

Background: Lupus nephritis (LN) is one of the most common and severe organ manifestations in patients with systemic lupus erythematosus (SLE). The risk factors for lupus nephritis had been reported. The uses of hydroxychloroquine and belimumab were associated with decreased risk of de novo LN flares in patients with non-renal diseases. The effects of B cell-suppressing therapy with rituximab on the lupus nephritis development remained to be elucidated.
Objectives: To evaluate the occurrence and risk factors of lupus nephritis flares in SLE patients treated with rituximab and azathioprine for non-renal causes.
Methods: We retrospectively collected SLE patients starting either rituximab or azathioprine for non-renal causes from a tertiary center in Taiwan. The baseline demographics, systemic lupus erythematosus disease activity index-2000 (SLEDAI-2K) score, serial laboratory reports, renal biopsy reports, and medication history were collected. The transient proteinuria was defined as one occasion of proteinuria > 0.5 g (or surrogated with urine protein-Patients with lupus nephritis within 2 years (defined as a persistent proteinuria > 0.5 g daily for 2 occasions or biopsy-proven lupus nephritis) before the start of rituximab or azathioprine, patients exposed to cyclophosphamide, mycophenolate mofetil, or belimumab at baseline or during the follow up, and incomplete rituximab and azathioprine administrations were excluded. The primary endpoint was the occurrence of lupus nephritis, defined by proteinuria > 0.5 g daily for 2 occasions or biopsy-proven lupus nephritis.
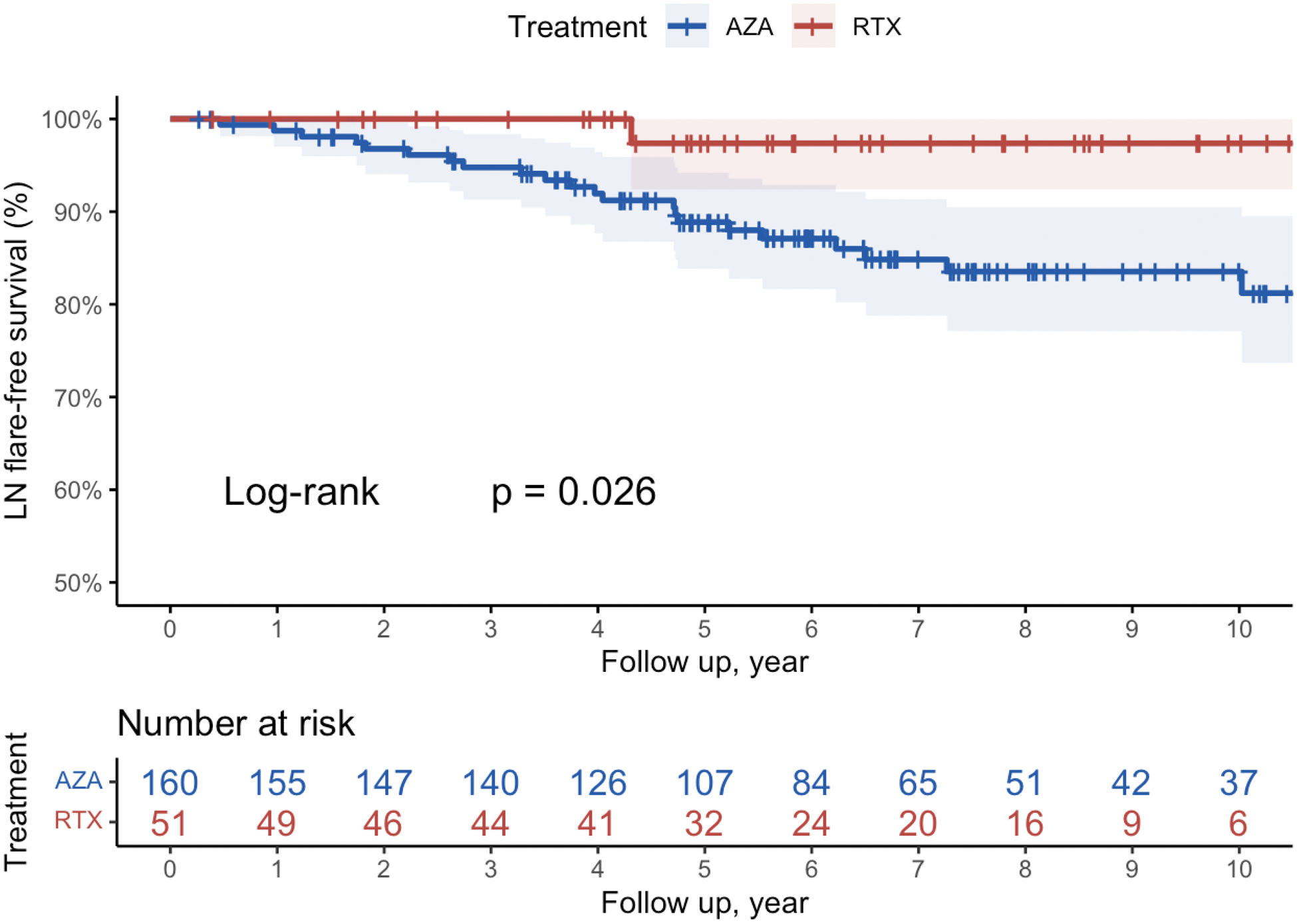
Results: From 2006 to 2023, 388 SLE patients receiving either rituximab and azathioprine for non-renal causes were identified, and 211 patients were enrolled into the final analysis. The median (IQR) age on enrollment was 37 (29-49) years and 89% were female. 97% patients took concomitant hydroxychloroquine, and all received oral glucocorticoids. The median cycle of rituximab course was 6.0 (3.0-9.5). Rituximab patients had higher baseline SLEDAI (10.0 vs. 7.5, p = 0.02) compared with those receiving azathioprine. 5 patients (9.8%) in rituximab group had transient proteinuria > 0.5 g/day before starting the therapy, compared with 9 (5.6%) in azathioprine group. At a median follow-up duration of 6.1 (4.3-8.8) years, one (2%) patient receiving rituximab patients developed lupus nephritis and 26 (16%) in azathioprine group (p = 0.026) (Figure 1). On multivariable and multivariable Cox regression analyses, the baseline SLEDAI score (HR: 1.13, 95% CI: 1.04-1.23, p = 0.004), history of transient proteinuria (HR: 5.87, 95% CI: 2.27-15.2, p < 0.001), were independent risk factors for subsequent LN flares. The use of rituximab (HR: 0.04, 95% CI: 0.00-0.36, p = 0.004) was associated with decreased risk for LN development (Table 1).
Conclusion: Compared with azathioprine, rituximab was associated with decreased rate of lupus nephritis flares in patients with non-renal SLE. Higher baseline SLEDAI and the history of transient proteinuria were independent risk factors for lupus nephritis development. The observations warrant further investigations on disease-modifying effects of rituximab and other immunomodulators.
REFERENCES: [1] Gomez A, Jägerback S, Sjöwall C, Parodis I. Belimumab and antimalarials combined against renal flares in patients treated for extra-renal systemic lupus erythematosus: results from 4 phase III clinical trials. Rheumatology (Oxford). 2024;63(2):338-348. doi:10.1093/rheumatology/kead253.
[2] Askanase AD, Furie RA, Dall’Era M, et al. Disease-modifying therapies in systemic lupus erythematosus for extrarenal manifestations. Lupus Sci Med. 2024;11(1):e001124. Published 2024 May 22. doi:10.1136/lupus-2023-001124.
Lupus nephritis flare-free survivals in patients treated with AZA or RTX.
Univariable and multivariable Cox regression of baseline predictors for LN flares.
| Risk factors | N | Univariable | Multivariable | ||||
|---|---|---|---|---|---|---|---|
| HR | 95% CI | p-value | HR | 95% CI | p-value | ||
| Treatment with RTX | 211 | 0.14 | 0.02-1.06 | 0.009 | 0.04 | 0.00-0.36 | 0.004 |
| SLEDAI score | 211 | 1.10 | 1.03-1.17 | 0.006 | 1.13 | 1.04-1.23 | 0.004 |
| Transient proteinuria | 211 | 6.04 | 2.52-14.5 | <0.001 | 5.87 | 2.27-15.2 | <0.001 |
Acknowledgements: NIL.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (