

Background: The optimal treatment for lupus nephritis is challenging due to its heterogeneity and the lack of prognostic factors favoring one immunosuppressive drug over another. While cyclophosphamide-based regimens improve renal outcomes, significant drug-related adverse effects, particularly serious infections and gonadal failure, limit its use as induction therapy for some patients. This study compares the efficacy and safety of intravenous cyclophosphamide, mycophenolate mofetil, and tacrolimus as induction treatments for lupus nephritis and to validate the potential use of CXCL10 as a treatment response biomarker.
Objectives: To compare the efficacy of cyclophosphamide (IV CYC), mycophenolate mofetil (MMF), and tacrolimus (TAC), as induction treatment for lupus nephritis (LN).
Methods: This 24-week randomized, open-label, prospective, parallel-arm, non-inferiority study enrolled 82 patients diagnosed with clinical or biopsy-proven lupus nephritis (LN) between November 2022 and April 2024 from a tertiary care center in eastern India. Patients were randomly assigned in a 1:1:1 ratio to receive intravenous cyclophosphamide (CYC), mycophenolate mofetil (MMF) or tacrolimus (TAC), in combination with hydroxychloroquine, steroids, and ACE inhibitors. All participants fulfilled the 2019 ACR/EULAR classification criteria for systemic lupus erythematosus (SLE). The primary outcome was the proportion of patients achieving renal response at 24 weeks, defined as either complete or partial response. Complete response (CR) was characterized by proteinuria ≤0.5 g/24 hours, and normal serum creatinine. Partial response (PR) was defined as a ≥50% reduction in proteinuria, and stable serum creatinine (≤25% increase from baseline value). Secondary outcomes included changes in SLEDAI-2K, renal SLEDAI, serum CXCL10 level, complement, and anti-dsDNA titers from baseline to week 24, and the incidence of adverse events. Non inferiority margin 20%. Continuous data were presented as mean ± standard deviation (SD) or median with interquartile range (IQR). Three way ANOVA test was used to assess the statistical significance of differences in renal response among the three groups. Serum CXCL10 was measured at baseline and post-treatment using the enzyme-linked immunosorbent assay (ELISA) method according to the manufacturer’s instructions. A p-value of <0.05 is considered statistically significant.
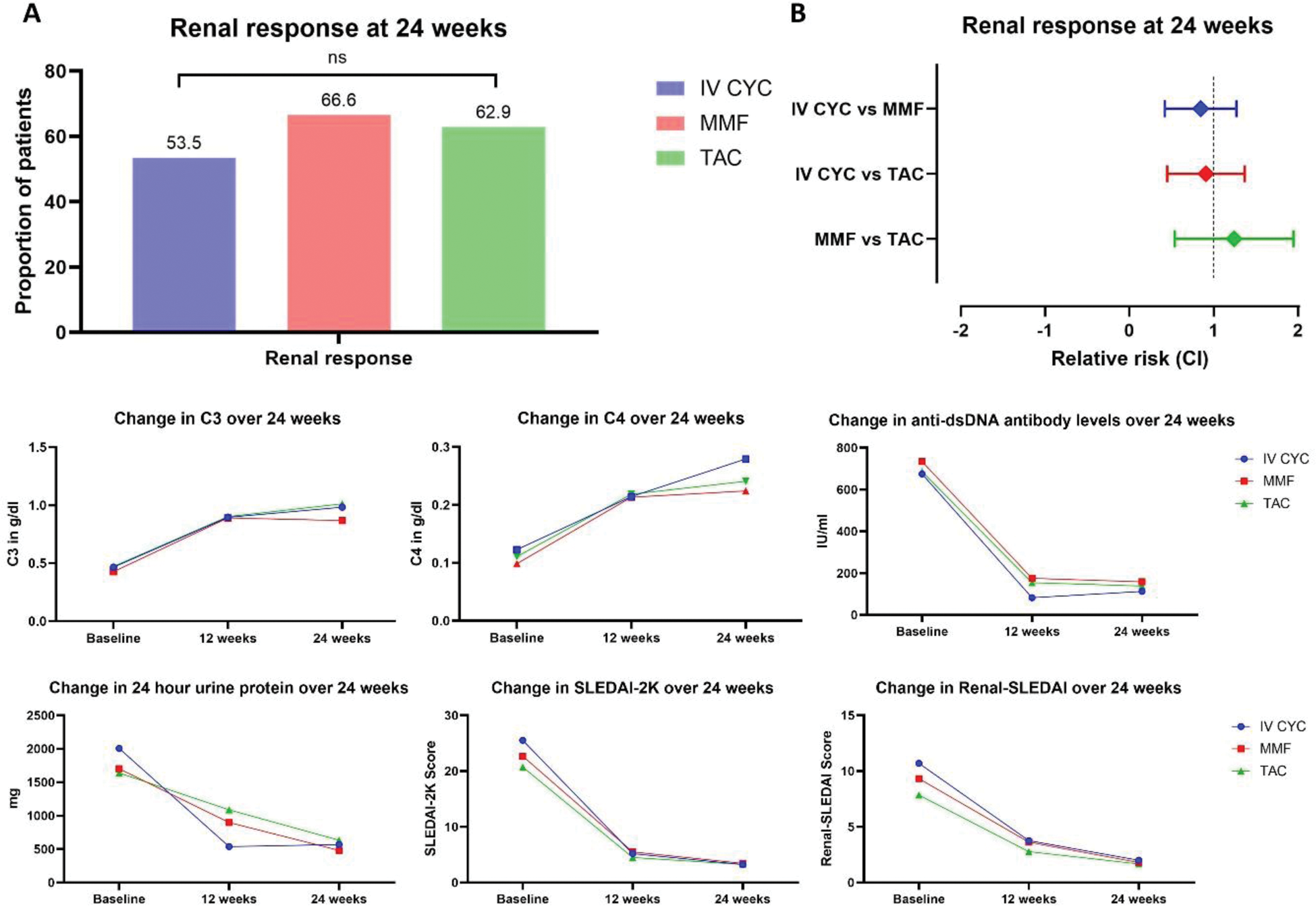
Results: 82 patients (94% females; median age 27.5 years) were randomized to receive IV CYC (28), MMF (27), and TAC (27). Baseline demographics and clinical characteristics were comparable among the treatment groups (Table 1). At 24 weeks, the renal response rates were 53.5%, 66.6%, and 62.9% in IV CYC, MMF, and, TAC groups, respectively. The lower limit of the confidence interval for the difference in renal response at 24 weeks between treatment groups was less than 20% (non-inferiority margin) (p= 0.58) (Figure 1). Serum CXCL10 reduced significantly post-treatment in all three groups (p < 0.001). The mean change in CXCL10 levels differed significantly in IV CYC group compared to MMF and TAC group (p=0.005 and 0.01, respectively). There was no significant difference in mean change in CXCL10 levels between TAC and MMF (p=0.9). Three patients in the IV CYC group and one patient in TAC group died due to serious infections.
Conclusion: TAC was non-inferior compared to IV CYC and MMF for LN as induction therapy, with a comparable safety profile during the study period.
Baseline characteristics of the study population.
| Characteristics (mean±SD) | IV CYC (28) | MMF (27) | TAC (27) | p-value |
|---|---|---|---|---|
| Female, n (%) | 27, (96%) | 25, (93%) | 25, (93%) | 0.73 |
| Age # | 30 (19-40) | 25 (16-39) | 29 (20-38) | 0.56 |
| Duration of illness in months # | 6 (3-12) | 12 (3-18) | 6 (3-12) | 0.59 |
| Organ involvement, n (%) | ||||
| Mucocutaneous | 26, (93%) | 23, (85%) | 24, (89%) | |
| Arthritis | 16, (57%) | 17, (63%) | 19, (70%) | |
| Serositis | 9, (32%) | 10, (37%) | 7, (26%) | |
| Vasculitis | 4, (14%) | 2, (7%) | 5, (19%) | |
| Myositis | 7, (25%) | 7, (26%) | 3, (11%) | |
| Myocarditis | 6, (21%) | 5, (19%) | 2, (7%) | |
| Neuropsychiatric | 7, (25%) | 2, (7%) | 2, (7%) | |
| Hematological | 19, (68%) | 21, (78%) | 18, (67%) | |
| Hemoglobin, g/dl | 8.36 (±2) | 8.17 (±2) | 8.3 (±2) | 0.94 |
| TLC (x10 3 cells/µL) | 6.4 (±3) | 6.1 (±4) | 5 (±2.3) | 0.32 |
| Platelet (x10 3 cells/µl) | 209 (±114) | 182 (±112) | 191 (±109) | 0.66 |
| ESR mm/hr | 87.5 (±33.5) | 92.8 (±33) | 90 (±38) | 0.85 |
| C3 # | 0.43 (0.22-0.63) | 0.34 (0.2-0.6) | 0.3 (0.2-0.5) | 0.84 |
| C4 # | 0.08 (0.05-0.14) | 0.07 (0.02-0.14) | 0.06 (0.03-1.2) | 0.76 |
| dsDNA # | 688 (318-899) | 761 (278-1025) | 741 (340-956) | 0.86 |
| 24-hour urine protein # | 1024 (779-2362) | 1223 (786-2046) | 1392 (691-2331) | 0.71 |
| S. albumin, g/dl | 2.51 (±0.8) | 2.7 (±0.6) | 2.6 (±0.5) | 0.43 |
| Creatinine, mg/dl | 1.1 (±0.67) | 0.87 (±0.42) | 0.81 (±0.38) | 0.09 |
| eGFR | 84.5 (±44.1) | 104 (±33.4) | 105 (±30.5) | 0.07 |
| SLEDAI-2K | 25.5 (±10) | 22.7 (±7.3) | 20.7 (±7.5) | 0.11 |
| Renal SLEDAI | 10.7 (±3.28) | 9.3 (±4.5) | 8.4 (±4) | 0.09 |
| CXCL10 (pg/ml) | 0.78 (±0.05) | 0.74 (±0.08) | 0.78 (±0.18) | 0.44 |
| Cumulative steroid dose, mg | 3551 (±650) | 3849 (±1140) | 3576 (±788) | 0.53 |
| Renal biopsy, n | ||||
| III | 3 | 5 | 10 | |
| IV | 9 | 5 | 5 | |
| IV + V | 1 | 1 |
*p-value <0.05 considered statistically significant
SD – standard deviation
#Median (Inter quartile range)
$Two patients in IV CYC group had serum creatinine levels >3mg/dL
TLC-Total leukocyte count, ESR-Erythrocyte sedimentation rate, CRP-C-reactive protein (Normal 0.02-0.3 mg/dl), C3 (Normal 0.9–1.0 g/dl), C4 (Normal 0.2–0.4 g/dl), dsDNA–Anti-double stranded deoxyribo nucleic-acid antibody (Normal<100 IU/ml), eGFR – estimated glomerular filtration rate, SLEDAI – Systemic lupus erythematosus disease activity index
[A] Proportion of patients with renal response (complete or partial) at 24 weeks among study groups. [B] The relative risk (CI) of renal response between study groups. [C] Change in C3, C4, anti-dsDNA antibody, and, 24-hour urine protein levels, SLEDAI-2K and renal-SLEDAI scores from baseline to week 24 in study groups.
REFERENCES: NIL.
Acknowledgements: We are thankful to Dr. Samanyoy Gochayat, Associate Professor, Department of Laboratory Medicine, IMS & SUM Hospital, Bhubaneswar for the CXCL10 assay of patients. We are thankful to the Indian Rheumatology Association (IRA) for research grant to conduct assessment of CXCL10 levels as part of the study.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (