

Background: Filgotinib (FIL), a selective Janus Kinase (JAK)1 inhibitor, has shown efficacy in rheumatoid arthritis (RA) in clinical trials, but real-world data remain limited. This study aimed to evaluate the effectiveness of FIL compared to other modes of action in a diverse Belgian RA population.
Objectives: This study aimed to evaluate the effectiveness of FIL compared to other modes of action in a diverse Belgian RA population.
Methods: Patients were included from the electronic platform “Tool for Administrative Reimbursement Drug Information Sharing” (TARDIS), which collects data on all Belgian RA patients undergoing biologic and targeted therapy. This platform is utilized during the submission of requests for initiation or continuation of reimbursement for these therapies. Adult patients (≥18 years) with a Disease Activity Score 28 (DAS28) >3.7, initiating FIL or a comparator biologic DMARD (bDMARD) treatment, were selected between August 2021 and December 2024. No other JAKi were included. Inclusion required attendance at a first follow-up visit, scheduled within 3 months for FIL or 6 months for bDMARDs, as recorded in TARDIS. Effectiveness was evaluated based on the proportion of patients achieving the following outcomes: remission (DAS28-CRP <2.6, or DAS28-ESR if DAS28-CRP was unavailable), low disease activity (LDA, DAS28 ≤3.2), Health Assessment Questionnaire Disability Index (HAQ-DI) ≤0.25 (HAQ25), HAQ-DI ≤0.5 (HAQ50), and minimum clinically important difference in HAQ-DI (MCID_HAQ), defined as a decrease of ≥0.22 from baseline. Logistic regression was used to adjust for potential confounders. Analyses were stratified by treatment line, defined as first (= advanced treatment-naive), second, and third (or more) line treatment. Analysis was performed in SAS Enterprise Guide and Python.
Results: In total, 816 patients on FIL and 5542 patients on bDMARDs were included. These populations were comparable on baseline demographics (Table 1). All effectiveness outcomes were higher in the FIL group compared to the bDMARD group (Figure 1). Logistic regression models per treatment line adjusted for baseline age, sex, disease duration, joint counts, PGA, CRP, and ESR confirmed these results (Figure 2).
Baseline characteristics of TARDIS patients.
| FIL population | bDMARD population | |
|---|---|---|
| Number | 816 | 5542 |
| Current Treatment | ||
| FIL | 816 (100%) | - |
| Abatacept | 453 (9.3%) | |
| Adalimumab | 1829 (33.0%) | |
| Certolizumab | 365 (6.6%) | |
| Etanercept | 1544 (27.9) | |
| Golimumab | 174 (3.1%) | |
| Infliximab | 116 (2.1%) | |
| Sarilumab | 354 (6.4%) | |
| Tocilizumab | 708 (12.8%) | |
| Age (mean ±SD, years) | 58.6 ±23.3 | 58.7 ±11.5 |
| Disease duration (mean ±SD, years) | 7.0 ±14.7 | 6.7 ±6.9 |
| Sex (women, n (%)) | 601 (73.6%) | 3741 (67.5%) |
| ESR (mean ±SD, mm/hour) | 24.0 ±33.6 | 26.9 ±18.6 |
| CRP (mean ±SD, mg/L) | 13.8 ±32.1 | 14.6 ±16.7 |
| SJC28 (mean ±SD) | 6.1 ±7.2 | 6.2 ±3.6 |
| TJC28 (mean ±SD) | 8.8 ±9.7 | 8.6 ±4.5 |
| DAS28 (mean ±SD, years) | 4.9 ±1.5 | 4.9 ±0.7 |
| HAQ-DI (mean ±SD, 0-3)* | 1.5 ±1.1 | 1.6 ±0.5 |
| PGA (mean ±SD, 0-100) | 66.5 ±33.1 | 67.4 ±15.5 |
| Advanced Therapy Naive, yes | 459 (56.3%) | 4474 (80.7%) |
| Advanced Therapy Experienced, yes | 358 (43.7%) | 1068 (19.3%) |
| 2nd line | 143 (17.5%) | 602 (10.9%) |
| 3th line + | 214 (26.2%) | 467 (8.4%) |
Legend: Number given are mean ± SD or number, proportion. JAKi = Janus Kinase inhibitor, HAQ= health assessment questionnaire, PGA= Patient Global assessment; CRP= C-reactive protein; ESR= erythrocyte sedimentation rate; TJC= tender joint count; SJC= Swollen joint Count; DAS28 = disease activity score based on the 28joints. *Missing HAQ(0-3) scores were imputed by regression using age and HAQ(0-60) scores.
Effectiveness outcomes of FIL versus bDMARDs per treatment line.
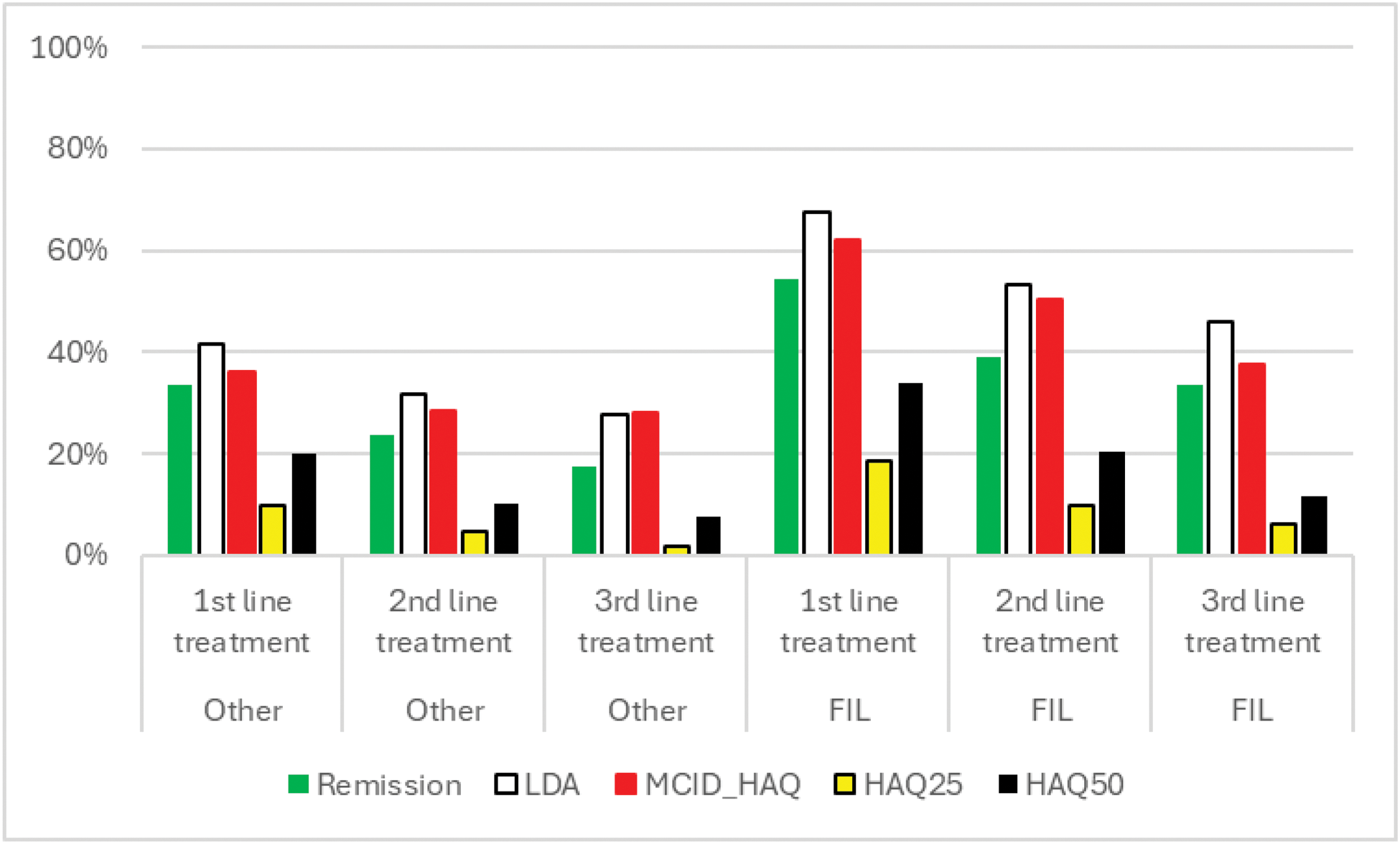
Legend: remission (DAS-28 <2.6), LDA - low disease activity (DAS-28 ≤3.2), HAQ-DI≤0.25 (HAQ25) and minimum HAQ-DI-decrease (MCID_HAQ) since baseline of 0.22.
Forest Plot of Odds Ratios By Treatment Line of FIL versus other bDMARDs.
Legend: remission (DAS-28 <2.6), LDA - low disease activity (DAS-28 ≤3.2), HAQ-DI≤0.25 (HAQ25) and minimum HAQ-DI-decrease (MCID_HAQ) since baseline of 0.22.
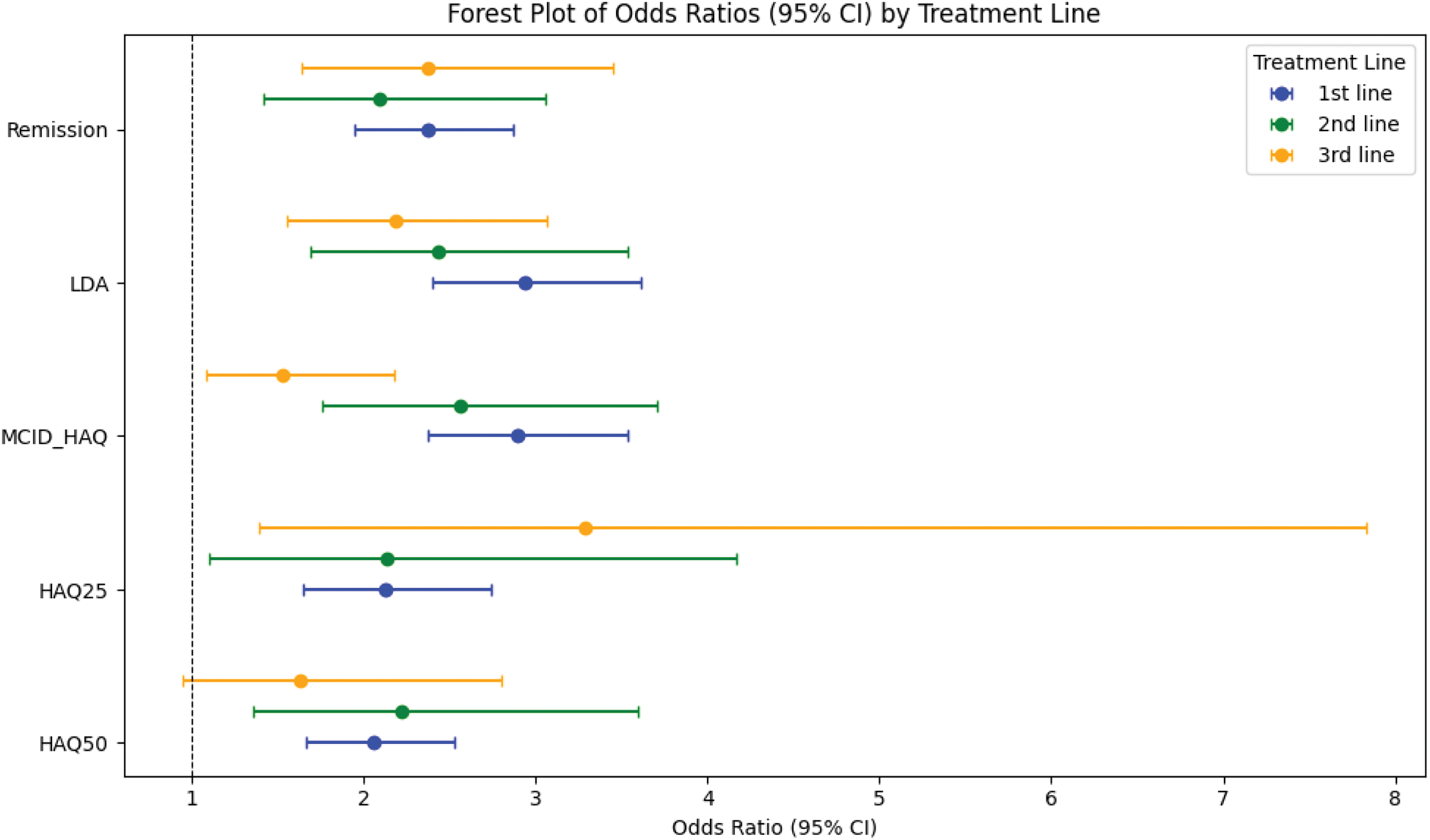
Conclusion: FIL treatment demonstrated in every treatment line improved clinical and functional outcomes compared to a contemporary bDMARD cohort in the short term among patients with moderately to severely active RA in a real-world setting. While adjusted analyses were employed, the potential influence of selection bias and timing of measurement cannot be excluded. Furthermore, the predominant use of DAS28CRP as the outcome measure may have biased remission rates in favour of FIL.
REFERENCES: NIL.
Acknowledgements: Galapagos/Alfasigma gave unrestricted support for this research.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (