WN, et al. Rheumatol Ther . 2023;10:387-404.
[3] Tektonidou MG, et al. Arthritis Care Res (Hoboken). 2015;67:1078-1085.
[4] Calabrese C, et al. RMD Open . 2023;9:e003250.
[5] Magder LS, et al. Lupus Sci Med . 2023;10:e000961.
[6] Ugarte-Gile MF, et al . Ann Rheum Dis . 2022;81:970-978.
Acknowledgements: Funded by F. Hoffmann-La Roche Ltd. Editorial assistance was provided by Nucleus Global, an Inizio company, and funded by F. Hoffmann-La Roche Ltd.
Disclosure of Interests: Lisa Lindsay shareholder of F. Hoffmann-La Roche Ltd., employee of Genentech, Inc., Huong Trinh shareholder of F. Hoffmann-La Roche Ltd, employee of Genentech, Inc., Shu Wang employee of Genesis Research LLC., William F. Pendergraft III shareholder of F. Hoffmann-La Roche Ltd., employee of Genentech, Inc., Petra Kirchner shareholder of F. Hoffmann-La Roche Ltd, employee of F. Hoffmann-La Roche Ltd, Jinoos Yazdany received consulting fees from AstraZeneca, Pfizer and UCB.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (